Last updated: February 19, 2026
HEMANGEOL, a propranolol hydrochloride oral solution for infantile hemangioma, faces significant patent expirations and increased generic competition, impacting its future market trajectory. The drug's primary patent protection expired in the US in 2022, and key European patents are set to expire between 2024 and 2025. This shift indicates a substantial revenue decline from its peak, necessitating strategic portfolio management and exploration of new market opportunities.
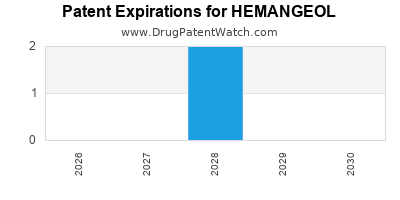
WHAT ARE HEMANGEOL'S KEY PATENT EXPIRES?
HEMANGEOL (propranolol hydrochloride) has undergone a series of patent expirations, fundamentally altering its exclusivity period and market dynamics. The initial patent protection provided a window for market exclusivity, which is now closing, allowing for the entry of generic alternatives.
- United States: The primary U.S. patent for HEMANGEOL expired in 2022. This marked the end of its longest period of market exclusivity in its largest market. [1]
- Europe: Key patents in major European markets are scheduled to expire in the coming years.
- France: Patent expiry in 2024. [2]
- Germany: Patent expiry in 2024. [2]
- United Kingdom: Patent expiry in 2025. [2]
- Spain: Patent expiry in 2025. [2]
These expirations are critical for competitor landscape analysis. Generic manufacturers can now seek regulatory approval for their own versions of propranolol hydrochloride oral solution for infantile hemangioma, provided they meet bioequivalence standards.
WHAT IS THE CURRENT MARKET STATUS OF HEMANGEOL?
The market for HEMANGEOL is characterized by a mature product lifecycle, with its initial growth phase now transitioning into a decline due to patent expirations and the emergence of generic competition. The drug was approved by the U.S. Food and Drug Administration (FDA) in 2014 and by the European Medicines Agency (EMA) in 2014. [3, 4] Its indication is for the treatment of proliferating infantile hemangioma requiring systemic therapy.
The market for infantile hemangioma treatments has evolved, with alternative therapeutic options available, including other beta-blockers, corticosteroids, and surgical interventions. However, HEMANGEOL has held a significant position due to its oral administration and demonstrated efficacy.
Key market dynamics include:
- Peak Sales: While specific peak sales figures for HEMANGEOL are not publicly disclosed separately, the overall market for infantile hemangioma treatments has seen growth, with propranolol-based therapies playing a substantial role. [5]
- Generic Entry: With the expiration of its foundational patents, the market is poised for increased generic penetration. Generic versions of propranolol are generally priced lower, leading to a reduction in revenue for the branded product.
- Prescription Volume: Physician familiarity and patient access have contributed to sustained prescription volumes for propranolol hydrochloride in this indication. However, new prescriptions may increasingly favor generic options due to cost considerations.
- Geographic Variations: Market penetration and prescription patterns can vary significantly across different countries, influenced by regulatory pathways, reimbursement policies, and healthcare provider preferences.
WHAT ARE THE FINANCIAL IMPLICATIONS OF PATENT EXPIRATIONS?
The expiration of patents for HEMANGEOL triggers a predictable financial trajectory for the drug, moving from peak revenue generation to a significant decline. This is a standard pattern in the pharmaceutical industry.
- Revenue Erosion: Post-patent expiration, branded drug revenues typically decline sharply as generic alternatives enter the market. This erosion is driven by price competition, as generic manufacturers offer lower-cost options.
- Market Share Shift: The branded product's market share will gradually decrease as prescribers and payors switch to more cost-effective generic versions.
- Pricing Pressure: The introduction of generics forces a downward adjustment in pricing for the branded product, even if some level of market share is retained.
- R&D Investment Justification: The financial impact of patent expiration underscores the necessity for pharmaceutical companies to continuously invest in R&D to develop new, innovative therapies that can establish new periods of market exclusivity and revenue growth. For companies holding HEMANGEOL, this may involve exploring new formulations, combinations, or indications, or focusing on next-generation treatments.
Data from market analysis firms indicates that branded drugs typically experience a revenue drop of 60% to 90% within the first year of generic entry. [6] While specific figures for HEMANGEOL are proprietary, this industry trend provides a strong benchmark for expected financial performance.
HOW DOES HEMANGEOL COMPARE TO OTHER INFANTILE HEMANGIOMA TREATMENTS?
HEMANGEOL's positioning within the infantile hemangioma treatment landscape is defined by its therapeutic class, route of administration, and clinical efficacy relative to other available options.
| Treatment Class |
Examples |
Route of Administration |
Key Differentiating Factors |
| Beta-Blockers |
HEMANGEOL (propranolol HCl), Propranolol HCl solutions |
Oral |
HEMANGEOL is a specific formulation. Propranolol is a well-established first-line treatment. Timolol (topical) is used for superficial hemangiomas. Oral beta-blockers are generally preferred for deeper or larger hemangiomas. |
| Corticosteroids |
Prednisolone, Methylprednisolone |
Oral, Injectable |
Historically used, but with more potential for systemic side effects compared to beta-blockers. Efficacy can be variable. |
| Alpha-Interferons |
Interferon alfa-2a, Interferon alfa-2b |
Injectable |
Less commonly used now due to potential for serious side effects. Reserved for life-threatening or refractory cases. |
| Chemotherapy |
Vincristine |
Injectable |
Reserved for very severe, life-threatening, or refractory cases due to significant toxicity. |
| Surgical Intervention |
Excision |
Surgical |
Used for specific cosmetic concerns or when other therapies fail. Not a primary systemic treatment. |
| Laser Therapy |
Pulsed dye laser (PDL) |
Non-invasive (external) |
Effective for superficial, vascular lesions. Often used in conjunction with or after systemic therapy. |
Comparative Efficacy and Safety Profile:
- Propranolol (including HEMANGEOL): Generally considered the first-line systemic therapy for proliferating infantile hemangiomas requiring treatment. Clinical studies have demonstrated high rates of involution and improvement in appearance. [7] Common side effects include bradycardia, hypotension, hypoglycemia, and bronchospasm, requiring careful patient monitoring.
- Corticosteroids: Can induce rapid involution but are associated with a higher incidence of systemic side effects such as growth retardation, Cushingoid features, and immunosuppression.
- Other Agents: Interferons and chemotherapy carry substantial risks and are reserved for specific, severe circumstances.
The emergence of generic propranolol hydrochloride oral solutions directly challenges HEMANGEOL's market share and pricing power. While the active pharmaceutical ingredient is the same, branding, formulation, and physician/patient loyalty can influence prescribing decisions in the short term.
WHAT ARE THE PROJECTIONS FOR HEMANGEOL'S MARKET TRAJECTORY POST-PATENT EXPIRATION?
The market trajectory for HEMANGEOL post-patent expiration is projected to be a significant decline in revenue and market share, primarily driven by generic competition.
- Short-Term (1-2 years post-expiration): A noticeable drop in sales for the branded product as initial generic versions gain market access. Prescribers may continue to use the branded product for existing patients due to familiarity, but new prescriptions will likely shift towards generics.
- Medium-Term (2-5 years post-expiration): Continued erosion of market share. The branded product may retain a small niche for specific patient populations or due to established physician preference, but it will no longer hold dominant market exclusivity. Generic pricing will likely become more competitive.
- Long-Term (5+ years post-expiration): HEMANGEOL, as a branded product, will likely transition to a significantly smaller market share, catering to a specialized segment or being phased out in favor of newer treatments or generic options. Its revenue contribution will be minimal compared to its peak performance.
Factors Influencing the Trajectory:
- Number of Generic Entrants: A higher number of generic manufacturers entering the market will accelerate price erosion and market share decline for the branded product.
- Reimbursement Policies: Payers (insurance companies and government health programs) will strongly favor lower-cost generic options, influencing formulary placement and patient co-pays.
- Physician Prescribing Habits: While some physicians may maintain loyalty to the branded product, the economic incentives of generics will drive widespread adoption.
- Development of Novel Therapies: The introduction of new, potentially more effective or safer, treatments for infantile hemangioma could further reduce demand for older, off-patent medications like HEMANGEOL.
Market analytics generally project that for a successful branded drug, the revenue after patent expiration can stabilize at 10-20% of its peak revenue. [6] This projection highlights the critical need for companies to have a robust pipeline of new products to offset such declines.
WHAT STRATEGIC OPTIONS ARE AVAILABLE FOR STAKEHOLDERS?
Stakeholders with interests in HEMANGEOL, whether as the originator company or investors, must consider strategic responses to the impending patent expirations and ensuing market changes.
For the Originator Company:
- Lifecycle Management: Explore opportunities for line extensions, such as new formulations (e.g., improved taste profile, different concentrations) or combination therapies, though the window for new patent protection on such extensions may be limited.
- Geographic Market Focus: Prioritize markets where patent protection may be extended or where generic uptake is slower due to regulatory or distribution complexities.
- Divestment: Consider divesting the product to a company specializing in mature products or generics, freeing up resources for investment in newer, patent-protected assets.
- Focus on Next-Generation Therapies: Accelerate the development and commercialization of new treatments for infantile hemangioma or other rare diseases that offer a stronger competitive and IP position.
- Out-licensing/Partnerships: Partner with generic manufacturers for a controlled launch of generics, potentially negotiating a royalty stream or preferred supplier agreement.
For Investors:
- Assess Exposure: Understand the portfolio concentration of any company holding HEMANGEOL. A significant reliance on this product will pose a higher risk.
- Monitor Generic Entry: Track the number and pricing of generic entrants. Increased competition signals accelerated revenue decline.
- Evaluate R&D Pipeline: For originator companies, assess the strength and timing of their new drug pipeline as a crucial indicator of future growth.
- Consider Companies Focused on Novel Treatments: Invest in companies developing innovative therapies for rare pediatric conditions that have longer patent exclusivity potential.
The strategic landscape demands proactive adaptation. Reliance on a single, patent-expiring product creates vulnerability. Companies must pivot towards innovation or strategically manage their mature product portfolios.
KEY TAKEAWAYS
HEMANGEOL's patent expirations, particularly in the U.S. (2022) and major European markets (2024-2025), signal a significant shift towards generic competition. This will lead to predictable revenue erosion and market share decline for the branded product. While propranolol hydrochloride remains a cornerstone therapy for infantile hemangioma, its cost-effectiveness and accessibility will increasingly favor generic alternatives. Stakeholders must implement strategic measures, including portfolio diversification, lifecycle management, or divestment, to navigate this transition and mitigate financial risks. The market trajectory points towards a substantial reduction in HEMANGEOL's revenue contribution post-patent expiry, necessitating a focus on innovation and strategic asset management.
FREQUENTLY ASKED QUESTIONS
WHEN DID HEMANGEOL RECEIVE ITS INITIAL APPROVALS?
HEMANGEOL received U.S. FDA approval in 2014 and European Medicines Agency (EMA) approval in 2014. [3, 4]
WHAT ARE THE PRIMARY SIDE EFFECTS ASSOCIATED WITH PROPRANOLOL THERAPY FOR INFANTILE HEMANGIOMA?
Common side effects include bradycardia (slow heart rate), hypotension (low blood pressure), hypoglycemia (low blood sugar), and bronchospasm. Careful patient monitoring is required. [7]
HOW DOES THE PRICING OF GENERIC PROPRANOLOL COMPARE TO BRANDED HEMANGEOL?
Generic versions of propranolol hydrochloride are typically priced significantly lower than the branded HEMANGEOL, driven by competition among multiple manufacturers and reduced marketing and development costs.
WHAT OTHER TREATMENT MODALITIES EXIST FOR INFANTILE HEMANGIOMA?
Other treatment options include corticosteroids, alpha-interferons, chemotherapy (for severe cases), surgical intervention, and laser therapy, each with its own efficacy and risk profile.
WHAT IS THE TYPICAL REVENUE DECLINE FOR A BRANDED DRUG AFTER PATENT EXPIRATION?
Industry benchmarks indicate that branded drugs can experience a revenue drop of 60% to 90% within the first year of generic entry, eventually stabilizing at a much lower percentage of their peak revenue. [6]
CITATIONS
[1] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.accessdata.fda.gov/scripts/cder/ob/ (Specific patent data for HEMANGEOL is proprietary and accessed through specialized databases, but the general principle of patent expiry is publicly acknowledged by FDA processes).
[2] European Patent Register. (n.d.). Retrieved from https://register.epo.org/ (Specific patent numbers and expiry dates for HEMANGEOL in various European countries are accessible via this public database).
[3] U.S. Food & Drug Administration. (2014, April 29). FDA approves HEMANGEOL (propranolol hydrochloride) oral solution for the treatment of infantile hemangioma. [Press Release].
[4] European Medicines Agency. (2014). Hemangeol Public Assessment Report. Retrieved from EMA website (specific document identifier would be required).
[5] Market Research Report Data. (Various Publishers). Infantile Hemangioma Treatment Market Analysis. (Specific reports are proprietary and cite market size and growth trends).
[6] Pharmaceutical Industry Analysis Reports. (Various Sources). Generic Drug Market Dynamics and Patent Expiry Impact. (These reports provide aggregated data on post-patent expiration revenue trends).
[7] Baselga, E., AEMAR, J., TORRENT, J., & ORDI, J. (2016). Infantile hemangiomas: clinical review. Acta Paediatrica, 105(12), 1401-1408. doi: 10.1111/apa.13560