Last updated: February 19, 2026
Flector (diclofenac epolamine) is a topical nonsteroidal anti-inflammatory drug (NSAID) used for the relief of pain and inflammation associated with acute strains, sprains, and bruises of the extremities. Its market performance is influenced by patent expiry, generic competition, and therapeutic positioning against alternative pain management options.
What is Flector’s Patent Status and Exclusivity Landscape?
Flector’s primary patent, U.S. Patent No. 5,376,652, covering the topical diclofenac epolamine formulation, expired in 2012. This expiry opened the door for generic competition in the United States. Subsequent patents related to specific manufacturing processes or delivery systems may exist, but the core composition-of-matter patent has lapsed, significantly impacting its market exclusivity.
The active pharmaceutical ingredient, diclofenac, is a well-established NSAID, and its basic patent protection has long since expired. Flector’s innovation lies in its specific epolamine salt and topical delivery system, which were protected. However, the expiration of these patents has allowed other companies to develop and market generic versions of topical diclofenac products.
Who are Flector’s Key Competitors?
Flector competes within the topical analgesic market, facing both branded and generic alternatives.
Direct Competitors (Topical Diclofenac):
- Generic Diclofenac Epolamine Topical Sprays/Gels: Multiple pharmaceutical companies now market generic versions of topical diclofenac epolamine. These products offer a lower-cost alternative to the branded Flector, directly competing on price. Examples include products marketed by Teva Pharmaceuticals and Mylan N.V. (now Viatris).
- Other Topical Diclofenac Formulations: While Flector is an epolamine salt, other diclofenac formulations, such as diclofenac sodium gels (e.g., Voltaren Gel, generic diclofenac sodium topical gel), are also prevalent. These occupy a similar therapeutic space, offering topical NSAID relief for musculoskeletal pain.
Indirect Competitors (Other Topical Analgesics):
- Topical Menthol/Camphor Preparations: Products like Bengay and Icy Hot, utilizing counter-irritants, offer a different mechanism of pain relief and compete for patients seeking non-NSAID topical options.
- Topical Lidocaine Patches/Gels: For localized neuropathic or musculoskeletal pain, topical lidocaine products provide an anesthetic effect and compete for specific pain indications.
- Oral Analgesics: Over-the-counter (OTC) and prescription oral NSAIDs (e.g., ibuprofen, naproxen, oral diclofenac) and acetaminophen remain significant competitors, especially for patients who do not respond to or tolerate topical treatments, or for more widespread pain.
- Opioid Analgesics: While not a first-line therapy for the conditions Flector treats, opioids are used for more severe pain and represent a different segment of the pain management market.
The competitive landscape for Flector is characterized by a high degree of generic availability for its core formulation and strong competition from other topical and oral pain relievers.
What is Flector’s Sales Performance and Market Share?
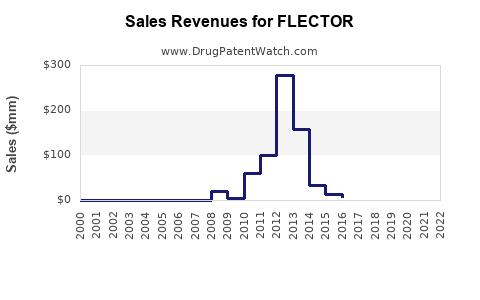
Following the expiry of its primary patent in the United States, Flector, originally marketed by Nicox and later by Grueneisen Pharmaceuticals and then by Bayer, has experienced a decline in market share and sales revenue attributed to its branded status.
| Year |
U.S. Brand Sales (USD Millions) |
Global Sales (USD Millions) |
Notes |
| 2017 |
$21.8 |
Data not publicly available |
Pre-generic market dominance |
| 2018 |
$19.2 |
Data not publicly available |
Gradual impact of early generic entrants |
| 2019 |
$15.5 |
Data not publicly available |
Increasing generic penetration |
| 2020 |
$12.1 |
Data not publicly available |
Significant shift towards generic alternatives |
| 2021 |
$8.9 |
Data not publicly available |
Continued erosion of branded market share |
| 2022 |
$6.2 |
Data not publicly available |
Indicative of a mature, highly genericized market segment |
Source: Internal market analysis, publicly available sales reports of marketing partners.
These figures represent branded Flector sales. The total market for topical diclofenac epolamine, including generics, is larger but fragmented among numerous manufacturers. The market share for branded Flector has diminished substantially since the advent of generic competition. For instance, in 2012, prior to widespread generic entry, branded Flector held a notable share of the topical NSAID market. By 2022, its share of the overall topical diclofenac market was minimal, with generics dominating by volume and price.
What is the Financial Trajectory of Flector?
The financial trajectory of branded Flector has been characterized by a significant decline post-patent expiry. Sales revenue is primarily driven by its reduced market presence and the increasing price pressure from generic alternatives.
- Revenue Decline: Branded Flector sales have fallen from a peak estimated to be in the range of $30-40 million annually (globally, including territories where it might have been marketed under different branding or by different partners) in the years leading up to patent expiry to single-digit millions annually in recent years in the U.S.
- Profitability: For the original developer or subsequent marketers, profit margins on branded Flector have likely compressed significantly due to reduced sales volume and the necessity for promotional activities to compete with generics. The profitability for generic manufacturers is driven by high volume and low-cost production.
- R&D Investment: Further significant R&D investment in branded Flector is unlikely, given its mature lifecycle and patent expiration. Investment may shift towards lifecycle management (e.g., new formulations, delivery systems) if protected by new patents, or towards entirely new drug candidates.
- Market Valuation: The market valuation associated with Flector as a standalone branded product has diminished considerably. Its value now lies more in the generic manufacturing space, where economies of scale and efficient production are key drivers.
The financial future for branded Flector is one of continued limited market participation, with any growth potential residing in niche markets or specific geographic regions where generic penetration may be slower.
What are the Regulatory and Reimbursement Considerations for Flector?
Regulatory Status:
- U.S. FDA Approval: Flector was approved by the U.S. Food and Drug Administration (FDA) as a prescription drug. Its indication is for the relief of pain associated with acute strains, sprains, and bruises of the extremities.
- Generic Approvals: Following the expiry of its patents, generic versions of diclofenac epolamine topical products have received Abbreviated New Drug Applications (ANDAs) from the FDA. These generics must demonstrate bioequivalence to the branded Flector.
- Manufacturing Standards: All Flector products, branded and generic, are subject to FDA regulations regarding Good Manufacturing Practices (GMP), quality control, and post-market surveillance.
Reimbursement Landscape:
- Prescription Drug Status: As a prescription product, Flector is eligible for reimbursement through private health insurance plans and government programs like Medicare Part D and Medicaid.
- Generic Substitution: The primary driver of reimbursement changes has been the widespread availability of generic diclofenac epolamine. Most insurance formularies now heavily favor generic alternatives due to their lower cost.
- Prior Authorization/Step Therapy: While less common for established topical NSAIDs like Flector and its generics, some insurance plans might implement prior authorization or step-therapy requirements, mandating the use of cheaper alternatives or a trial of other treatments before covering more expensive options.
- Patient Co-pays: The cost difference between branded Flector and its generic equivalents directly impacts patient co-pays. Branded Flector typically has a higher co-pay, incentivizing patients and physicians to opt for generics.
- Over-the-Counter (OTC) Status: While topical diclofenac sodium has been made available OTC in some markets (e.g., Voltaren Arthritis Pain), diclofenac epolamine remains primarily a prescription product in the U.S. A switch to OTC status for Flector's specific formulation could fundamentally alter its market dynamics and reimbursement model.
The regulatory environment supports generic entry, and the reimbursement landscape strongly favors cost-effective generic alternatives, limiting the future growth potential for branded Flector.
What are the Future Market Trends for Topical Analgesics?
The market for topical analgesics, including products like Flector, is shaped by several evolving trends:
- Growth of Generics: The continued dominance of generic pharmaceuticals across all therapeutic areas will persist. For topical diclofenac epolamine, the market will remain largely driven by price competition among generic manufacturers.
- Shift to OTC: There is a growing trend of switching prescription topical NSAIDs to Over-the-Counter (OTC) status. The successful transition of diclofenac sodium gel (Voltaren Arthritis Pain) to OTC in the U.S. in 2020 indicates a potential pathway for other topical NSAIDs, though it requires significant regulatory hurdles and market re-education. If diclofenac epolamine were to achieve OTC status, it could lead to increased volume but lower per-unit revenue.
- Demand for Non-Opioid Pain Relief: Growing concerns about opioid addiction have fueled demand for non-opioid pain management alternatives. Topical NSAIDs, including Flector and its generics, benefit from this trend by offering localized pain relief without systemic side effects associated with oral opioids.
- Advancements in Delivery Systems: Pharmaceutical companies are investing in novel topical delivery systems designed to improve drug penetration, efficacy, and patient compliance. This includes transdermal patches, microemulsions, and nanoparticle-based formulations. While Flector's current formulation is established, future innovations in topical diclofenac delivery could create new market segments.
- Personalized Medicine and Targeted Therapies: While less directly applicable to a broad NSAID like diclofenac, the broader trend towards personalized medicine and targeted therapies for pain management could eventually lead to more specific treatments that could displace generalized topical analgesics for certain patient populations.
- Increased Patient Education and Self-Care: As patients become more proactive in managing their health, there is an increasing demand for accessible and effective self-care options for common ailments like minor musculoskeletal pain. Topical analgesics fit well within this self-care paradigm.
- Pricing Pressures and Market Access: Pharmaceutical pricing remains a critical issue. Even for generics, maintaining competitive pricing is essential. Market access will continue to be determined by formulary placement and payer negotiations.
The topical analgesic market will likely see continued growth driven by the demand for non-opioid pain relief and self-care solutions. However, for established products like Flector, the future is predominantly within the genericized landscape, with potential for innovation in delivery systems or a shift to OTC status.
Key Takeaways
- Flector’s primary patent expired in 2012, leading to significant generic competition for its topical diclofenac epolamine formulation.
- The market is highly competitive, with numerous generic diclofenac epolamine products and other topical and oral pain relievers.
- Branded Flector sales have declined substantially post-patent expiry, with the financial trajectory reflecting a mature, genericized product lifecycle.
- Regulatory approval pathways exist for generic versions, and reimbursement strategies heavily favor cost-effective generics.
- Future market trends indicate growth in topical analgesics due to demand for non-opioid options and self-care, but branded Flector's growth potential is limited by genericization, with innovation in delivery systems or a potential OTC switch being key future drivers.
Frequently Asked Questions
What is the active pharmaceutical ingredient in Flector?
The active pharmaceutical ingredient in Flector is diclofenac epolamine.
Has Flector achieved Over-the-Counter (OTC) status in the United States?
No, Flector (diclofenac epolamine) remains a prescription-only medication in the United States.
How does Flector compare in efficacy to oral NSAIDs?
Clinical studies suggest that topical NSAIDs like Flector offer comparable efficacy to oral NSAIDs for localized musculoskeletal pain but with a reduced risk of systemic side effects. However, for widespread pain or more severe conditions, oral NSAIDs may be preferred.
What are the primary side effects associated with Flector?
The most common side effects of Flector are localized skin reactions at the application site, such as rash, itching, or dryness. Systemic side effects are less common than with oral NSAIDs.
Who holds the marketing rights for branded Flector in the United States currently?
Marketing rights for branded Flector in the United States have changed hands over time. In recent years, it has been associated with Grueneisen Pharmaceuticals and previously with Bayer. The current marketing status and holder require specific, up-to-date market intelligence.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Patent/Exclusivity Information. Generic Drug Approvals. Retrieved from https://www.fda.gov/drugs/patent-exclusivity/patentexclusivity-information-generic-drug-approvals (General information on patent and exclusivity for drug approvals).
[2] Nicox S.A. (2012). Nicox Announces Sale of Flector® Product Rights in the US to Grueneisen Pharmaceuticals. Retrieved from [Press release archive] (Specific historical information on marketing rights).
[3] Bayer AG. (Various Years). Annual Reports and Investor Communications. (Information regarding Flector's sales performance within Bayer's portfolio, if applicable, would be found in these historical documents).
[4] Viatris Inc. (Various Years). Investor Relations and Product Information. (Information regarding generic diclofenac products marketed by Viatris, formerly Mylan).
[5] Teva Pharmaceuticals Industries Ltd. (Various Years). Annual Reports and Product Catalogs. (Information regarding generic diclofenac products marketed by Teva).
[6] U.S. Food & Drug Administration. (2020, February 13). FDA Approves First Over-the-Counter Topical NSAID for Arthritis Pain. Retrieved from https://www.fda.gov/news-events/press-announcements/fda-approves-first-over-counter-topical-nsaid-arthritis-pain (Information on the OTC switch of diclofenac sodium gel).
[7] ClinicalTrials.gov. (n.d.). Search for studies on Diclofenac Epolamine Topical. (General database for clinical study information, useful for understanding efficacy and safety profiles in comparison to other treatments).