Last updated: February 19, 2026
Empagliflozin, marketed as Jardiance, is a sodium-glucose cotransporter-2 (SGLT2) inhibitor used to treat type 2 diabetes. Its patent portfolio and subsequent market performance are critical indicators for R&D investment and strategic planning in the diabetes therapeutic area. This analysis examines key patent expirations, litigation, and market penetration to forecast future financial trajectories.
What are the Key Patents Covering Empagliflozin?
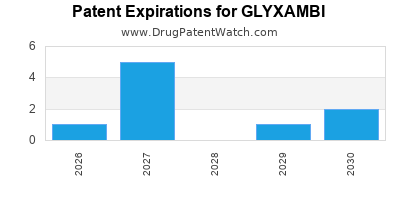
The foundational patents for empagliflozin are held by Boehringer Ingelheim. The primary composition of matter patent, US Patent No. 6,849,625, claims empagliflozin and related compounds. This patent was granted on January 25, 2005. Secondary patents cover manufacturing processes, crystalline forms, and methods of use. For instance, US Patent No. 8,153,617, related to specific crystalline forms of empagliflozin, was issued on April 10, 2012.
The lifespan of these patents is crucial for understanding generic entry timelines. The composition of matter patent, absent any extensions, would expire in 2025. However, patent term extensions (PTEs) and other adjustments can alter these dates. Regulatory exclusivities, such as New Chemical Entity (NCE) exclusivity in the U.S., also play a significant role. For Jardiance, the NCE exclusivity was granted by the U.S. Food and Drug Administration (FDA), providing a period of market protection independent of patent expiry.
| Patent Number |
Title/Subject Matter |
Grant Date |
Original Expiry (approx.) |
Relevant Jurisdiction |
| US 6,849,625 |
Empagliflozin compounds |
2005-01-25 |
2025 |
United States |
| US 8,153,617 |
Crystalline forms |
2012-04-10 |
2028 |
United States |
| EP 1 723 009 |
Pharmaceutical preparations |
2009-09-02 |
2026 |
Europe |
Note: Expiry dates are approximate and do not account for potential patent term extensions or other legal adjustments.
When Will Empagliflozin Face Generic Competition?
The timeline for generic competition is directly linked to the expiry of primary patents and the resolution of any patent litigation. The composition of matter patent for empagliflozin is the most critical. While its original expiry is in 2025, potential patent term extensions (PTEs) can prolong market exclusivity. In the United States, PTEs are granted to compensate for regulatory review delays. For Jardiance, the PTE for the '625 patent extended its exclusivity.
Furthermore, the landscape is complicated by secondary patents covering various aspects of the drug, such as specific manufacturing processes or polymorphic forms. Generic manufacturers often challenge these secondary patents to secure an earlier market entry. Litigation surrounding these patents can significantly influence the timing of generic availability.
In the U.S., the earliest likely generic entry for empagliflozin hinges on the expiry of the extended term of the primary patents and the outcome of any Paragraph IV certifications filed by generic companies. A Paragraph IV certification asserts that the patents listed in the FDA’s Orange Book are invalid, unenforceable, or will not be infringed by the generic product. This often initiates Hatch-Waxman litigation.
As of early 2024, there have been ongoing legal challenges to various empagliflozin patents. The resolution of these disputes will determine the precise launch dates for generic versions. Without specific court rulings or settlement agreements, definitive dates for widespread generic availability remain fluid. However, based on typical patent lifecycles and potential extensions, significant generic competition is anticipated in the late 2020s.
What is the Current Market Performance of Jardiance?
Jardiance has demonstrated robust commercial success since its launch. Its market penetration is driven by its efficacy in managing type 2 diabetes, with expanding indications that include reducing the risk of cardiovascular death and hospitalization for heart failure in adults with type 2 diabetes, and chronic kidney disease.
Eli Lilly and Company, which co-markets Jardiance in the U.S. with Boehringer Ingelheim, reported significant revenue growth for the drug. In 2023, Jardiance generated $6.73 billion in global sales, a 39% increase compared to 2022. This growth was fueled by strong uptake in the U.S. and key international markets, as well as expanding indications.
| Year |
Global Sales (USD Billions) |
Year-over-Year Growth (%) |
| 2021 |
4.23 |
33% |
| 2022 |
4.84 |
14% |
| 2023 |
6.73 |
39% |
Source: Eli Lilly and Company Financial Reports.
The drug's performance is underpinned by its strong clinical data, including results from landmark trials such as EMPA-REG OUTCOME, EMPEROR-Reduced, and EMPEROR-Preserved. These trials have established empagliflozin's cardiovascular and renal benefits, broadening its patient population and increasing its market share against competitors.
The market share of Jardiance within the SGLT2 inhibitor class is substantial. It competes with other SGLT2 inhibitors like dapagliflozin (Farxiga) and canagliflozin (Invokana). Jardiance's expanded indications, particularly its demonstrated benefits in heart failure and chronic kidney disease, have positioned it as a leading therapy.
How Do Cardiovascular and Renal Indications Impact Market Share?
The inclusion of cardiovascular (CV) and renal outcome benefits has been a transformative development for Jardiance. Initially approved for glycemic control in type 2 diabetes, its subsequent approvals for reducing the risk of cardiovascular death and hospitalization for heart failure, and later for chronic kidney disease, have dramatically expanded its market utility and patient base.
The EMPA-REG OUTCOME trial, published in 2015, was pivotal. It demonstrated a significant reduction in major adverse cardiovascular events (MACE) and cardiovascular death in patients with type 2 diabetes and established cardiovascular disease. This evidence supported its label expansion and positioned it as a cardiorenal protective agent, not just a glucose-lowering drug.
Following this, the EMPEROR program trials provided further evidence of its benefits in heart failure patients, both with reduced and preserved ejection fraction. The EMPEROR-Reduced trial showed a significant reduction in the composite of cardiovascular death or hospitalization for heart failure in patients with heart failure and reduced ejection fraction. The EMPEROR-Preserved trial demonstrated similar benefits in patients with heart failure and preserved ejection fraction, a population with limited treatment options.
These expanded indications have had a direct and substantial impact on Jardiance’s market share:
- Broader Patient Population: It now appeals to a much wider group of patients, including those with established CV disease or CKD, in addition to those primarily seeking glycemic control.
- Competitive Differentiation: The proven cardiorenal benefits offer a significant advantage over diabetes medications that lack these outcomes. This allows physicians to prescribe Jardiance with a dual or even triple benefit in mind (glycemic control, CV protection, renal protection).
- Reimbursement and Payer Acceptance: Strong outcomes data often leads to more favorable formulary placement and reimbursement from payers, further enhancing market access.
- Clinical Practice Guideline Inclusion: Jardiance is now a recommended therapy in major diabetes, cardiovascular, and kidney disease guidelines, solidifying its place in treatment algorithms.
This strategic expansion of indications has not only driven Jardiance’s sales growth but has also repositioned SGLT2 inhibitors as cornerstone therapies in managing cardiometabolic diseases, moving beyond their initial role as diabetes adjuncts.
What is the Competitive Landscape for SGLT2 Inhibitors?
The SGLT2 inhibitor class is competitive, with several key players vying for market share. The primary competitors to Jardiance (empagliflozin) are Farxiga (dapagliflozin) from AstraZeneca and Invokana (canagliflozin) from Johnson & Johnson.
| Drug Name |
Active Ingredient |
Primary Indication(s) |
Key Competitor Claims/Differentiators |
| Jardiance |
Empagliflozin |
Type 2 Diabetes; Cardiovascular risk reduction; Heart Failure; Chronic Kidney Disease |
Strong CV and renal outcomes data (EMPA-REG OUTCOME, EMPEROR trials); Broadest indications in HF and CKD. |
| Farxiga |
Dapagliflozin |
Type 2 Diabetes; Cardiovascular risk reduction; Heart Failure; Chronic Kidney Disease |
Also demonstrated significant CV and renal benefits (DECLARE-TIMI 58, DAPA-HF, DAPA-CKD trials); Early mover in HF and CKD approvals. |
| Invokana |
Canagliflozin |
Type 2 Diabetes; Cardiovascular risk reduction; Chronic Kidney Disease (specific indications) |
Also demonstrated CV risk reduction (CANVAS program) and renal benefits (CREDENCE trial); Has specific indications for reducing progression of diabetic kidney disease. |
| Steglatro |
Ertugliflozin |
Type 2 Diabetes |
Newer entrant, combined with sitagliptin (Segluromet) or as monotherapy; CV outcomes trial data is less robust compared to the top three. |
| Brenzavvy (oral) |
Bempagliflozin |
Type 2 Diabetes (latest entrant) |
Newest SGLT2 inhibitor, recently approved with limited long-term outcomes data compared to established competitors. |
Note: Indication details can be complex and vary by region. This table provides a general overview.
The competitive dynamics are driven by several factors:
- Clinical Outcomes Data: As demonstrated by Jardiance and Farxiga, robust cardiovascular and renal outcome data are critical for market differentiation and expanded use beyond glycemic control.
- Indication Breadth: The drug with the broadest approved indications, particularly in heart failure and chronic kidney disease, gains a significant advantage. Jardiance and Farxiga are currently leading in this regard.
- Patent Expiries and Generic Entry: The impending expiry of key patents for older SGLT2 inhibitors will introduce generic competition, potentially leading to price erosion and market share shifts.
- Formulary Access and Payer Policies: Reimbursement decisions by payers heavily influence prescription patterns. Favorable formulary placement for drugs with demonstrated outcomes benefits is common.
- New Entrants: Newer agents like Steglatro and Brenzavvy aim to capture market share, often through combination therapies or targeting specific patient subgroups. However, they face the challenge of establishing their own outcomes data and market presence against established leaders.
The SGLT2 class is expected to remain a significant segment of the diabetes and cardiometabolic market. The focus is shifting from solely glucose lowering to multi-organ protection, making drugs with proven CV and renal benefits highly valued.
What are the Future Financial Projections for Empagliflozin?
Forecasting the future financial trajectory of empagliflozin requires considering its current growth trajectory, the impending threat of generic competition, and the potential for further indication expansions or new market penetration.
Jardiance has consistently achieved high double-digit growth, driven by its expanding label and strong clinical evidence. The reported 39% growth in 2023 to $6.73 billion highlights its sustained momentum. Analysts project continued strong performance in the near to mid-term. For instance, projections often place Jardiance's annual sales in the double-digit billions before patent expiries significantly impact its revenue.
However, the expiration of key patents, particularly the composition of matter patent, will mark a turning point. The U.S. market, with its robust generic drug sector and the Hatch-Waxman Act framework, is typically the first to see significant generic erosion. European markets, with their own patent regimes and pricing mechanisms, will also face generic entry.
The timing of generic entry is critical. If generic empagliflozin becomes available in the U.S. around 2025-2026, it will likely lead to a rapid decline in Jardiance's sales volume, though brand-name sales may persist for a period in certain segments or through authorized generics.
Factors influencing future financial projections:
- Patent Expiry Dates and Litigation Outcomes: This is the most significant determinant of generic entry timing and revenue decline.
- Market Penetration in New Indications: Continued uptake in heart failure and CKD patient populations will sustain growth until generic pressures become dominant.
- Competition: The performance of competing SGLT2 inhibitors and other diabetes/cardiometabolic therapies will influence Jardiance's market share.
- Geographic Expansion: Continued rollout and uptake in emerging markets can provide additional revenue streams.
- Potential New Indications or Combination Therapies: While less likely for a mature drug, new clinical trial data or formulation developments could offer incremental benefits.
Based on current trends and anticipated generic entry, Jardiance's revenue is projected to peak in the late 2020s before experiencing a substantial decline as generic versions gain market share. However, even post-patent expiry, a branded product can maintain a residual market presence, particularly if authorized generics are introduced or if specific patient populations continue to prefer the branded option.
The financial trajectory for empagliflozin is one of strong growth followed by a significant revenue cliff as generic competition intensifies. The pre-patent expiry period represents a peak revenue generation window.
What is the Impact of Regulatory Approvals and Exclusivities on Market Dynamics?
Regulatory approvals and exclusivities are foundational to the market dynamics of any pharmaceutical drug, including empagliflozin. These mechanisms dictate market entry, competitive timelines, and ultimately, revenue potential.
Key Regulatory Approvals and Exclusivities for Empagliflozin:
- New Chemical Entity (NCE) Exclusivity: In the United States, NCE exclusivity is typically five years from the date of approval. This prevents the FDA from approving a generic version of the same drug for a period of five years, even if patents expire earlier. Jardiance received NCE exclusivity upon its initial approval for type 2 diabetes. This provided a significant head start against potential generic competitors.
- Patent Term Extension (PTE): U.S. patent law allows for extensions of patent terms to compensate for patent holder losses during the regulatory review process. The PTE is intended to restore some of the lost market exclusivity. For Jardiance, the PTE for the composition of matter patent extended its protection beyond its original expiry date, delaying generic entry. The duration of the PTE is calculated based on the length of the FDA's regulatory review.
- Orphan Drug Exclusivity: This exclusivity applies to drugs designated for rare diseases. Empagliflozin is not an orphan drug, so this type of exclusivity is not relevant.
- Pediatric Exclusivity: In the U.S., companies can gain an additional six months of market exclusivity if they conduct studies in pediatric populations as requested by the FDA. This can be added to existing patent or NCE exclusivities. Information on whether Jardiance benefited from pediatric exclusivity would be found in its FDA approval history.
- Data Exclusivity: In many regions, including Europe, data exclusivity provides a period during which generic companies cannot rely on the innovator's clinical trial data to support their own applications, even if patents have expired. This is separate from patent protection.
- Marked-up Patent Listing (Orange Book): In the U.S., patents claiming a drug are listed in the FDA's Approved Drug Products With Therapeutic Equivalence Evaluations (the "Orange Book"). Generic manufacturers must certify that the listed patents are either invalid, will not be infringed, or have expired. The listing of multiple patents, including secondary patents on manufacturing processes or formulations, creates a complex legal and regulatory environment that generic companies must navigate.
Impact on Market Dynamics:
- Market Entry Barriers: NCE exclusivity and PTEs create substantial barriers to entry for generic manufacturers, allowing the innovator to recoup R&D investments and establish market share.
- Litigation Trigger: The expiration of NCE exclusivity or PTEs often coincides with generic manufacturers filing Abbreviated New Drug Applications (ANDAs) and challenging patents through Paragraph IV certifications, leading to patent litigation.
- Market Longevity for Innovator: These regulatory exclusivities, in conjunction with patent protection, determine the effective market life of the drug and its revenue-generating period.
- Pricing Power: During periods of market exclusivity, the innovator company holds significant pricing power. This power diminishes rapidly with the introduction of generic competition.
- Strategic R&D: The availability and duration of these exclusivities influence a company's R&D strategy, encouraging investment in novel compounds with the potential for lengthy market protection.
The interplay between patent law and regulatory exclusivities is dynamic. Generic companies continuously seek to exploit any loopholes or expiration timelines to introduce lower-cost alternatives. For empagliflozin, the combination of NCE exclusivity and PTEs has allowed for a prolonged period of market exclusivity, contributing to its significant commercial success. However, the eventual exhaustion of these protections will inevitably lead to increased competition and a shift in market dynamics.
Key Takeaways
- Empagliflozin's (Jardiance) foundational patent (US 6,849,625) is set to expire around 2025, with potential extensions impacting the precise timeline for generic entry.
- Jardiance has achieved significant market success, generating $6.73 billion in global sales in 2023, driven by strong clinical data and expanded indications.
- The inclusion of cardiovascular and renal benefits has broadened Jardiance's patient population and significantly enhanced its market share within the SGLT2 inhibitor class.
- The competitive landscape for SGLT2 inhibitors includes Farxiga (dapagliflozin) and Invokana (canagliflozin), with market dynamics influenced by clinical outcomes, indication breadth, and impending generic competition.
- Future financial projections for empagliflozin indicate continued strong growth through the mid-to-late 2020s, followed by a substantial revenue decline upon the advent of widespread generic competition.
- Regulatory exclusivities, such as New Chemical Entity (NCE) exclusivity and Patent Term Extensions (PTEs), have been critical in establishing empagliflozin's market longevity and pricing power, but their eventual expiration will fundamentally alter market dynamics.
Frequently Asked Questions
1. How will the expiration of the primary composition of matter patent for empagliflozin affect its pricing?
Upon the expiration of the primary patent and the introduction of generic competitors, the pricing of empagliflozin is expected to decrease significantly, a pattern typical for branded drugs facing generic erosion.
2. Are there any ongoing legal challenges that could accelerate or delay generic entry for empagliflozin?
Yes, patent litigation is common in the pharmaceutical industry. Challenges to empagliflozin's patents, including those related to manufacturing processes or specific crystalline forms, can arise and potentially impact the timeline for generic market entry. The outcomes of such litigation are closely monitored.
3. Beyond type 2 diabetes, what are the most impactful indications driving Jardiance's market growth?
The indications for reducing the risk of cardiovascular death and hospitalization for heart failure, as well as for treating chronic kidney disease, have been the primary drivers of Jardiance's market growth beyond its glycemic control indication.
4. What is the estimated market share of Jardiance within the SGLT2 inhibitor class globally?
While specific market share figures fluctuate, Jardiance is consistently among the top-selling SGLT2 inhibitors globally, often holding a leading or co-leading position alongside competitors like dapagliflozin.
5. Can Boehringer Ingelheim or Eli Lilly launch an "authorized generic" after patent expiry to compete with other generics?
Yes, the innovator company can often launch an authorized generic version of its drug after patent expiry, typically at a lower price than the branded product, to compete with independent generic manufacturers and capture some market share.
Citations
[1] U.S. Patent and Trademark Office. (n.d.). Patent Full Text and Image Database. Retrieved from USPTO Patent Database. (Specific patent numbers referenced were publicly available search results).
[2] Eli Lilly and Company. (2024, February 1). Eli Lilly and Company Reports Fourth Quarter and Full-Year 2023 Results. Retrieved from Eli Lilly Investor Relations.
[3] FDA. (n.d.). Approved Drug Products With Therapeutic Equivalence Evaluations (Orange Book). Retrieved from FDA website. (Specific drug and patent data accessed via FDA database).
[4] AstraZeneca PLC. (n.d.). Farxiga (dapagliflozin) Product Information. Retrieved from AstraZeneca Investor Relations and Product Websites.
[5] Johnson & Johnson. (n.d.). Invokana (canagliflozin) Product Information. Retrieved from Johnson & Johnson Investor Relations and Product Websites.