GLYXAMBI Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Glyxambi, and when can generic versions of Glyxambi launch?
Glyxambi is a drug marketed by Boehringer Ingelheim and is included in one NDA. There are fifteen patents protecting this drug and one Paragraph IV challenge.
This drug has five hundred and twenty-one patent family members in forty-five countries.
The generic ingredient in GLYXAMBI is empagliflozin; linagliptin. There are twenty-two drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the empagliflozin; linagliptin profile page.
DrugPatentWatch® Generic Entry Outlook for Glyxambi
Glyxambi was eligible for patent challenges on May 2, 2015.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be April 14, 2030. This may change due to patent challenges or generic licensing.
There have been fifty-two patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
There are three tentative approvals for the generic drug (empagliflozin; linagliptin), which indicates the potential for near-term generic launch.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for GLYXAMBI?
- What are the global sales for GLYXAMBI?
- What is Average Wholesale Price for GLYXAMBI?
Summary for GLYXAMBI
| International Patents: | 521 |
| US Patents: | 15 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 2 |
| Drug Prices: | Drug price information for GLYXAMBI |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for GLYXAMBI |
| What excipients (inactive ingredients) are in GLYXAMBI? | GLYXAMBI excipients list |
| DailyMed Link: | GLYXAMBI at DailyMed |
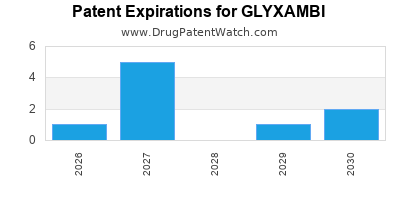
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for GLYXAMBI
Generic Entry Date for GLYXAMBI*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for GLYXAMBI
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Humanis Saglk Anonim Sirketi | PHASE1 |
Pharmacology for GLYXAMBI
Paragraph IV (Patent) Challenges for GLYXAMBI
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| GLYXAMBI | Tablets | empagliflozin; linagliptin | 10 mg/5 mg and 25 mg/5 mg | 206073 | 9 | 2018-08-01 |
US Patents and Regulatory Information for GLYXAMBI
GLYXAMBI is protected by fifteen US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of GLYXAMBI is ⤷ Start Trial.
This potential generic entry date is based on patent ⤷ Start Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim | GLYXAMBI | empagliflozin; linagliptin | TABLET;ORAL | 206073-002 | Jan 30, 2015 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Boehringer Ingelheim | GLYXAMBI | empagliflozin; linagliptin | TABLET;ORAL | 206073-002 | Jan 30, 2015 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Boehringer Ingelheim | GLYXAMBI | empagliflozin; linagliptin | TABLET;ORAL | 206073-001 | Jan 30, 2015 | RX | Yes | No | ⤷ Start Trial | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Boehringer Ingelheim | GLYXAMBI | empagliflozin; linagliptin | TABLET;ORAL | 206073-002 | Jan 30, 2015 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for GLYXAMBI
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim | GLYXAMBI | empagliflozin; linagliptin | TABLET;ORAL | 206073-002 | Jan 30, 2015 | ⤷ Start Trial | ⤷ Start Trial |
| Boehringer Ingelheim | GLYXAMBI | empagliflozin; linagliptin | TABLET;ORAL | 206073-001 | Jan 30, 2015 | ⤷ Start Trial | ⤷ Start Trial |
| Boehringer Ingelheim | GLYXAMBI | empagliflozin; linagliptin | TABLET;ORAL | 206073-001 | Jan 30, 2015 | ⤷ Start Trial | ⤷ Start Trial |
| Boehringer Ingelheim | GLYXAMBI | empagliflozin; linagliptin | TABLET;ORAL | 206073-002 | Jan 30, 2015 | ⤷ Start Trial | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for GLYXAMBI
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim International GmbH | Glyxambi | empagliflozin, linagliptin | EMEA/H/C/003833Glyxambi, fixed dose combination of empagliflozin and linagliptin, is indicated in adults aged 18 years and older with type 2 diabetes mellitus:to improve glycaemic control when metformin and/or sulphonylurea (SU) and one of the monocomponents of Glyxambi do not provide adequate glycaemic control;when already being treated with the free combination of empagliflozin and linagliptin. | Authorised | no | no | no | 2016-11-11 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for GLYXAMBI
When does loss-of-exclusivity occur for GLYXAMBI?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 7970
Estimated Expiration: ⤷ Start Trial
Patent: 7657
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 08288407
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 0815331
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 96558
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 08002427
Estimated Expiration: ⤷ Start Trial
China
Patent: 1784270
Estimated Expiration: ⤷ Start Trial
Patent: 4288166
Estimated Expiration: ⤷ Start Trial
Patent: 4353077
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 51239
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0170022
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 18308
Estimated Expiration: ⤷ Start Trial
Patent: 17017
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 87879
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 109977
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 8608
Estimated Expiration: ⤷ Start Trial
Patent: 1000321
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 87879
Estimated Expiration: ⤷ Start Trial
Patent: 98152
Estimated Expiration: ⤷ Start Trial
Patent: 06156
Estimated Expiration: ⤷ Start Trial
Patent: 39577
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 01721
Estimated Expiration: ⤷ Start Trial
Patent: 03351
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 30158
Estimated Expiration: ⤷ Start Trial
Patent: 700020
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 2886
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 95914
Estimated Expiration: ⤷ Start Trial
Patent: 10535850
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 187879
Estimated Expiration: ⤷ Start Trial
Patent: 2017014
Estimated Expiration: ⤷ Start Trial
Patent: 87879
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 2037
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 10001696
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 573
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 612
Estimated Expiration: ⤷ Start Trial
Netherlands
Patent: 0872
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 3242
Estimated Expiration: ⤷ Start Trial
Norway
Patent: 17020
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 090938
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 87879
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 87879
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 205
Patent: FARMACEUTSKA KOMPOZICIJA KOJA SADRŽI GLUKOPIRANOZILOM SUPSTITUISANI DERIVAT BENZENA (PHARMACEUTICAL COMPOSITION COMPRISING A GLUCOPYRANOSYL-SUBSTITUTED BENZENE DERIVATIVE)
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 87879
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 0909105
Patent: Pharmaceutical composition comprising a glucopyranosyl-substituted benzene derivative
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1491554
Estimated Expiration: ⤷ Start Trial
Patent: 100049595
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 02748
Estimated Expiration: ⤷ Start Trial
Taiwan
Patent: 72325
Estimated Expiration: ⤷ Start Trial
Patent: 0914030
Patent: Pharmaceutical composition comprising a glucopyranosyl-substituted benzene derivative
Estimated Expiration: ⤷ Start Trial
Patent: 1436798
Patent: Pharmaceutical composition comprising a glucopyranosyl-substituted benzene derivative
Estimated Expiration: ⤷ Start Trial
Tunisia
Patent: 10000073
Patent: COMPOSITON PHARMACEUTIQUE CONTENANT UN DERIVE DE BENZENE SUBSTITUE PAR GLUCOPYRANOSYLE
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 0384
Patent: ФАРМАЦЕВТИЧНА КОМПОЗИЦІЯ, ЩО МІСТИТЬ ГЛЮКОПІРАНОЗИЛЗАМІЩЕНУ ПОХІДНУ БЕНЗОЛУ ТА ІНГІБІТОР DPP IV (PHARMACEUTICAL COMPOSITION COMPRISING A GLUCOPYRANOSYL-SUBSTITUTED BENZENE DERIVATIVE)
Estimated Expiration: ⤷ Start Trial
Uruguay
Patent: 296
Patent: COMPOSICION FARMACÉUTICA QUE COMPRENDE UN DERIVADO DE BENCENO SUSTITUIDO CON GLUCOPIRANOSILO
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering GLYXAMBI around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Australia | 2013251292 | 8-[3-amino-piperidin-1-yl]-xanthines, the production thereof and the use of the same as medicaments | ⤷ Start Trial |
| Montenegro | 01941 | ⤷ Start Trial | |
| Croatia | P20150003 | ⤷ Start Trial | |
| Cyprus | 1123253 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for GLYXAMBI
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1730131 | 200 5026-2014 | Slovakia | ⤷ Start Trial | PRODUCT NAME: EMPAGLIFLOZIN; REGISTRATION NO/DATE: EU/1/14/930 20140527 |
| 1532149 | 91889 | Luxembourg | ⤷ Start Trial | 91889, EXPIRES: 20260824 |
| 1084705 | SPC/GB14/086 | United Kingdom | ⤷ Start Trial | PRODUCT NAME: VILDAGLIPTIN; REGISTERED: UK EU/1/07/414/001-017 20070928 |
| 1084705 | C01084705/03 | Switzerland | ⤷ Start Trial | PRODUCT NAME: SAXAGLIPTIN; REGISTRATION NO/DATE: SWISSMEDIC 59390 05.02.2010 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Empagliflozin (Jardiance) Patent Landscape and Market Trajectory
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.
