Last updated: February 19, 2026
Hydroxyzine pamoate is an antihistamine with anxiolytic properties, primarily used for treating anxiety, tension, pruritus, and as a pre-operative sedative. Its market trajectory is influenced by patent expirations, generic competition, evolving treatment guidelines, and the emergence of novel therapies.
What is the Current Market Size and Projected Growth for Hydroxyzine Pamoate?
The global market for hydroxyzine pamoate is a mature segment within the antihistamine and anxiolytic categories. Precise current market size figures are not readily available as it is often aggregated within broader drug class reports. However, based on its established use and generic availability, the market is characterized by stable, albeit low, single-digit growth, primarily driven by its cost-effectiveness and continued prescription for specific indications.
- Estimated Annual Revenue: While specific recent figures are proprietary, industry estimates place the global market for hydroxyzine pamoate in the tens to low hundreds of millions of USD annually. This is significantly smaller than blockbuster drug markets.
- Growth Rate: The projected annual growth rate is anticipated to be between 1% and 3%. This growth is sustained by its entrenched position in certain treatment protocols and its affordability compared to newer, branded alternatives.
- Key Market Drivers:
- Cost-effectiveness: As a widely available generic, hydroxyzine pamoate is a cost-effective option for patients and healthcare systems, particularly in developing markets.
- Established Efficacy: Decades of clinical use have validated its efficacy for its approved indications, ensuring continued physician confidence.
- Pediatric Use: Its palatability in liquid suspension form contributes to its use in pediatric populations for pruritus and sedation.
- Market Restraints:
- Sedation Side Effects: Significant drowsiness can limit its use in patients requiring alertness.
- Cardiovascular Risks: Potential for QT interval prolongation, though rare, requires careful patient selection and monitoring.
- Emergence of Newer Therapies: Development of benzodiazepine alternatives, SSRIs, and SNRIs for anxiety disorders offers broader efficacy and potentially fewer side effects, diverting market share.
- Limited Efficacy for Severe Conditions: It is not a first-line treatment for severe anxiety disorders or major depressive disorder.
What is the Patent Landscape and Generic Competition for Hydroxyzine Pamoate?
Hydroxyzine pamoate's original patents have long expired, paving the way for extensive generic competition. This has fundamentally shaped its market economics.
- Original Patent Expiration: The foundational patents for hydroxyzine pamoate have expired decades ago, with the last significant protection likely lapsing in the late 20th century.
- Generic Market Dominance: The market is overwhelmingly dominated by generic manufacturers. This leads to intense price competition and low profit margins for individual manufacturers.
- Key Generic Players: Numerous pharmaceutical companies produce generic hydroxyzine pamoate. Prominent manufacturers include Teva Pharmaceuticals, Mylan (now Viatris), Sandoz (Novartis), and numerous smaller regional players.
- Impact of Generic Entry:
- Price Erosion: The introduction of generics led to a rapid and significant decline in the drug's price.
- Increased Accessibility: Generic availability has made the drug accessible to a wider patient base.
- Limited R&D Investment: The low profit margins discourage significant investment in new formulations or novel indications by generic manufacturers.
- Formulations: Hydroxyzine pamoate is primarily available in oral suspension and capsule forms. The oral suspension is particularly important for pediatric and geriatric patients.
- Intellectual Property (IP) Focus: While the core composition patent is expired, companies might still hold patents on specific novel formulations, manufacturing processes, or combinations that could offer limited market exclusivity, but these are generally not disruptive to the established generic market.
How Do Regulatory Policies and Approvals Impact Hydroxyzine Pamoate?
Regulatory bodies play a crucial role in ensuring the safety and efficacy of hydroxyzine pamoate, influencing its market access and prescriber confidence.
- FDA Approval: Hydroxyzine pamoate is approved by the U.S. Food and Drug Administration (FDA) for the symptomatic relief of anxiety and tension, pruritus due to allergic conditions, and as a pre-operative and post-operative sedative. The original New Drug Application (NDA) and subsequent approvals have long been established.
- ANDA Pathway: Generic versions are approved through the Abbreviated New Drug Application (ANDA) pathway, requiring demonstration of bioequivalence to the reference listed drug.
- Manufacturing Standards: Manufacturers must adhere to Current Good Manufacturing Practices (cGMP) as enforced by regulatory agencies worldwide. This ensures product quality, purity, and consistency.
- Pharmacovigilance: Post-market surveillance by regulatory agencies monitors for adverse events and safety signals. Any significant new safety concerns could lead to updated labeling requirements, risk mitigation strategies, or, in rare cases, market withdrawal.
- Labeling Updates: Regulatory agencies can mandate label updates to reflect new safety information, contraindications, or warnings. For hydroxyzine, this includes warnings regarding potential QT interval prolongation and significant central nervous system (CNS) depression.
- Off-Label Prescribing: While approved for specific indications, off-label use in areas such as nausea and vomiting can contribute to its market demand, though this is not directly supported by regulatory approval.
- European Medicines Agency (EMA) and Other International Bodies: Similar regulatory frameworks exist in other major markets, with EMA approval being critical for European market access.
What are the Key Therapeutic Applications and Their Market Share Contribution?
Hydroxyzine pamoate serves a defined set of therapeutic niches, with its market share distribution reflecting the prevalence of these conditions and the availability of alternative treatments.
- Pruritus (Itching): This is a significant indication, particularly for allergic skin conditions like eczema and urticaria. Its antihistaminic properties make it effective in reducing itching and associated inflammation.
- Market Contribution: Substantial, especially in over-the-counter (OTC) preparations (though hydroxyzine pamoate itself is typically prescription-only in many regions, its hydrochloride salt is widely OTC). This indication remains a stable driver.
- Anxiety and Tension: Hydroxyzine pamoate possesses anxiolytic properties due to its CNS depressant effects, acting on histamine H1 receptors and potentially other neurotransmitter systems.
- Market Contribution: Moderate. While historically used for mild to moderate anxiety, newer agents like SSRIs and SNRIs are now often preferred for chronic or severe anxiety disorders due to more targeted mechanisms and potentially better safety profiles for long-term use. It remains an option for short-term situational anxiety or when other agents are contraindicated.
- Sedation (Pre-operative/Post-operative): Its sedative effects make it useful for calming patients before surgery and aiding in post-operative recovery and management of post-anesthetic nausea and vomiting.
- Market Contribution: Moderate. It competes with benzodiazepines and other sedatives in this space. Its advantage lies in its perceived lower abuse potential compared to benzodiazepines in some clinical contexts.
- Other Uses (Less Significant):
- Nausea and Vomiting: Off-label use, particularly post-operatively.
- Motion Sickness: Limited use.
- Competitive Landscape within Indications:
- Pruritus: Competes with other antihistamines (e.g., cetirizine, loratadine), topical corticosteroids, and emollients.
- Anxiety: Competes with benzodiazepines (e.g., alprazolam, lorazepam), SSRIs (e.g., fluoxetine, sertraline), and SNRIs (e.g., venlafaxine, duloxetine).
- Sedation: Competes with benzodiazepines (e.g., midazolam, propofol), barbiturates, and other sedative-hypnotics.
What are the Key Financial Trends and Profitability Factors?
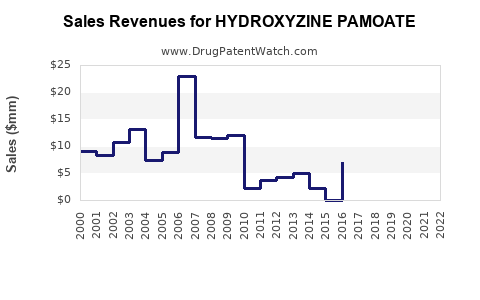
The financial landscape for hydroxyzine pamoate is characterized by the realities of a mature generic drug market, emphasizing volume and cost efficiency over high margins.
- Low Profit Margins: Due to intense generic competition and price controls in many healthcare systems, profit margins per unit are very low.
- Volume-Driven Revenue: Manufacturers rely on high sales volumes to generate meaningful revenue.
- Cost of Goods Sold (COGS): Efficient manufacturing and supply chain management are critical to maintaining profitability. Active Pharmaceutical Ingredient (API) sourcing and production costs are key variables.
- Distribution Channels: Sales are primarily through wholesale distributors to pharmacies, hospitals, and clinics. Direct-to-consumer marketing is minimal for prescription generics.
- Reimbursement: Coverage by insurance plans and government healthcare programs is widespread, contributing to consistent demand. However, payer policies often favor the lowest-cost generics.
- R&D Investment: For most generic manufacturers, R&D investment related to hydroxyzine pamoate is minimal, focused on process improvements or bioequivalence studies for new markets rather than novel drug development.
- Market Access Strategies: Generic companies focus on securing contracts with major distributors and pharmacy benefit managers (PBMs) and ensuring regulatory compliance in target markets.
- Pricing Power: Manufacturers have very little pricing power. Prices are largely dictated by market competition and payer negotiations.
- Financial Trajectory: The financial trajectory is stable but unexciting. Revenue is tied to overall market demand for its indications and the number of generic competitors. Significant revenue growth is unlikely without a major shift in treatment paradigms or a new, patent-protected indication.
What are the Future Market Outlook and Potential Disruptions?
The future of hydroxyzine pamoate is largely predictable within its current framework, but external factors could introduce shifts.
- Continued Generic Dominance: The market will likely remain dominated by generics for the foreseeable future.
- Stable Demand for Specific Indications: Demand for pruritus and short-term anxiety/sedation is expected to remain consistent, driven by its cost-effectiveness and established use.
- Potential for Increased Competition: New entrants into the generic market, particularly from emerging economies, could further intensify price pressure.
- Regulatory Scrutiny: Ongoing pharmacovigilance may lead to further label warnings or restrictions, potentially impacting prescribing patterns if significant safety issues are identified.
- Advancements in Anxiety Treatment: The continued development and adoption of newer, more targeted therapies for anxiety disorders could gradually erode hydroxyzine pamoate's market share in this indication.
- Shifting Guidelines: Changes in clinical practice guidelines for conditions like pruritus or pre-operative sedation could either favor or disfavor its use.
- Supply Chain Vulnerabilities: Like all pharmaceuticals, hydroxyzine pamoate production and distribution are susceptible to global supply chain disruptions, impacting availability and potentially price volatility.
- Emergence of Novel Formulations (Limited Potential): While unlikely to be a major driver, a company might develop a novel, patentable formulation (e.g., extended-release, improved taste profile) that could carve out a niche, but this is a low-probability event given the market's economics.
Key Takeaways
- Hydroxyzine pamoate operates in a mature, generic-dominated market with low single-digit growth projected.
- Its financial trajectory is characterized by low profit margins and volume-driven revenue, heavily influenced by intense generic competition.
- The drug's primary market drivers are its cost-effectiveness and established efficacy for pruritus, short-term anxiety, and sedation.
- Regulatory approvals and adherence to cGMP are essential for market access and product integrity.
- Future market outlook suggests continued stability, with potential disruptions arising from evolving treatment guidelines and increased generic competition.
Frequently Asked Questions
-
What is the primary mechanism of action for hydroxyzine pamoate?
Hydroxyzine pamoate is a first-generation antihistamine that acts as a competitive antagonist of histamine H1 receptors. It also exhibits anticholinergic, antiemetic, and anxiolytic properties, likely through effects on central nervous system neurotransmitters.
-
Are there any significant side effects associated with hydroxyzine pamoate that limit its use?
Yes, significant side effects include profound drowsiness and sedation, dry mouth, and potential for anticholinergic effects such as blurred vision and urinary retention. A rare but serious risk is QT interval prolongation, which can lead to potentially fatal arrhythmias.
-
How does hydroxyzine pamoate compare to newer antihistamines for allergies?
Hydroxyzine pamoate is a first-generation antihistamine, known for causing more sedation than second-generation antihistamines (e.g., cetirizine, loratadine). While effective for pruritus, its sedating effect makes newer agents often preferred for daily allergic rhinitis or when daytime alertness is critical.
-
Can hydroxyzine pamoate be used long-term for anxiety disorders?
While it has anxiolytic properties, hydroxyzine pamoate is generally not recommended for long-term, daily management of chronic anxiety disorders. Its sedative effects and potential for tolerance make it more suitable for short-term situational anxiety or intermittent use. Newer agents like SSRIs and SNRIs are typically preferred for long-term anxiety management.
-
What are the key differentiators for generic manufacturers in the hydroxyzine pamoate market?
Given the commoditized nature of the product, key differentiators for generic manufacturers lie in manufacturing efficiency, supply chain reliability, broad market access through distribution networks, and competitive pricing strategies to secure contracts with payers and wholesalers.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/orange-book-approved-drug-products-therapeutic-equivalence-evaluations
[2] EMA. (n.d.). European Medicines Agency. Retrieved from https://www.ema.europa.eu/
[3] U.S. Pharmacopeia. (n.d.). General Chapter <1078> Good Manufacturing Practices. Retrieved from https://www.uspharmacopeia.org/
[4] PDR. (n.d.). Hydroxyzine Pamoate. Retrieved from https://www.pdr.net/ (Note: PDR access typically requires subscription or professional login, but it is a primary source for drug information).
[5] National Institutes of Health. (n.d.). PubMed. Retrieved from https://pubmed.ncbi.nlm.nih.gov/ (Used for general scientific literature review on efficacy and safety profiles).