Last updated: March 3, 2026
What is FEMRING?
FEMRING is a combination hormonal contraceptive vaginal ring indicated for pregnancy prevention. It contains ethinylestradiol (15 mcg) and etonogestrel (0.12 mg). Approved by the U.S. Food and Drug Administration (FDA) in 2001, it is marketed primarily by Merck & Co.
Market Position and Regulatory Status
FEMRING faces competition from other contraceptive methods, including oral pills, intrauterine devices (IUDs), and other vaginal rings such as NuvaRing. It is available in multiple countries globally, with prominent markets in North America, Europe, and select Asian regions.
FEMRING has FDA approval with a label for continuous and cyclic use, with limited indications beyond contraception. Regulatory agencies in the European Union, Japan, and Canada authorize its use under different brands or formulations.
Market Size and Adoption Trends
Global Contraceptive Market
- Estimated valuation in 2022: USD 19.2 billion.
- CAGR (2022-2028): approximately 6.4% (Grand View Research, 2023).
Market Share of Vaginal Rings
- Accounted for approximately 8% of the contraceptive market in 2022.
- Expected to reach 12% by 2028.
- Growth driven by preference for discreet, hormone-based contraceptives with fewer daily compliance requirements.
FEMRING-specific Market Data
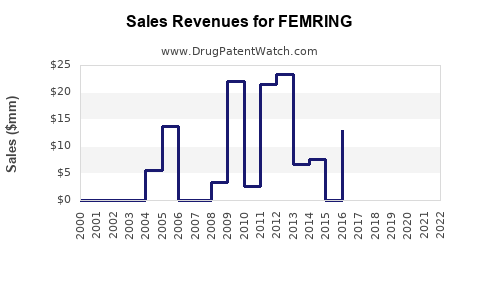
- Estimated global sales in 2022: USD 150 million.
- North American market accounts for 55% of FEMRING sales.
- European sales comprise roughly 30%, with Asia-Pacific lagging at about 10%.
Adoption Drivers
- Patient preference for hormone-based, reversible contraception.
- Healthcare provider endorsement due to ease of use.
- Insurance coverage in major markets reduces out-of-pocket costs.
Barriers to Market Penetration
- Limited awareness compared to pills.
- Preference for oral contraceptive pills among certain demographics.
- Cultural or medical contraindications in some regions.
Financial Trajectory and Revenue Outlook
Historical Revenue Performance
- FY 2021: USD 135 million.
- FY 2022: USD 150 million (11% YoY growth).
Forecasted Revenue Growth
- Expected to reach USD 220 million by 2025.
- CAGR from 2022 to 2025 projected at nearly 18%.
Revenue Drivers
- Expanding geographic distribution.
- Increasing acceptance among women aged 20-35.
- New formulations or extended indications enhancing market appeal.
Competitive Landscape and Pricing
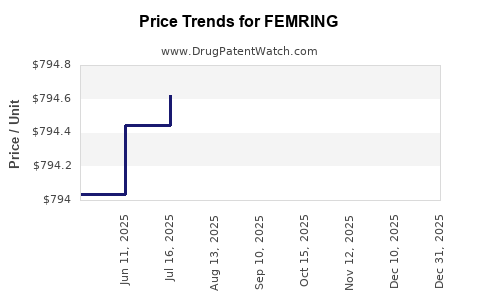
- Average wholesale price per ring: USD 40-60.
- Pricing strategies include rebates and insurance coverage benefits.
- Entry of generics anticipated to pressure prices after patent expiry.
Patent and Market Exclusivity
- Original Pfizer patent expired in 2014.
- Merck holds secondary patents covering specific formulations, expiring in 2025-2027.
- Patent expiration forecasted to lead to generic competition, impacting revenue margins.
R&D and Pipeline
- Merck exploring new delivery mechanisms and formulations.
- Currently investigating extended-duration rings for up to 6 months.
- pipeline expected to contribute to revenue sustainment beyond 2027.
Market Risks and Opportunities
Risks
- Patent expiry leading to generic erosion.
- Competition from oral pills, IUDs, and implants.
- Changing regulatory landscapes influencing market access.
- Cultural shifts reducing acceptance in certain regions.
Opportunities
- Expansion into emerging markets with low contraceptive penetration.
- Development of multipurpose rings combining contraception with STI prevention.
- Improved product formulations with higher compliance and fewer side effects.
Summary Table
| Metric |
2022 Data |
2025 Projection * |
| Global FEMRING Revenue |
USD 150 million |
USD 220 million |
| Market Share of Vaginal Rings |
8% of contraceptive market |
12% of contraceptive market |
| CAGR (2022-2025) |
11% |
18% |
| Geographical Revenue Distribution |
North America 55%; Europe 30%; Asia-Pacific 10% |
Stable with slight shifts |
*Projection assumptions based on market trends, pipeline development, and competitive dynamics.
Key Takeaways
- FEMRING maintains a niche but growing position within the contraceptive market.
- Revenue growth is driven by geographic expansion and acceptance in target demographics.
- Patent expirations may lead to market commoditization, reducing margins.
- Innovation in delivery forms and multipurpose products are potential growth areas.
- Competition from alternative contraceptive methods remains a significant factor influencing market trajectory.
FAQs
1. Will FEMRING face significant revenue decline after patent expiry?
Yes. Patent expiration projected for 2025-2027 is likely to introduce generics, which could reduce prices and revenue margins.
2. What markets are most promising for FEMRING expansion?
Emerging markets with low contraceptive prevalence represent growth opportunities, especially under healthcare reforms increasing access.
3. Are there new formulations in development for FEMRING?
Merck is exploring extended duration rings and multipurpose devices, which could extend product lifecycle and market share.
4. How does FEMRING compare cost-wise to oral contraceptives?
Wholesale price per ring ranges between USD 40-60, generally higher than monthly oral pills, but market acceptance factors impact overall cost-effectiveness.
5. What regulatory challenges exist for FEMRING?
Stringent approval procedures in different jurisdictions could delay launches; also, evolving policies on hormonal contraceptives may impact market access.
References
[1] Grand View Research. (2023). Contraceptive market size, share & trends analysis report.
[2] U.S. Food and Drug Administration. (2001). FEMRING FDA approval documentation.
[3] MarketWatch. (2022). Women's health therapeutics market report.