ILEVRO Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Ilevro, and what generic alternatives are available?
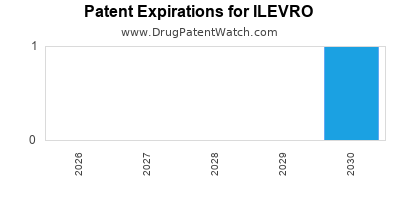
Ilevro is a drug marketed by Harrow Eye and is included in one NDA. There are two patents protecting this drug and one Paragraph IV challenge.
This drug has fifty-five patent family members in twenty-seven countries.
The generic ingredient in ILEVRO is nepafenac. There are eight drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the nepafenac profile page.
DrugPatentWatch® Generic Entry Outlook for Ilevro
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be March 31, 2032. This may change due to patent challenges or generic licensing.
There have been two patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for ILEVRO?
- What are the global sales for ILEVRO?
- What is Average Wholesale Price for ILEVRO?
Summary for ILEVRO
| International Patents: | 55 |
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 93 |
| Clinical Trials: | 6 |
| Patent Applications: | 2,905 |
| Drug Prices: | Drug price information for ILEVRO |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for ILEVRO |
| What excipients (inactive ingredients) are in ILEVRO? | ILEVRO excipients list |
| DailyMed Link: | ILEVRO at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for ILEVRO
Generic Entry Date for ILEVRO*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
SUSPENSION/DROPS;OPHTHALMIC |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for ILEVRO
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Research Insight LLC | PHASE4 |
| Vance Thompson Vision - MT | Phase 4 |
| Alcon Research | N/A |
Pharmacology for ILEVRO
| Drug Class | Nonsteroidal Anti-inflammatory Drug |
| Mechanism of Action | Cyclooxygenase Inhibitors |
US Patents and Regulatory Information for ILEVRO
ILEVRO is protected by two US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of ILEVRO is ⤷ Start Trial.
This potential generic entry date is based on patent 8,921,337.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Harrow Eye | ILEVRO | nepafenac | SUSPENSION/DROPS;OPHTHALMIC | 203491-001 | Oct 16, 2012 | RX | Yes | Yes | 9,662,398 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Harrow Eye | ILEVRO | nepafenac | SUSPENSION/DROPS;OPHTHALMIC | 203491-001 | Oct 16, 2012 | RX | Yes | Yes | 8,921,337 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for ILEVRO
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Harrow Eye | ILEVRO | nepafenac | SUSPENSION/DROPS;OPHTHALMIC | 203491-001 | Oct 16, 2012 | 7,947,295 | ⤷ Start Trial |
| Harrow Eye | ILEVRO | nepafenac | SUSPENSION/DROPS;OPHTHALMIC | 203491-001 | Oct 16, 2012 | 5,475,034 | ⤷ Start Trial |
| Harrow Eye | ILEVRO | nepafenac | SUSPENSION/DROPS;OPHTHALMIC | 203491-001 | Oct 16, 2012 | 6,403,609 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for ILEVRO
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Novartis Europharm Limited | Nevanac | nepafenac | EMEA/H/C/000818Nevanac is indicated for:, , , prevention and treatment of postoperative pain and inflammation associated with cataract surgery;, reduction in the risk of postoperative macular oedema associated with cataract surgery in diabetic patients., , | Authorised | no | no | no | 2007-12-11 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for ILEVRO
When does loss-of-exclusivity occur for ILEVRO?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 0572
Patent: SUSPENSIONES DE NANOPARTICULAS QUE CONTIENEN POLIMERO DE CARBOXIVINILO
Estimated Expiration: ⤷ Start Trial
Patent: 2463
Patent: SUSPENSIONES DE NANOPARTÍCULAS QUE CONTIENEN POLÍMERO DE CARBOXIVINILO
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 10326099
Patent: Carboxyvinyl polymer-containing nanoparticle suspension
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2012013503
Patent: suspensões de nanopartícula contendo polímero de carboxivinila
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 81254
Patent: SUSPENSION DE NANOPARTICULES CONTENANT UN POLYMERE DE TYPE CARBOXYVINYLE (CARBOXYVINYL POLYMER-CONTAINING NANOPARTICLE SUSPENSION)
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 12001402
Patent: Composicion farmaceutica en suspension oftalmica acuosa de administracion topica que comprende polimero de carboxivinilo, galactomanana, borato, compuesto en nanoparticulas escasamente soluble, agentes para ajustar ph y tonicidad, conservante y quelante; metodo para tratar trastornos oftalmicos, metodo para mantener viscosidad.
Estimated Expiration: ⤷ Start Trial
China
Patent: 2724965
Patent: Carboxyvinyl polymer-containing nanoparticle suspensions
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0140016
Estimated Expiration: ⤷ Start Trial
Patent: 0181337
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 14871
Estimated Expiration: ⤷ Start Trial
Patent: 20585
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 06831
Estimated Expiration: ⤷ Start Trial
Patent: 86426
Estimated Expiration: ⤷ Start Trial
Patent: 65749
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 06831
Patent: SUSPENSION DE NANOPARTICULES CONTENANT UN POLYMÈRE DE TYPE CARBOXYVINYLE (CARBOXYVINYL POLYMER-CONTAINING NANOPARTICLE SUSPENSIONS)
Estimated Expiration: ⤷ Start Trial
Patent: 86426
Patent: Suspension de nanoparticules contenant un polymère de type carboxyvinyle (Carboxyvinyl polymer-containing nanoparticle suspensions)
Estimated Expiration: ⤷ Start Trial
Patent: 65749
Patent: SUSPENSIONS DE NANOPARTICULES CONTENANT UN POLYMÈRE CARBOXYVINYLIQUE (CARBOXYVINYL POLYMER-CONTAINING NANOPARTICLE SUSPENSIONS)
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 70164
Patent: CARBOXYVINYL POLYMER-CONTAINING NANOPARTICLE SUSPENSIONS
Estimated Expiration: ⤷ Start Trial
Patent: 78802
Patent: 含毫微顆粒懸浮體的羧基乙烯聚合物 (CARBOXYVINYL POLYMER-CONTAINING NANOPARTICLE SUSPENSIONS)
Estimated Expiration: ⤷ Start Trial
Patent: 17091
Patent: 含羧乙烯基聚合物的納米顆粒混懸液 (CARBOXYVINYL POLYMER-CONTAINING NANOPARTICLE SUSPENSIONS)
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 25838
Estimated Expiration: ⤷ Start Trial
Patent: 38821
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 64433
Estimated Expiration: ⤷ Start Trial
Patent: 55849
Estimated Expiration: ⤷ Start Trial
Patent: 13512912
Estimated Expiration: ⤷ Start Trial
Patent: 15061886
Patent: カルボキシビニルポリマー含有ナノ粒子懸濁物 (CARBOXYVINYL POLYMER-CONTAINING NANOPARTICLE SUSPENSION)
Estimated Expiration: ⤷ Start Trial
Patent: 16106155
Patent: カルボキシビニルポリマー含有ナノ粒子懸濁物 (CARBOXYVINYL POLYMER-CONTAINING NANOPARTICLE SUSPENSIONS)
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 65749
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 12006231
Patent: SUSPENSION DE NANOPARTICULAS QUE CONTIENEN POLIMERO DE CARBOXIVINILO. (CARBOXYVINYL POLYMER-CONTAINING NANOPARTICLE SUSPENSION.)
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 012501111
Patent: CARBOXYVINYL POLYMER-CONTAINING NANOPARTICLES SUSPENSIONS
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 06831
Estimated Expiration: ⤷ Start Trial
Patent: 86426
Estimated Expiration: ⤷ Start Trial
Patent: 65749
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 06831
Estimated Expiration: ⤷ Start Trial
Patent: 86426
Estimated Expiration: ⤷ Start Trial
Patent: 65749
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 71078
Patent: СУСПЕНЗИИ НАНОЧАСТИЦ, СОДЕРЖАЩИЕ КАРБОКСИВИНИЛОВЫЙ ПОЛИМЕР (SUSPENSIONS OF NANOPARTICLES, CONTAINING CARBOXYVINYL POLYMER)
Estimated Expiration: ⤷ Start Trial
Patent: 12127675
Patent: СУСПЕНЗИИ НАНОЧАСТИЦ, СОДЕРЖАЩИЕ КАРБОКСИВИНИЛОВЫЙ ПОЛИМЕР
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 085
Patent: SUSPENZIJE NANOČESTICA KOJE SADRŽE KARBOKSIVINILNI POLIMER (CARBOXYVINYL POLYMER-CONTAINING NANOPARTICLE SUSPENSION)
Estimated Expiration: ⤷ Start Trial
Patent: 465
Patent: NANOČESTIČNE SUSPENZIJE KOJE SADRŽE KARBOKSIVINIL POLIMER (CARBOXYVINYL POLYMER-CONTAINING NANOPARTICLE SUSPENSIONS)
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 06831
Estimated Expiration: ⤷ Start Trial
Patent: 86426
Estimated Expiration: ⤷ Start Trial
Patent: 65749
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1203770
Patent: CARBOXYVINYL POLYMER-CONTAINING NANOPARTICLE SUSPENSIONS
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1543613
Estimated Expiration: ⤷ Start Trial
Patent: 1809484
Estimated Expiration: ⤷ Start Trial
Patent: 120099269
Patent: CARBOXYVINYL POLYMER- CONTAINING NANOPARTICLE SUSPENSION
Estimated Expiration: ⤷ Start Trial
Patent: 150063587
Patent: 카복시비닐 폴리머를 포함하는 나노입자 현탁액 (CARBOXYVINYL POLYMER-CONTAINING NANOPARTICLE SUSPENSIONS)
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 41420
Estimated Expiration: ⤷ Start Trial
Patent: 50942
Estimated Expiration: ⤷ Start Trial
Patent: 84752
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 1129395
Patent: Carboxyvinyl polymer-containing nanoparticle suspensions
Estimated Expiration: ⤷ Start Trial
Patent: 86178
Estimated Expiration: ⤷ Start Trial
Uruguay
Patent: 081
Patent: SUSPENSIONES DE NANOPARTÍCULAS QUE CONTIENEN POLÍMEROS DE CARBOXIVINILO
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering ILEVRO around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Hungary | E038821 | ⤷ Start Trial | |
| Hong Kong | 1170164 | CARBOXYVINYL POLYMER-CONTAINING NANOPARTICLE SUSPENSIONS | ⤷ Start Trial |
| Russian Federation | 2571078 | СУСПЕНЗИИ НАНОЧАСТИЦ, СОДЕРЖАЩИЕ КАРБОКСИВИНИЛОВЫЙ ПОЛИМЕР (SUSPENSIONS OF NANOPARTICLES, CONTAINING CARBOXYVINYL POLYMER) | ⤷ Start Trial |
| Denmark | 2506831 | ⤷ Start Trial | |
| Serbia | 53085 | SUSPENZIJE NANOČESTICA KOJE SADRŽE KARBOKSIVINILNI POLIMER (CARBOXYVINYL POLYMER-CONTAINING NANOPARTICLE SUSPENSION) | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ILEVRO
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0999825 | 122013000085 | Germany | ⤷ Start Trial | PRODUCT NAME: NEPAFENAC (OPHTHALMISCHE SUSPENSION); REGISTRATION NO/DATE: EU 1/07/433/002 20130503 |
| 0999825 | C300622 | Netherlands | ⤷ Start Trial | PRODUCT NAME: NEPAFENAC; REGISTRATION NO/DATE: EU/1/07/433/002 20130503 |
| 0999825 | CA 2013 00055 | Denmark | ⤷ Start Trial | PRODUCT NAME: NEPAFENAC (3 MG/ML), HERUNDER NEPAFENAC I KOMBINATION MED GLACTOMANNANPOLYMERER, ISAER 3 MG/ML NEPAFENAC I KOMBINATION MED GALACTOMANNANPOLYMERER, SAMT OFTALMISKE SAMMENSAETNINGER DERAF; REG. NO/DATE: EU1/07/433/002 20130503 |
| 0716600 | C00716600/01 | Switzerland | ⤷ Start Trial | PRODUCT NAME: NEPAFENAC; REGISTRATION NUMBER/DATE: SWISSMEDIC 58745 24.09.2008 |
| 0999825 | 92301 | Luxembourg | ⤷ Start Trial | PRODUCT NAME: NEPAFENAC-SUSPENSION OPHTALMIQUE |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
ILEVRO: Patent Landscape, Market Dynamics, and Financial Outlook
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.