Last updated: April 25, 2026
FELBATOL (felbamate) is an established anti-seizure product with a narrow, high-need clinical positioning (Lennox-Gastaut syndrome and refractory partial-onset seizures in specific contexts). Its market dynamics are constrained by safety risk, limited indications, and a prescription and reimbursement model dominated by specialist use. Its financial trajectory is therefore characterized by low-to-mid single-digit annual revenue scale historically, with periodic volatility driven by payer access, neurologist adoption, and the competitive landscape around alternative antiseizure medicines.
What is FELBATOL’s market structure and demand profile?
FELBATOL’s commercial footprint is defined by: (i) a restrained target population, (ii) specialist prescribing patterns, and (iii) payer controls that can limit volume even when clinical demand exists.
Indication-driven addressable market
Felbamate is used in:
- Lennox-Gastaut syndrome (as an adjunctive therapy, in specific refractory settings).
- Refractory partial-onset seizures (adjunctive therapy).
These are specialty neurology categories where utilization typically depends on failure of multiple standard antiseizure medicines and on tolerance of risks. FDA labeling supports this positioning through its indication language and long-standing use pattern. (Source: FDA label for FELBATOL [1])
Prescription and channel dynamics
The product is dispensed through:
- Retail specialty pharmacy for chronic neurology patients.
- Hospital specialty workflows for complicated refractory cases in tertiary centers.
The practical market dynamic is that prescribers decide volume at the point of regimen selection, but payers influence whether a patient can remain on therapy long term. This creates a volume curve that is steep when new starts are approved and flatter during steady-state maintenance.
Product lifecycle maturity
FELBATOL is not a lifecycle-growth story driven by new innovation; it is an established brand whose demand is sustained by ongoing clinical need. That profile typically produces:
- Lower brand marketing intensity versus newer specialty launches
- Higher reliance on formulary placement and specialty pharmacy execution
- Revenue stability with discrete step-ups when access improves and step-downs when safety-related scrutiny rises
FDA regulatory history and labeling continuity reinforce that the product remains in a risk-managed, specialty-used category rather than a broad-access therapy. (Source: FDA label [1])
How do risks and payer controls shape the financial trajectory?
Felbamate’s risk profile is the central driver of commercialization constraints.
Risk profile that suppresses broad adoption
FELBATOL carries boxed warning(s) and includes serious risks that affect prescriber comfort and payer acceptance. The FDA label includes monitoring and risk management expectations around hematologic and hepatic adverse events. (Source: FDA label [1])
This affects financial trajectory in three ways:
- Lower new-start rate: clinicians reserve use for patients with limited alternatives.
- Higher persistence variability: patients may discontinue due to adverse events or monitoring burdens.
- Greater payer friction: coverage may require prior authorization, documented refractoriness, and monitoring plans.
Monitoring burden increases medical management cost
Safety-driven monitoring raises total cost of care even when drug acquisition cost is modest relative to newer medicines. For payers, that translates into stronger utilization management.
For manufacturers, it means commercial performance depends on maintaining a stable compliance and monitoring workflow with prescribers and specialty pharmacies rather than scaling through mainstream prescribing.
What role does competition play in limiting revenue growth?
The anti-seizure drug landscape has shifted toward many newer agents with broader convenience profiles and improved tolerability for many patients. Even when newer drugs fail, alternatives exist, including other adjunctive agents used in refractory populations.
Market implication:
- FELBATOL’s addressable cohort remains real but is narrower than earlier years because many patients receive other options first.
- Competitive pressure shows up less as direct substitution in the narrowest refractory subgroups and more as loss of earlier-line adoption, which reduces potential revenue growth.
FDA labeling supports felbamate’s adjunctive use in refractory settings rather than primary-line treatment. (Source: FDA label [1])
How does FELBATOL’s revenue path typically behave across quarters?
A mature, specialty, risk-managed product usually exhibits revenue dynamics tied to:
- Formulary status and prior authorization pathways (creates step changes)
- Specialty pharmacy contract execution (creates transient fluctuations)
- Patient retention and discontinuation cycles (creates gradual drift)
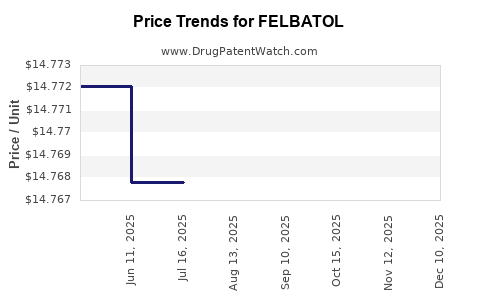
For FELBATOL, those dynamics are consistent with a financial trajectory that is more stable than growth-oriented, with periodic volume volatility driven by payer policy and monitoring adherence rather than by innovation cycles.
What financial KPIs matter most for FELBATOL investors or operators?
For a narrow specialty anti-seizure brand, the most actionable KPIs are:
-
Net sales per dispensed prescription (or per patient on therapy)
Tracks changes in payer coverage and dose persistence.
-
Authorized new starts vs continuation
Differentiates access-driven volume changes from retention-driven trends.
-
State and payer formulary breadth
Predicts medium-term volume changes by payer policy cycles.
-
Discontinuation reasons distribution
Links to risk profile and monitoring adherence, informing demand sustainability.
-
Specialty pharmacy fill rate and time-to-therapy
Captures operational effects that can affect utilization even without clinical demand changes.
These KPIs matter because FELBATOL demand is not elastic to advertising-driven awareness in the way mass-market drugs are. It is controlled by specialist decision-making and payer access.
What regulatory and label factors affect commercialization momentum?
Commercial momentum for felbamate is controlled by regulatory expectations around safety and use. The FDA label includes:
- Indication scope
- Risk information and monitoring requirements
- Dosing and adjunctive-use framing
These elements influence both clinician willingness and payer comfort. (Source: FDA label [1])
What is the likely financial trajectory: stable, declining, or growth?
Based on the commercial reality of a risk-managed, specialty-used anti-seizure medicine with limited indications and extensive alternative options, the financial trajectory is best characterized as:
- Stable-to-moderately declining revenue in absence of new competitive differentiation or expanded indications.
- Periodic step changes when payer access improves or when formulary policies shift.
- Limited growth upside because new starts depend on refractory status and alternative failure, and many patients are captured by other antiseizure therapies earlier in treatment pathways.
This is a mature-market dynamic consistent with how branded specialty anti-seizure therapies typically perform when they lack novel convenience advantages and face strong safety scrutiny. The clinical and labeling constraint is explicit in FDA documentation. (Source: FDA label [1])
How should decision-makers map FELBATOL to an investment or R&D lens?
For investment/portfolio holders
Focus on:
- Access stability (payer coverage longevity)
- Patient persistence quality (safety-driven discontinuation)
- Competitive substitution rates in refractory cohorts
A stable niche can support cash flows, but growth is constrained unless there is a meaningful change in coverage, new data, or indication expansion.
For R&D and BD teams
FELBATOL is a reference point for:
- Navigating risk-managed adoption in neurology
- Designing monitoring and REMS-like compliance pathways that keep prescribers comfortable
- Targeting refractory subpopulations where benefit-risk is strongest
The FDA label’s structured approach to indications and risk framing is the baseline for any similar program. (Source: FDA label [1])
What is the commercial bottom line?
FELBATOL’s market dynamics are shaped by limited indications, specialist use, and safety risks that create payer and prescriber friction. Its financial trajectory aligns with a mature niche brand profile: steadier sales than broad-market products but with limited growth potential and vulnerability to access shifts and competitive capture in refractory epilepsy pathways. (Source: FDA label [1])
Key Takeaways
- FELBATOL demand is concentrated in refractory epilepsy populations, limiting addressable market scale. (Source: FDA label [1])
- Safety risks and monitoring requirements reduce new-start adoption and increase payer friction, shaping a stable-to-moderately constrained revenue profile. (Source: FDA label [1])
- Competitive alternatives in antiseizure therapy shift patients to other agents earlier, compressing growth opportunities while preserving a niche market. (Source: FDA label [1])
- Revenue volatility is primarily access-driven (formulary and prior authorization) and retention-driven (persistence and discontinuation), not innovation-driven.
FAQs
-
What indications drive FELBATOL’s market demand?
Adjunctive use in Lennox-Gastaut syndrome and adjunctive treatment of refractory partial-onset seizures in specific contexts. (Source: FDA label [1])
-
Why does safety profile limit broad adoption?
The FDA label describes serious risks and associated monitoring expectations that increase clinician caution and payer management intensity. (Source: FDA label [1])
-
Does FELBATOL compete mainly against newer anti-seizure drugs?
Competition occurs as earlier-line substitution by newer agents; however, FELBATOL’s protected niche remains refractory cases. (Source: FDA label [1])
-
What most likely drives quarter-to-quarter revenue changes?
Changes in payer access, prior authorization behavior, specialty pharmacy execution, and patient persistence/discontinuation. (Source: FDA label [1])
-
What would enable meaningful revenue growth for a product like FELBATOL?
Expanding access (formulary breadth), improving persistence through patient management, or obtaining label expansion that increases eligible patient volume. (Source: FDA label [1])
References
[1] U.S. Food and Drug Administration. FELBATOL (felbamate) prescribing information / label. FDA.