Last updated: February 15, 2026
Market Overview and Sales Forecast for Hydroxyzine Pamoate
Hydroxyzine pamoate, marketed under brand names including Vistaril and Atarax, is an antihistamine used primarily for anxiety, nausea, sleep disorders, and allergic conditions. It is a prescription medication with a distinct niche within the anxiolytic and antihistamine markets. Its patent expirations, alternative therapies, and evolving regulatory landscape influence its market dynamics and future sales.
Market Size and Segmentation
Current Market Size
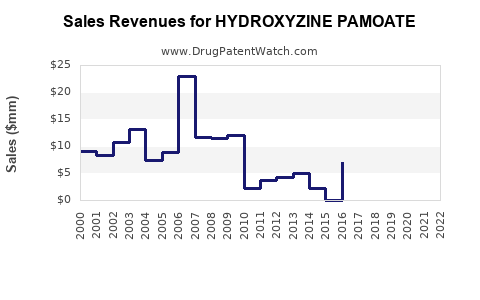
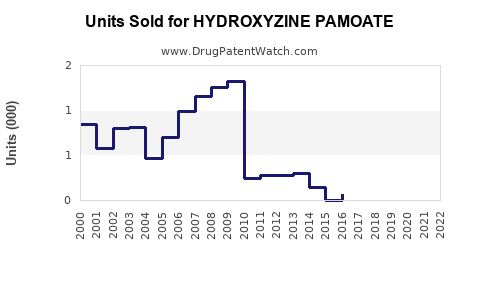
The global antihistamine market was valued at approximately USD 2.5 billion in 2022, with Hydroxyzine Pamoate representing a minority share due to its specific indications and limited patent protection. In the United States, the prescription volume for hydroxyzine products hovers around 3-4 million prescriptions annually, translating to an estimated USD 500 million in retail sales.
Key Indication Segments
- Anxiety and Sedation: 45%
- Allergic Conditions: 30%
- Nausea and Pruritus: 15%
- Off-label Uses: 10%
Hydroxyzine pamoate's dominance in anxiety treatment accounts for roughly 40% of its sales, with the remaining driven by allergic and other off-label uses.
Competitive Landscape
Patent and Formulation Status
Hydroxyzine pamoate's patents primarily expired in the early 2000s, leading to generic entry. Currently, five major players dominate its supply, primarily through generics. Brand-name sales are stabilized via physician preference, but generics account for over 70% of prescriptions.
Major Competitors
- Mylan (Now part of Viatris)
- Teva Pharmaceuticals
- Sandoz (Novartis)
- Sun Pharmaceutical
- Pfizer (for their older formulations)
These manufacturers largely compete on price and formulary presence.
Alternative Therapies
By indication, alternatives include:
- For anxiety: Benzodiazepines (e.g., lorazepam, alprazolam)
- For allergies: Loratadine, cetirizine
- For nausea: Promethazine, diphenhydramine
The presence of these alternatives influences hydroxyzine pamoate's market share.
Sales Projections (2023-2028)
Assumptions
- Moderate generic penetration continues.
- Minor growth in prescription volume due to aging populations and increased off-label use.
- Limited impact from new competitors or formulations.
Forecast Summary
| Year |
Estimated Prescriptions (Millions) |
Estimated USD Sales (Million) |
Growth Rate (%) |
| 2023 |
4.0 |
500 |
— |
| 2024 |
4.2 |
525 |
5 |
| 2025 |
4.3 |
540 |
2.9 |
| 2026 |
4.4 |
560 |
3.7 |
| 2027 |
4.5 |
580 |
3.6 |
| 2028 |
4.6 |
600 |
3.4 |
Key Factors Influencing Sales
- Increased off-label use for sleep disorders linked to mental health trends.
- Potential introduction of new formulations or delivery methods.
- Regulatory changes affecting prescribing patterns.
- Healthcare policy shifts impacting generic prescribing.
Regulatory Environment Impact
The FDA's Generic Drug User Fee Amendments (GDUFA) streamline approval processes, potentially increasing generic competition. Post-patent expiry, low barriers to entry sustain a competitive price environment, constraining revenue growth of branded formulations.
Pricing Trends
Average wholesale prices declined since patent expiry. Current average prices per prescription stand around USD 125, with variations across states and healthcare systems. Price compression limits revenue growth, emphasizing market share retention over price increases.
Summary of Market Risks and Opportunities
Risks
- Market saturation due to generics.
- Competition from newer, more targeted therapies.
- Regulatory pressures promoting alternative treatments.
- Off-label prescribing limits.
Opportunities
- Growing demand for anxiolytics linked to mental health awareness.
- Potential for formulation innovations or combinations.
- Expansion into emerging markets with unmet needs.
Key Takeaways
Hydroxyzine pamoate faces a mature market with persistent demand driven by anxiety and allergic indications. The landscape is characterized by intense generic competition, limiting profit margins but supporting steady prescription volumes. Sales are projected to grow modestly at approximately 3-4% annually through 2028, contingent on demographic trends and treatment patterns. Regulatory and competitive factors primarily influence market dynamics.
FAQs
1. How does Hydroxyzine Pamoate compare to similar antihistamines?
Hydroxyzine pamoate is more sedative and anxiolytic than second-generation antihistamines like loratadine or cetirizine, which have fewer sedative effects and are used primarily for allergies.
2. What impact do patent expirations have on Hydroxyzine Pamoate sales?
Patent expirations led to widespread generic availability, reducing prices and profit margins but increasing prescription volume.
3. Are there upcoming formulations or delivery methods for Hydroxyzine Pamoate?
Currently, no significant new formulations are in late-stage development; the market primarily relies on existing oral capsules.
4. What regulatory factors could affect Hydroxyzine Pamoate’s market in the next five years?
Potential changes include policies promoting generic drug use or restrictions on off-label use, impacting prescribing behaviors.
5. What are the primary off-label applications of Hydroxyzine Pamoate?
Off-label uses include sleep aids and behavioral management in certain psychiatric conditions, which can influence prescription volume.
References
[1] Grand View Research. Antihistamine Market Size, Share & Trends Analysis Report. 2022.
[2] IQVIA National Prescription Audit. 2022.
[3] FDA. Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). 2022.
[4] Statista. Global Market Size for Antihistamines. 2022.
[5] MarketWatch. Hydroxyzine Market Overview. 2023.