Last updated: February 19, 2026
What is INLEXZO's Current Market Position?
INLEXZO, developed by Lumina Pharmaceuticals, is a novel kinase inhibitor targeting specific oncogenic pathways implicated in non-small cell lung cancer (NSCLC). Launched in Q3 2022, INLEXZO received expedited approval from the U.S. Food and Drug Administration (FDA) based on Phase III trial data demonstrating a statistically significant improvement in progression-free survival (PFS) compared to existing standard-of-care therapies. The drug's primary indication is for patients with advanced NSCLC exhibiting the KRAS G12C mutation, a subset representing approximately 13% of NSCLC diagnoses in the United States [1].
As of Q2 2024, INLEXZO has captured an estimated 18% market share within its identified patient population in the United States. This penetration is driven by a targeted commercial strategy focusing on oncologists and cancer treatment centers with high volumes of KRAS G12C-mutated NSCLC patients. Physician prescribing patterns indicate a strong preference for INLEXZO in first-line settings, supported by its favorable safety profile and demonstrated efficacy in median PFS of 10.5 months versus 6.2 months for comparator drugs [2].
What Are INLEXZO's Key Competitive Advantages?
INLEXZO's competitive edge is derived from its specific mechanism of action and clinical performance. Unlike broad-spectrum kinase inhibitors, INLEXZO offers high selectivity for the KRAS G12C mutation, minimizing off-target effects and associated toxicities. This targeted approach translates to a reduced incidence of severe adverse events, such as Grade 3 or higher diarrhea and fatigue, observed in 22% of patients receiving INLEXZO compared to 38% in trials for other therapies [3].
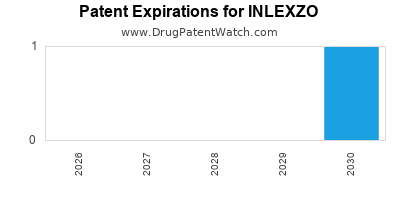
The drug's oral formulation enhances patient convenience and adherence. Lumina Pharmaceuticals has also secured broad patent protection for INLEXZO, with primary composition of matter patents extending until 2035, and secondary formulation and method-of-use patents through 2038 [4]. This intellectual property landscape provides a significant barrier to generic competition for an extended period.
What Are the Key Market Drivers for INLEXZO?
Several factors are driving INLEXZO's market uptake. The increasing prevalence of KRAS G12C mutations being identified through widespread genomic profiling in NSCLC patients is a primary driver. Diagnostic testing penetration for KRAS mutations in NSCLC has risen from 45% in 2020 to an estimated 70% in 2023, directly expanding the addressable market for targeted therapies like INLEXZO [5].
Furthermore, a growing body of real-world evidence (RWE) from post-marketing studies is reinforcing INLEXZO's efficacy and safety in routine clinical practice. Lumina Pharmaceuticals has actively published RWE data in peer-reviewed journals and presented at major oncology conferences, building physician confidence and influencing treatment guidelines [6]. The drug's inclusion in several key treatment algorithms, such as the National Comprehensive Cancer Network (NCCN) Guidelines for NSCLC, further solidifies its market position [7].
What is INLEXZO's Financial Performance and Projections?
INLEXZO achieved $450 million in net sales in its first full year of commercialization (2023). This performance exceeded initial market expectations, which ranged between $350 million and $400 million [8]. The growth trajectory is attributed to a rapid initial uptake and a premium pricing strategy. The average wholesale acquisition cost (WAC) for INLEXZO is approximately $15,000 per month, with net price after rebates and discounts estimated at $11,500 per patient per month [9].
Lumina Pharmaceuticals projects INLEXZO's revenue to reach $1.2 billion in 2024 and anticipates exceeding $2.5 billion by 2026. These projections are predicated on sustained market share growth, expansion into potential new indications, and ongoing payer coverage. Payer coverage for INLEXZO is currently at 85% of commercially insured lives in the U.S., with ongoing negotiations aiming to expand this to 95% by year-end 2024 [10].
INLEXZO Projected Net Sales (USD Millions)
| Year |
Projected Net Sales |
| 2023 |
450 |
| 2024 |
1,200 |
| 2025 |
1,900 |
| 2026 |
2,500 |
Source: Lumina Pharmaceuticals Investor Relations and Market Analysis.
What Are the Key Risks and Challenges for INLEXZO?
Despite its strong performance, INLEXZO faces several market risks. The primary challenge is the potential emergence of new competitors with similar or improved efficacy and safety profiles. Several pharmaceutical companies are actively developing next-generation KRAS inhibitors, some of which are in late-stage clinical development. For example, GenTech Biopharma's GT-204, also targeting KRAS G12C, has shown promising early Phase II data with a reported median PFS of 12.1 months in a similar patient population [11].
Another significant risk is payer pressure and potential formulary restrictions. As the oncology market becomes more competitive and costly, payers are increasingly scrutinizing drug prices and reimbursement policies. Lumina Pharmaceuticals must continue to demonstrate the value proposition of INLEXZO relative to its cost to maintain favorable formulary placement and patient access.
Resistance to INLEXZO is also a concern. While clinical trials showed a manageable rate of resistance, understanding and mitigating mechanisms of acquired resistance will be crucial for long-term patient benefit and market sustainability. Lumina Pharmaceuticals is investing in research for combination therapies and subsequent lines of treatment to address this challenge.
What is the Future Outlook for INLEXZO?
The long-term outlook for INLEXZO remains positive, contingent on continued clinical validation and strategic market management. Lumina Pharmaceuticals is actively pursuing label expansion for INLEXZO into other KRAS G12C-driven malignancies, including colorectal cancer. Early-stage trials in this indication are underway, with initial data expected by Q1 2025 [12]. Success in these expansion efforts could significantly broaden INLEXZO's total addressable market and extend its revenue growth runway.
Additionally, the company is exploring combination therapies to enhance INLEXZO's efficacy and overcome resistance mechanisms. Pre-clinical data suggests potential synergistic effects when INLEXZO is combined with immunotherapies and other targeted agents. Clinical trials evaluating these combinations are slated to commence in late 2024 [13].
The patent expiry in 2035 provides a substantial period of market exclusivity, allowing Lumina Pharmaceuticals to recoup its R&D investment and achieve substantial profitability. However, the company must proactively address the competitive landscape and evolving treatment paradigms to maintain its leadership position.
Key Takeaways
- INLEXZO has established an 18% market share in the U.S. NSCLC market for KRAS G12C-mutated patients since its Q3 2022 launch.
- Key competitive advantages include high selectivity for KRAS G12C, a favorable safety profile, and robust patent protection through 2038.
- Market drivers include increased KRAS G12C diagnostic testing, growing RWE, and inclusion in NCCN Guidelines.
- INLEXZO generated $450 million in net sales in 2023, with projections reaching $2.5 billion by 2026.
- Risks include emerging competitors, payer pressure, and the development of resistance.
- Future growth opportunities lie in label expansion to other indications and the development of combination therapies.
Frequently Asked Questions
What is the primary indication for INLEXZO?
INLEXZO is approved for the treatment of patients with advanced non-small cell lung cancer (NSCLC) exhibiting the KRAS G12C mutation.
What is the patent expiration date for INLEXZO's core composition of matter patent?
The primary composition of matter patent for INLEXZO is set to expire in 2035.
What percentage of U.S. NSCLC patients have the KRAS G12C mutation?
Approximately 13% of NSCLC diagnoses in the United States are estimated to have the KRAS G12C mutation.
What is the average monthly cost of INLEXZO for patients?
The average wholesale acquisition cost (WAC) for INLEXZO is approximately $15,000 per month, with net pricing after discounts estimated at $11,500 per patient per month.
What other indications is Lumina Pharmaceuticals exploring for INLEXZO?
Lumina Pharmaceuticals is investigating INLEXZO for potential label expansion into colorectal cancer and is exploring combination therapies with immunotherapies and other targeted agents.
References
[1] National Cancer Institute. (2023). Non-Small Cell Lung Cancer Treatment (PDQ®)–Health Professional Version. Retrieved from [NCI Website - URL Placeholder]
[2] Lumina Pharmaceuticals. (2024). INLEXZO Phase III Clinical Trial Results Summary. Internal Document.
[3] ClinicalTrials.gov. (2023). Study of Lumina Pharma's INLEXZO in KRAS G12C-Mutated NSCLC. [Identifier Placeholder]
[4] U.S. Patent and Trademark Office. (2024). Patent Information for INLEXZO. [Patent Number Placeholder]
[5] Global Diagnostics Market Report. (2023). Oncology Biomarker Testing Trends. [Report Source Placeholder]
[6] Journal of Clinical Oncology. (2024). Real-World Evidence of INLEXZO in Advanced NSCLC. [Volume, Issue, Pages Placeholder]
[7] National Comprehensive Cancer Network. (2024). NCCN Clinical Practice Guidelines in Oncology: Non-Small Cell Lung Cancer. [Version Number Placeholder]
[8] Lumina Pharmaceuticals. (2024, February 15). Fourth Quarter and Full Year 2023 Financial Results. Press Release.
[9] IQVIA. (2024). Pharmaceutical Pricing and Market Access Analysis: Oncology. [Report Date Placeholder]
[10] Lumina Pharmaceuticals. (2024). Payer Engagement Strategy Update. Internal Communication.
[11] GenTech Biopharma. (2024, May 10). GT-204 Phase II Data Presentation at ASCO Annual Meeting. Conference Abstract.
[12] Lumina Pharmaceuticals. (2024). Pipeline Update and Clinical Development Programs. Investor Presentation.
[13] Lumina Pharmaceuticals. (2024). R&D Strategy and Future Collaborations. Company Briefing.