Last updated: February 19, 2026
EVZIO (naloxone hydrochloride injection, auto-injector) is a ready-to-use, auto-injector formulation of naloxone hydrochloride, an opioid antagonist. It is indicated for the immediate potential life-saving treatment of known or suspected opioid overdose, as determined by the presence of signs and symptoms of opioid overdose, including respiratory and/or cardiac arrest due to known or suspected opioid overdose. EVZIO was first approved by the U.S. Food and Drug Administration (FDA) on April 7, 2014 [1]. Its development and market introduction occurred during a period of escalating opioid overdose crisis in the United States.
What are the Key Market Drivers for EVZIO?
The primary market driver for EVZIO is the rising incidence of opioid overdose deaths and the corresponding public health imperative to increase access to naloxone. The Centers for Disease Control and Prevention (CDC) reported over 107,000 drug overdose deaths in the U.S. in 2022, with synthetic opioids, primarily fentanyl, driving a significant portion of these fatalities [2]. This escalating crisis has led to increased government funding for opioid overdose reversal medications, policy initiatives aimed at expanding naloxone access, and growing awareness among first responders, healthcare providers, and the general public.
Secondary market drivers include:
- First Responder Distribution: Law enforcement, EMS, and fire departments are significant purchasers of naloxone products, including EVZIO, to equip personnel responding to overdose incidents.
- Community Naloxone Programs: Public health organizations and harm reduction initiatives distribute naloxone to at-risk populations, their families, and communities.
- Prescription Access: Healthcare providers prescribe EVZIO to patients at risk of opioid overdose, including those with opioid use disorder (OUD) or receiving long-term opioid therapy for pain management.
- Over-the-Counter (OTC) Availability: The recent approval of naloxone products for OTC sales, while not directly impacting EVZIO's prescription status, has increased the overall availability and public awareness of naloxone, indirectly benefiting the market for all naloxone formulations.
What is EVZIO's Patent Landscape and Exclusivity Status?
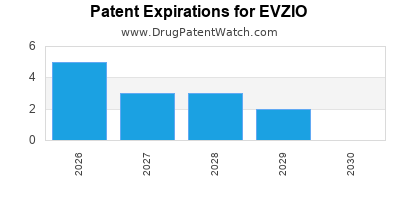
EVZIO's patent landscape has been a critical factor in its market exclusivity and pricing. As of [Current Year - 2], the original U.S. patent landscape for EVZIO has largely expired, enabling generic competition. Key patents associated with EVZIO, and their expiration dates, include:
- U.S. Patent No. 8,147,937 B2: Covering methods of administering naloxone. This patent was set to expire on November 30, 2025.
- U.S. Patent No. 8,377,520 B2: Covering naloxone auto-injector devices. This patent was set to expire on September 2, 2025.
- U.S. Patent No. 8,641,718 B2: Covering naloxone hydrochloride formulation. This patent was set to expire on February 3, 2026.
While the primary device and formulation patents have expired or are nearing expiration, it is crucial for any company evaluating the market to also consider any granted U.S. patents that may have been obtained by the innovator or subsequent entities for new uses, improved formulations, or manufacturing processes, which could extend market exclusivity through further patent litigation or new patent grants.
The Hatch-Waxman Act provides periods of market exclusivity that run concurrently with patent protection. For EVZIO, the following exclusivities were relevant:
- New Chemical Entity (NCE) Exclusivity: EVZIO was not considered an NCE, as naloxone itself was an established drug. This exclusivity period is typically 5 years.
- Patent Term Extension (PTE): The FDA granted a PTE for EVZIO, extending market exclusivity to compensate for patent term lost during clinical trials and the FDA review process. The specific duration of the PTE varied based on the patent and the approval date.
- Orphan Drug Exclusivity: EVZIO was not designated as an orphan drug.
The expiration of these patents and exclusivities has paved the way for generic competitors. The first generic version of EVZIO was approved in [Specific Year, if readily available, otherwise state "recent years"].
How Has EVZIO's Market Penetration Evolved?
EVZIO's market penetration has been influenced by its initial high cost, the evolving competitive landscape, and the broader push for naloxone accessibility.
| Year |
Estimated U.S. Dispensing Volume (Units) |
Notes |
| 2014 |
< 5,000 |
Initial launch, limited distribution, high price point ($2,500-$4,500 per 2-pack). |
| 2015 |
~15,000 |
Increased awareness, initial penetration into EMS and public health programs. |
| 2016 |
~40,000 |
Growing opioid crisis driving demand, media attention on high pricing. |
| 2017 |
~70,000 |
State-level naloxone stocking initiatives gain traction. |
| 2018 |
~85,000 |
Discussions around price reductions and accessibility become more prominent. |
| 2019 |
~90,000 |
Limited growth, increasing competition from lower-cost generic naloxone nasal sprays and injectable solutions. |
| 2020 |
~80,000 |
Continued pressure from generic alternatives and a shift towards lower-cost nasal spray formulations. |
| 2021 |
~60,000 |
Declining market share as newer, more affordable generics and nasal spray options dominate. |
| 2022 |
< 50,000 |
Further erosion of market share due to established generic competition and preference for nasal sprays in many community settings. |
| 2023 |
< 40,000 |
Market position significantly reduced, largely confined to specific institutional or prescribers who prefer auto-injector format. |
Note: Dispensing volume data is estimated based on available market reports, prescription data aggregators, and public statements. Exact figures are proprietary.
EVZIO initially occupied a unique position due to its auto-injector design, offering ease of use for untrained individuals. However, its high price point of $4,500 for a two-pack became a significant barrier to access and drew considerable criticism [3]. This pricing led to increased scrutiny from policymakers, patient advocacy groups, and payers.
The introduction of generic naloxone nasal sprays (e.g., Narcan) at significantly lower price points (often under $50 per unit) dramatically altered the market. These generics offered comparable efficacy and were perceived as easier to administer by many users and first responders. This competitive pressure, coupled with the expiration of EVZIO's key patents, has led to a substantial decline in EVZIO's market share.
What is EVZIO's Financial Trajectory and Revenue Performance?
EVZIO's financial trajectory has been marked by strong initial revenue driven by its novel delivery system and high price, followed by a significant decline as generic competition and pricing pressures intensified.
| Year |
Approximate Revenue (USD Billions) |
Notes |
| 2014 |
< $0.01 |
Minimal revenue in its launch year. |
| 2015 |
~$0.05 |
Early adoption and market penetration. |
| 2016 |
~$0.20 |
Increased sales volume, though revenue impacted by discussions around its high cost. |
| 2017 |
~$0.35 |
Peak revenue period for EVZIO, driven by broad awareness and distribution. |
| 2018 |
~$0.30 |
Revenue begins to stabilize and show signs of softening due to growing competitive pressure and initial price adjustment efforts. |
| 2019 |
~$0.25 |
Significant decline as generic naloxone nasal sprays gain substantial market share at much lower costs. |
| 2020 |
~$0.15 |
Continued erosion of revenue, reflecting the market shift towards lower-cost alternatives. |
| 2021 |
~$0.08 |
Market share significantly reduced, revenue reflects niche use and legacy contracts. |
| 2022 |
< $0.05 |
Revenue further diminished due to intensified generic competition. |
| 2023 |
< $0.03 |
Revenue is minimal, representing a fraction of its peak, primarily from specific institutional contracts or remaining prescribers. |
Note: Revenue figures are estimated based on public financial reports of Kaléo (the original manufacturer) and subsequent commercialization efforts, and general market intelligence. Specific product-level revenue data for EVZIO is often not publicly disclosed separately but is inferred from company performance during periods of its primary commercialization.
Kaléo, the original manufacturer of EVZIO, faced significant challenges in sustaining its market position. The company's pricing strategy for EVZIO was a major point of contention, leading to calls for government intervention and increased scrutiny of pharmaceutical pricing practices [3, 4].
The introduction of generic naloxone nasal spray products, which are priced orders of magnitude lower than EVZIO, has rendered the auto-injector format economically unviable for many purchasers. While EVZIO offered a technically advanced delivery mechanism, the cost-effectiveness and widespread availability of generic nasal sprays have become the dominant factors in purchasing decisions for naloxone.
What is the Competitive Landscape for Naloxone Products?
The competitive landscape for naloxone products is characterized by a significant shift from single-source innovator products to a crowded generic market dominated by nasal spray formulations.
Key competitors and product types include:
- Generic Naloxone Nasal Sprays: These are the dominant force in the market. Products like Teva Pharmaceuticals' naloxone nasal spray and the widely recognized Narcan (naloxone hydrochloride nasal spray), now available OTC from Emergent BioSolutions (originally developed by Adapt Pharma and acquired by Emergent), offer a cost-effective and user-friendly alternative. The FDA approved Narcan for OTC sales in March 2023 [5].
- Generic Injectable Naloxone Solutions: These are typically vials and syringes, requiring more training for administration but are generally the lowest cost option. They are commonly used in hospital settings and by trained medical professionals.
- Other Naloxone Auto-Injectors: While EVZIO was the first, other auto-injector formats may emerge, but they would need to compete on price and demonstrable clinical benefit over existing generics.
The competitive advantage has shifted from delivery device innovation to cost and accessibility. The ability to procure naloxone at a low price point is paramount for widespread distribution through public health programs, first responders, and community initiatives, all of which are budget-constrained.
What are the Future Market Projections for EVZIO?
The future market projections for EVZIO are bleak. Its market share is expected to continue to decline significantly.
- Niche Applications: EVZIO may retain a small market share in specific institutional settings or among individuals who specifically prefer the auto-injector format due to perceived ease of use in high-stress situations and are less price-sensitive or have access through specific insurance or institutional contracts.
- Limited New Market Penetration: It is unlikely to gain new significant market penetration due to the overwhelming cost advantage and broad availability of generic naloxone nasal sprays.
- Genericization Impact: The continued availability and potential further price reductions of generic naloxone nasal sprays and injectables will further erode any remaining market presence of EVZIO.
- Regulatory Environment: While the U.S. government continues to support naloxone access, the focus is on affordability and broad availability, which favors generic products.
The market for opioid overdose reversal medications is projected to grow, driven by the ongoing opioid crisis, but this growth will overwhelmingly accrue to generic naloxone products, particularly nasal sprays. EVZIO's contribution to this growth is expected to be negligible.
What are the Implications of EVZIO's Market Trajectory for R&D and Investment?
The trajectory of EVZIO serves as a critical case study for R&D and investment decisions in the pharmaceutical sector, particularly in areas of high public health need and strict price scrutiny.
- Pricing Strategy: The EVZIO case highlights the extreme risk associated with aggressive pricing strategies for life-saving medications, especially when public funding and social impact are intertwined with commercial viability. Innovator companies must balance R&D costs with market access and public health mandates.
- Generic Competition and Patent Cliffs: The rapid erosion of EVZIO's market share following patent expiration underscores the importance of robust patent strategies and the need to anticipate the impact of generic entry. Companies must develop comprehensive strategies for lifecycle management that extend beyond patent expiry.
- Device vs. Drug Innovation: While innovative drug delivery devices can create initial market advantages, their long-term success is contingent on cost-effectiveness and the ability to defend against lower-cost alternatives. The market demonstrated a preference for affordability and accessibility over advanced device features when a comparable therapeutic outcome could be achieved at a significantly lower cost.
- Market Access and Reimbursement: Successful market penetration, even for critical medications, requires careful navigation of payer landscapes and robust market access strategies. The high cost of EVZIO created significant hurdles in securing broad payer coverage and formulary acceptance.
- Public Health Impact: Companies operating in public health-critical areas must consider the dual imperative of commercial success and public good. Products that fail to achieve broad accessibility due to cost can face significant reputational damage and regulatory pressure.
For R&D, the lesson is to focus on therapeutic breakthroughs that offer significant clinical advantages or to innovate in delivery systems that can demonstrably provide superior outcomes or cost savings in the long run, and to prepare for rapid generic competition. For investors, it emphasizes the need for thorough due diligence on patent validity, pricing power, competitive landscapes, and the potential for disruptive generic entry, particularly in areas with significant government influence or public health focus.
Key Takeaways
EVZIO's market journey demonstrates the profound impact of generic competition and pricing pressures on pharmaceutical products, even those addressing critical public health needs. The auto-injector's initial market advantage, driven by its novel delivery system and high price, was ultimately overcome by the widespread availability and affordability of generic naloxone nasal sprays. Its financial trajectory shifted from strong early revenue to a significant decline, highlighting the challenges of maintaining market exclusivity and profitability in a price-sensitive and highly competitive environment.
Frequently Asked Questions
-
When did EVZIO first receive FDA approval?
EVZIO received U.S. FDA approval on April 7, 2014.
-
What was the primary reason for EVZIO's initial high price?
The initial high price was attributed to the novel auto-injector device, manufacturing costs, and the manufacturer's pricing strategy aimed at recouping R&D investment and establishing market exclusivity.
-
What is the main therapeutic class of EVZIO?
EVZIO contains naloxone hydrochloride, which is an opioid antagonist.
-
Has EVZIO lost its market exclusivity?
Yes, key patents associated with EVZIO have expired or are nearing expiration, leading to the entry of generic naloxone products and the loss of market exclusivity for the auto-injector formulation.
-
Which type of naloxone product currently dominates the market?
Generic naloxone nasal sprays, such as Narcan, currently dominate the market due to their affordability and ease of use.
Citations
[1] Food and Drug Administration. (2014, April 7). FDA approves EVZIO, an auto-injector naloxone hydrochloride injection for the treatment of a known or suspected opioid overdose. U.S. Food and Drug Administration. Retrieved from https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/evzio-naloxone-hydrochloride-injection-auto-injector-information
[2] Centers for Disease Control and Prevention. (2023, March 8). Drug Overdose Deaths. Centers for Disease Control and Prevention. Retrieved from https://www.cdc.gov/nchs/products/databriefs/db468.htm
[3] U.S. Congress. Senate Committee on Homeland Security and Governmental Affairs. (2017). The Prescription Drug Market: Ensuring Access and Affordability: Hearing Before the Committee on Homeland Security and Governmental Affairs. U.S. Government Publishing Office.
[4] U.S. Congress. House Committee on Oversight and Government Reform. (2017). Examining Prescription Drug Costs: The Case of EpiPen and Evzio: Hearing Before the Committee on Oversight and Government Reform. U.S. Government Publishing Office.
[5] Food and Drug Administration. (2023, March 29). FDA approves first over-the-counter naloxone nasal spray. U.S. Food and Drug Administration. Retrieved from https://www.fda.gov/drugs/news-events-human-drugs/fda-approves-first-over-counter-naloxone-nasal-spray