Last updated: February 19, 2026
This analysis examines the market dynamics and financial trajectory of EXPAREL, a liposomal bupivacaine injectable suspension marketed by Pacira BioSciences. Key patent expiries, competitive landscapes, and revenue performance are assessed to inform R&D and investment strategies.
What is EXPAREL and Its Core Technology?
EXPAREL is a non-opioid analgesic that utilizes DepoFoam drug delivery technology. This technology encapsulates the anesthetic bupivacaine within a biodegradable, multivesicular liposome. The liposomes are designed to release bupivacaine gradually over an extended period, providing prolonged pain relief following surgical procedures. The extended-release mechanism allows for a single administration to deliver pain management for up to 72 hours, thereby reducing the reliance on systemic opioid analgesics post-surgery.
The mechanism of action involves the slow dissolution of the liposome structure in the interstitial fluid at the injection site. As the liposomes degrade, bupivacaine is released incrementally, interacting with voltage-gated sodium channels on nerve fibers to inhibit nerve impulse transmission. This sustained local anesthetic effect is the primary differentiator for EXPAREL.
What are the Approved Indications and Target Patient Populations for EXPAREL?
EXPAREL has received regulatory approval for several indications across various surgical specialties, targeting patients undergoing these procedures.
Key Approved Indications:
- Hemorrhoidectomy: Approved by the U.S. Food and Drug Administration (FDA) in 2012. This was the initial indication.
- Major Depressive Disorder (MDD): Approved in 2014 as an adjunct to established antidepressant therapies. This indication was later withdrawn due to strategic commercial decisions by Pacira BioSciences, reflecting a refocusing of resources.
- Minimally Invasive Breast Augmentation: Approved in 2015.
- Minimally Invasive Abdominoplasty: Approved in 2015.
- Hernia Repair: Approved in 2016.
- Total Knee Arthroplasty (TKA): Approved in 2017.
- Total Hip Arthroplasty (THA): Approved in 2017.
- Deltoid Intramuscular Injection: Approved in 2017 for localized pain management in adults.
- Trauma: Approved in 2017 for management of post-surgical pain in patients 6 years of age and older following lower extremity
injuries.
- Post-Surgical Pain Management (Broad Label): Approved in 2018 for management of post-surgical pain in patients 12 years of age and older. This expanded label covers a wide range of surgical procedures.
- Intercostal Nerve Block: Approved in 2019 for intercostal nerve blocks to provide analgesia following thoracotomy.
- Bunionectomy: Approved in 2021 for patients 18 years of age and older.
- Arthroscopic Surgery: Approved in 2021 for patients 18 years of age and older.
The target patient populations encompass individuals undergoing surgical procedures where moderate to severe pain is anticipated and prolonged relief is desired without the immediate need for opioid interventions. This includes patients in orthopedic surgery (knee and hip replacements, foot surgery), general surgery (hernia repair, abdominoplasty), plastic surgery, and trauma.
What is the Intellectual Property Landscape and Patent Expiry Timeline for EXPAREL?
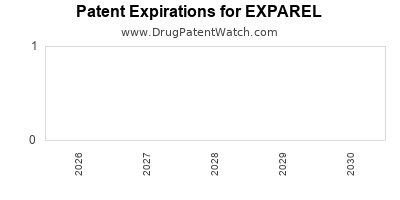
The intellectual property portfolio surrounding EXPAREL is critical to its commercial exclusivity. Pacira BioSciences has secured patents covering the composition of matter, formulation, method of use, and manufacturing processes. The primary patents protecting EXPAREL are the U.S. patents that will ultimately expire, opening the door for generic competition.
Key Patent Information and Expiry:
- U.S. Patent No. 7,011,847: This patent, titled "Liposome formulations of local anesthetics and uses thereof," covers the core liposome formulation of bupivacaine.
- Original Expiration: November 10, 2023.
- Patent Term Extension (PTE): A PTE was granted, extending the patent term by approximately 640 days. This extension pushed the expiration date to August 15, 2025.
- U.S. Patent No. 8,415,342: This patent covers methods of treating pain with EXPAREL and has an original expiration date of March 25, 2029.
- U.S. Patent No. 9,603,837: This patent, titled "Method of treating postsurgical pain with a liposomal bupivacaine formulation," covers the use of EXPAREL for post-surgical pain management. Its original expiration is May 28, 2030.
- U.S. Patent No. 9,649,439: This patent relates to specific dosage and administration methods. Its original expiration is May 16, 2034.
- U.S. Patent No. 9,943,525: This patent relates to methods of treating pain with EXPAREL. Its original expiration is March 21, 2035.
- U.S. Patent No. 10,195,207: This patent relates to methods of manufacturing EXPAREL. Its original expiration is December 15, 2035.
- U.S. Patent No. 10,675,322: This patent relates to methods of treating pain with EXPAREL. Its original expiration is December 15, 2035.
Key Observations on IP:
- The expiry of U.S. Patent No. 7,011,847 on August 15, 2025, represents the first significant patent cliff for the core formulation.
- Later-expiring patents, particularly those covering methods of use and manufacturing processes, provide a more extended period of protection.
- Litigation surrounding these patents has occurred, with varying outcomes impacting generic entry timelines. The landscape is subject to ongoing legal challenges.
What is the Competitive Landscape for EXPAREL?
The competitive landscape for EXPAREL includes other local anesthetics, opioid analgesics, and emerging non-opioid pain management strategies.
Direct Competitors (Local Anesthetics):
- Bupivacaine (standard formulation): Traditional bupivacaine hydrochloride injectables offer local anesthesia but lack the prolonged duration of action and sustained release of EXPAREL. These are significantly lower cost.
- Ropivacaine: Another long-acting local anesthetic used for similar indications. It provides a shorter duration of action compared to EXPAREL and is also available in standard formulations.
- Generic Liposomal Bupivacaine: The primary competitive threat will emerge from generic versions of EXPAREL once key patents expire and any associated litigation is resolved. The first generic entry is anticipated following the expiry of the '847 patent.
Indirect Competitors (Pain Management Modalities):
- Opioid Analgesics: While EXPAREL is positioned as a non-opioid alternative, opioids remain a standard of care for managing severe post-surgical pain. Their widespread availability and established reimbursement structures present a persistent competitive challenge, despite the push for opioid-sparing strategies.
- Non-Steroidal Anti-Inflammatory Drugs (NSAIDs): Systemic NSAIDs are commonly used for pain management and may be used in conjunction with or as an alternative to EXPAREL in certain scenarios.
- Regional Anesthesia Techniques: Epidurals, spinal blocks, and peripheral nerve blocks can provide effective pain relief but often require specialized administration and monitoring.
- Other Non-Opioid Analgesics: Acetaminophen and other non-opioid medications are used for pain management.
- Emerging Technologies: Research into novel pain management agents, including other long-acting local anesthetics, gene therapies, and neuromodulation techniques, represents future competitive pressures.
Market Dynamics Influencing Competition:
- Opioid Crisis: The ongoing opioid crisis continues to drive demand for non-opioid pain management solutions, benefiting EXPAREL.
- Value-Based Healthcare: Increased focus on healthcare economics and patient outcomes supports products that can demonstrate reduced hospital stays, fewer readmissions, and lower overall healthcare costs. EXPAREL's ability to reduce opioid consumption and potentially shorten hospital stays aligns with these initiatives.
- Payer Reimbursement: Reimbursement policies from private payers and government programs significantly impact market adoption. Favorable formulary placement and reimbursement rates for EXPAREL are crucial.
- Physician Adoption: Clinician familiarity, training, and perceived efficacy are key drivers of prescribing behavior.
What is the Revenue Performance and Financial Trajectory of EXPAREL?
EXPAREL has been a significant revenue driver for Pacira BioSciences, demonstrating consistent growth since its launch. Its financial trajectory is marked by increasing sales volumes, expansion into new indications, and the impact of commercial strategies.
Revenue Data (USD Millions):
| Year |
Net Sales of EXPAREL |
% Change (YoY) |
| 2019 |
$377.9 |
11.8% |
| 2020 |
$394.6 |
4.4% |
| 2021 |
$465.3 |
17.0% |
| 2022 |
$513.8 |
10.4% |
| 2023 |
$601.9 |
17.1% |
Data Source: Pacira BioSciences Annual Reports and SEC Filings.
Analysis of Revenue Trends:
- Consistent Growth: EXPAREL has shown a sustained upward trend in net sales.
- Impact of New Indications: The expansion into broader surgical indications, particularly total joint arthroplasty and broad post-surgical pain management, has been a key driver of growth.
- COVID-19 Impact: The company experienced a dip in growth in 2020, likely due to the widespread cancellation and postponement of elective surgeries during the COVID-19 pandemic. Recovery and accelerated growth were observed in subsequent years as surgical volumes rebounded.
- Strategic Focus: Pacira's strategic decisions, such as the withdrawal of the MDD indication, have allowed for greater concentration on its core surgical pain management franchise.
- Outlook: Projections indicate continued growth, although the rate may moderate as it approaches the patent cliff. The introduction of generics will be a significant factor influencing future revenue streams and potentially leading to price erosion.
Profitability and Margin Considerations:
EXPAREL, as a proprietary specialty pharmaceutical, typically commands higher gross margins compared to generic drugs. However, its manufacturing process, involving liposome encapsulation, is complex and can contribute to higher cost of goods sold. Research and development expenses related to new indications, ongoing clinical studies, and defense of intellectual property also impact net profitability.
What are the Key Growth Drivers and Strategic Considerations for EXPAREL?
The future growth of EXPAREL hinges on several key drivers and strategic imperatives.
Key Growth Drivers:
- Expansion into Underserved Surgical Procedures: Identifying and securing market access in surgical specialties where long-acting local anesthesia is underutilized.
- Value Proposition Demonstration: Continuously providing robust health economic data demonstrating reduced overall healthcare costs (e.g., shorter lengths of stay, fewer opioid-related adverse events, reduced readmission rates) to payers and providers.
- Physician Education and Advocacy: Ongoing training programs for surgeons and anesthesiologists on the appropriate use and benefits of EXPAREL.
- Market Penetration in Existing Indications: Increasing the adoption rate within already approved surgical areas, particularly in large-volume procedures like total joint replacements.
- Geographic Expansion: Exploring and executing market entry into international markets where appropriate regulatory approvals can be obtained.
Strategic Considerations Moving Forward:
- Generic Competition Preparedness: Developing a robust strategy to mitigate the impact of generic entrants post-patent expiry. This may involve:
- Developing next-generation formulations or delivery systems.
- Focusing on value-added services or patient support programs.
- Leveraging remaining patent protection on methods of use.
- Exploring strategic partnerships or licensing agreements.
- Lifecycle Management: Investigating opportunities for new indications or expanded use within existing therapeutic areas that can extend the product's commercial life.
- Portfolio Diversification: While EXPAREL is a cornerstone, Pacira BioSciences will likely continue to explore diversification through pipeline development or strategic acquisitions to reduce reliance on a single product.
- Pricing and Access Strategies: Adapting pricing models and actively engaging with payers to ensure continued access and reimbursement in a challenging healthcare environment.
Key Takeaways
- EXPAREL's core technology, DepoFoam, provides sustained pain relief, differentiating it from standard local anesthetics.
- The product has secured multiple FDA approvals across various surgical indications, targeting patients requiring post-operative pain management.
- Key U.S. patents protecting the core formulation expire in August 2025, signaling the eventual arrival of generic competition.
- Pacira BioSciences has achieved significant revenue growth with EXPAREL, driven by expanding indications and the market's shift towards non-opioid pain management.
- The competitive landscape includes traditional local anesthetics, opioids, and emerging pain management therapies, with generic liposomal bupivacaine posing the most direct future threat.
- Future growth will depend on continued market penetration, demonstrating value to payers, and strategic lifecycle management to address patent expirations.
Frequently Asked Questions
-
When is the primary U.S. patent for EXPAREL set to expire, allowing for generic entry?
The U.S. patent covering the core liposome formulation of EXPAREL (U.S. Patent No. 7,011,847), with a Patent Term Extension, is scheduled to expire on August 15, 2025.
-
What are the key approved indications for EXPAREL that contribute to its market success?
Key indications include post-surgical pain management following total knee and hip arthroplasty, hernia repair, bunionectomy, and broad post-surgical pain management in patients 12 years of age and older.
-
How has the opioid crisis influenced the market for EXPAREL?
The opioid crisis has served as a significant tailwind for EXPAREL by increasing demand for non-opioid analgesics that offer sustained pain relief without the risks associated with opioid dependence.
-
What are the primary revenue-generating indications for EXPAREL?
While specific revenue breakdowns by indication are not publicly disclosed, indications such as total joint arthroplasty and broader post-surgical pain management are considered major contributors due to the high volume of these procedures.
-
Beyond patent expiry, what other competitive pressures does EXPAREL face?
EXPAREL faces competition from other long-acting local anesthetics, continued reliance on opioid analgesics for severe pain, and the ongoing development of novel non-opioid pain management therapies and regional anesthesia techniques.
Citations
[1] Pacira BioSciences, Inc. (n.d.). Annual Reports. U.S. Securities and Exchange Commission. Retrieved from https://www.sec.gov/edgar/search-and-access
[2] U.S. Patent and Trademark Office. (n.d.). Patent Search. Retrieved from https://www.uspto.gov/patents/search