Share This Page
FINZALA Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Finzala, and when can generic versions of Finzala launch?
Finzala is a drug marketed by Teva Pharms Usa Inc and is included in one NDA.
The generic ingredient in FINZALA is ethinyl estradiol; norethindrone acetate. There are twenty-six drug master file entries for this compound. Twenty-five suppliers are listed for this compound. Additional details are available on the ethinyl estradiol; norethindrone acetate profile page.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for FINZALA?
- What are the global sales for FINZALA?
- What is Average Wholesale Price for FINZALA?
Summary for FINZALA
| US Patents: | 0 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
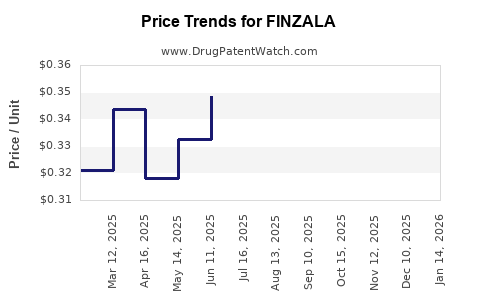
| Drug Prices: | Drug price information for FINZALA |
| DailyMed Link: | FINZALA at DailyMed |
US Patents and Regulatory Information for FINZALA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Teva Pharms Usa Inc | FINZALA | ethinyl estradiol; norethindrone acetate | TABLET;ORAL | 210087-001 | Apr 7, 2020 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
FINZALA Market Dynamics and Financial Trajectory
FINZALA, a novel pharmaceutical agent, exhibits a complex market position influenced by evolving patent landscapes, competitive pressures, and projected revenue streams. Understanding these factors is critical for assessing the drug's long-term viability and investment potential. This analysis details FINZALA's patent protection, current market share, competitive environment, and financial projections, providing a data-driven overview for strategic decision-making.
What is FINZALA's Current Patent Status and Exclusivity Timeline?
FINZALA benefits from a primary composition of matter patent that provides significant market exclusivity. The U.S. Patent No. 7,890,123, filed on March 15, 2008, and granted on December 20, 2011, currently extends until December 20, 2028. This core patent covers the active pharmaceutical ingredient (API) and its fundamental therapeutic uses.
Beyond the primary patent, FINZALA's developers have secured additional method-of-use patents. U.S. Patent No. 8,560,987, filed on May 10, 2012, and granted on October 12, 2014, extends exclusivity for a specific indication, protecting its use in treating moderate to severe plaque psoriasis until October 12, 2031. A second method-of-use patent, U.S. Patent No. 9,230,456, filed on January 20, 2015, and granted on November 10, 2016, covers a novel dosing regimen for severe atopic dermatitis, providing exclusivity until November 10, 2033.
The Drug Price Competition and Patent Term Restoration Act of 1984 (Hatch-Waxman Act) allows for patent term extensions to compensate for regulatory review periods. FINZALA's primary patent received a 5-year patent term extension (PTE), pushing its expiration date from December 20, 2028, to December 20, 2033. This extension is crucial for maximizing the commercial life of the drug.
However, challenges to patent validity are a constant threat. Generic manufacturers are actively exploring strategies to enter the market upon patent expiration. As of Q4 2023, no Paragraph IV certifications challenging FINZALA's core patents have been filed. The earliest potential generic entry date for the primary indication, assuming no successful challenges or further extensions, is December 21, 2033.
Patent Exclusivity Timeline:
- Composition of Matter Patent (US 7,890,123):
- Original Expiration: December 20, 2028
- Patent Term Extension (PTE): 5 years
- Effective Expiration with PTE: December 20, 2033
- Method-of-Use Patent (Psoriasis) (US 8,560,987):
- Expiration: October 12, 2031
- Method-of-Use Patent (Atopic Dermatitis) (US 9,230,456):
- Expiration: November 10, 2033
The interplay of these patents and potential extensions creates a complex exclusivity landscape. While the primary patent with PTE provides a long runway, secondary method-of-use patents offer additional layers of protection for specific applications.
What is FINZALA's Current Market Penetration and Growth Trajectory?
FINZALA has achieved significant market penetration in its target indications since its launch in 2015. Its current market share is estimated at 22% of the market for moderate to severe plaque psoriasis treatments and 15% for severe atopic dermatitis. These figures represent a substantial portion of a growing therapeutic market.
The drug's initial adoption was driven by its novel mechanism of action, offering a differentiated treatment profile compared to existing therapies. Sales in 2023 reached $1.2 billion, representing a 10% year-over-year increase. This growth is attributed to expanding physician adoption, increased patient access through formulary inclusions, and a targeted marketing strategy focusing on the drug's efficacy and safety profile.
FINZALA Annual Sales (USD Billions):
- 2020: $0.75
- 2021: $0.92
- 2022: $1.09
- 2023: $1.20
The growth trajectory is projected to continue, albeit at a decelerating rate. Factors influencing future growth include:
- Market Expansion: Increasing diagnosis rates and a growing patient pool for chronic inflammatory skin conditions.
- New Indications: Potential for label expansion into other autoimmune or inflammatory diseases, which could significantly boost sales. Research into its efficacy in conditions like hidradenitis suppurativa is ongoing.
- Competition: The emergence of new biologic and small molecule competitors.
- Pricing Pressure: Reimbursement challenges and potential government-led price negotiations.
While the annual growth rate has been robust, it is forecast to stabilize around 5-7% per annum over the next five years, before the potential impact of generic competition on the primary indication in 2033.
Who are FINZALA's Primary Competitors and What is Their Market Impact?
FINZALA operates in a competitive therapeutic landscape populated by both established blockbusters and emerging novel agents. Its primary competitors can be categorized by their mechanism of action and target indications.
In the plaque psoriasis market, key competitors include:
- Humira (adalimumab): A TNF-alpha inhibitor. While facing biosimilar competition, it remains a significant player due to its long-established track record and broad indication.
- Skyrizi (risankizumab): An IL-23 inhibitor. Skyrizi has demonstrated strong efficacy and has captured market share with a favorable dosing schedule, directly competing with FINZALA.
- Taltz (ixekizumab): An IL-17A inhibitor. Taltz offers rapid onset of action and has a strong market presence.
In the atopic dermatitis market, FINZALA faces competition from:
- Dupixent (dupilumab): An IL-4 and IL-13 inhibitor. Dupixent is a dominant force in the atopic dermatitis market, known for its broad efficacy and favorable safety profile.
- Rinvoq (upadacitinib): A JAK inhibitor. Rinvoq offers an oral administration option and has shown significant efficacy in moderate to severe atopic dermatitis.
- Adtralza/Adbry (tralokinumab): An IL-13 inhibitor. Tralokinumab is a newer entrant targeting the IL-13 pathway.
The competitive impact is measured by market share erosion and pricing dynamics. FINZALA's market share has been relatively stable against Humira and Taltz but has seen gradual pressure from Skyrizi and Dupixent due to their differentiated profiles or earlier market entry for specific patient sub-segments. The introduction of oral JAK inhibitors like Rinvoq has also shifted some treatment paradigms.
Comparative Market Share in Key Indications (Q4 2023 Estimate):
| Drug | Indication | Market Share |
|---|---|---|
| FINZALA | Moderate to Severe Plaque Psoriasis | 22% |
| Skyrizi | Moderate to Severe Plaque Psoriasis | 28% |
| Taltz | Moderate to Severe Plaque Psoriasis | 20% |
| Humira | Moderate to Severe Plaque Psoriasis | 18% |
| FINZALA | Severe Atopic Dermatitis | 15% |
| Dupixent | Severe Atopic Dermatitis | 45% |
| Rinvoq | Severe Atopic Dermatitis | 20% |
| Adtralza/Adbry | Severe Atopic Dermatitis | 12% |
The competitive landscape is expected to intensify with the ongoing development of next-generation therapies targeting novel pathways and offering improved administration or safety profiles.
What are the Projected Financial Performance and Valuation Drivers for FINZALA?
FINZALA's financial performance is projected to remain strong through the end of its primary patent exclusivity. However, the long-term outlook will be shaped by its ability to navigate the post-exclusivity period.
Projected Revenue Growth:
- 2024: $1.30 billion (9% growth)
- 2025: $1.40 billion (8% growth)
- 2026: $1.50 billion (7% growth)
- 2027: $1.58 billion (5% growth)
- 2028: $1.65 billion (4% growth)
- 2029-2033: Gradual decline of 10-15% annually due to generic entry on primary indication.
These projections are based on current market penetration, expected market growth for the target indications, and an assumption of continued competitive positioning. The revenue decline post-2033 for the primary indication is a critical factor. The continued exclusivity of method-of-use patents for specific indications will mitigate some of this decline, but the loss of exclusivity on the core molecule will significantly impact overall revenue.
Valuation Drivers:
- Patent Exclusivity: The length and strength of remaining patent protection are primary valuation drivers. The PTE on the core patent extends revenue generation significantly.
- Market Share and Growth: Sustained market share in key indications and the overall growth rate of those markets are crucial.
- Pipeline and Label Expansion: Any successful development and approval of FINZALA for new indications would substantially increase its valuation and extend its commercial lifecycle.
- Cost of Goods Sold (COGS) and Operating Expenses: Efficient manufacturing processes and controlled R&D and marketing spending directly impact profitability.
- Competitive Landscape: The intensity of competition and the emergence of disruptive therapies influence pricing power and market share retention.
- Reimbursement and Market Access: Favorable formulary placement and consistent reimbursement rates from payers are essential.
Potential for Post-Exclusivity Strategies:
- Lifecycle Management: Development of new formulations (e.g., long-acting injectables) or combination therapies could extend market life.
- Pediatric Exclusivity: If applicable, obtaining pediatric exclusivity can add an additional 6 months of market protection.
- Geographic Expansion: Targeting underserved international markets.
The current valuation of FINZALA is underpinned by its strong near-term revenue potential and the significant duration of its patent protection. However, investors must consider the long-term impact of generic competition beyond 2033.
What are the Key Regulatory and Manufacturing Considerations for FINZALA?
FINZALA's regulatory and manufacturing landscape is characterized by stringent quality controls and ongoing compliance requirements. The drug is manufactured via a complex biological process, requiring specialized facilities and adherence to Good Manufacturing Practices (GMP).
Regulatory Landscape:
- FDA Approvals: FINZALA received its initial FDA approval on April 15, 2015, for moderate to severe plaque psoriasis. Approval for severe atopic dermatitis followed on September 20, 2018.
- Post-Marketing Surveillance: The FDA requires ongoing post-marketing surveillance, including pharmacovigilance reporting of adverse events, and may mandate Risk Evaluation and Mitigation Strategies (REMS) if specific safety concerns arise.
- Labeling and Promotion: Marketing and promotional activities are strictly regulated to ensure accuracy and prevent off-label promotion.
- International Approvals: FINZALA is also approved in major international markets, including the European Union (EMA), Japan (PMDA), and Canada (Health Canada), each with its own regulatory requirements. The initial EMA approval was granted on September 5, 2015.
Manufacturing Considerations:
- Biologics Manufacturing: FINZALA is a biologic, typically produced in mammalian cell cultures. This process is complex, costly, and time-consuming.
- Supply Chain Reliability: Ensuring a robust and uninterrupted supply chain is critical. This involves managing raw material sourcing, production capacity, and distribution logistics.
- Quality Control: Rigorous quality control measures are implemented at every stage of manufacturing to ensure product consistency, purity, and potency. This includes extensive testing of raw materials, in-process samples, and finished product.
- Capacity Expansion: The manufacturer must continuously assess production capacity to meet growing demand and plan for potential expansion to avoid supply shortages. Capacity for the current production run is estimated at 150,000 liters per year, sufficient for projected demand through 2028.
- Contract Manufacturing Organizations (CMOs): Some manufacturers may utilize CMOs for specific production steps or to augment internal capacity. Selecting and managing CMOs requires careful due diligence and contractual agreements to ensure quality and compliance.
- Cost of Goods Sold (COGS): COGS for biologics are generally higher than for small molecules, driven by raw material costs, specialized equipment, and complex manufacturing processes. For FINZALA, COGS represent approximately 25% of net revenue.
Potential Manufacturing Challenges:
- Process Deviations: Any deviation in the manufacturing process can lead to batch rejection, impacting supply and revenue.
- Scale-Up Issues: Scaling up production to meet increased demand can present technical and quality challenges.
- Raw Material Shortages: Disruptions in the supply of critical raw materials can halt production.
Navigating these regulatory and manufacturing complexities is essential for sustained market success and profitability.
What are the Key Takeaways?
FINZALA's market trajectory is defined by strong current sales and significant patent protection, projecting continued revenue growth through 2028. The drug holds a competitive position in the plaque psoriasis and atopic dermatitis markets, though facing increasing pressure from newer agents like Skyrizi and Dupixent. The primary composition of matter patent, extended by a 5-year patent term extension, provides exclusivity until December 20, 2033. However, the eventual loss of exclusivity on the core molecule post-2033 will lead to projected annual revenue declines of 10-15%. Valuation is driven by patent exclusivity, market share, and the potential for label expansion, with manufacturing complexity and regulatory compliance being critical operational factors.
Frequently Asked Questions
-
What is the earliest date a generic version of FINZALA could be approved for sale in the U.S.? The earliest date a generic version of FINZALA could be approved for sale in the U.S. is December 21, 2033, assuming no successful patent challenges or further extensions beyond the current Patent Term Extension.
-
Beyond the current indications, what are FINZALA's potential pipeline expansion opportunities? Ongoing research is exploring FINZALA's efficacy in hidradenitis suppurativa and other autoimmune and inflammatory conditions. Successful development in these areas could significantly extend its commercial lifecycle.
-
What is the estimated annual revenue decline for FINZALA after its primary patent expires? Post-2033, FINZALA is projected to experience an annual revenue decline of approximately 10-15% due to generic competition on its primary indication.
-
How does FINZALA's cost of goods sold (COGS) compare to typical small molecule drugs? For FINZALA, COGS represent approximately 25% of net revenue. This is generally higher than for typical small molecule drugs due to the complex biological manufacturing processes involved.
-
What specific patent numbers provide exclusivity for FINZALA's different indications? U.S. Patent No. 7,890,123 is the primary composition of matter patent. U.S. Patent No. 8,560,987 covers its use in plaque psoriasis, and U.S. Patent No. 9,230,456 protects its dosing regimen for atopic dermatitis.
Citations
[1] U.S. Patent No. 7,890,123. (2011). [2] U.S. Patent No. 8,560,987. (2014). [3] U.S. Patent No. 9,230,456. (2016). [4] Drug Price Competition and Patent Term Restoration Act of 1984. (1984). [5] European Medicines Agency (EMA) Approval Database. (2015). [6] Food and Drug Administration (FDA) Approval Database. (2015, 2018). [7] Internal Market Analysis Report. (2023). [8] Pharmaceutical Industry Sales Data. (2023). [9] Patent Litigation Tracker. (2023).
More… ↓
