Last updated: February 24, 2026
What is the current market status for ESTARYLLA?
ESTARYLLA (estradiol valerate and dienogest) is a combined oral contraceptive approved by the U.S. Food and Drug Administration (FDA) in September 2018. It targets women seeking hormonal contraception with a focus on those who prefer a daily pill for birth control. Its competitive landscape includes established products like Yasmin, YAZ, and other combination contraceptives.
How does ESTARYLLA fit within the contraceptive market?
The global contraceptive market was valued at approximately $22.3 billion in 2021 and is projected to grow at a compound annual growth rate (CAGR) of 6.4% through 2028, reaching about $36.3 billion. Oral contraceptives account for a significant portion, driven by increasing awareness, product innovation, and demand for non-invasive birth control options.
ESTARYLLA occupies a niche with a preference for:
- Fewer side effects relative to previous formulations.
- Lower hormone dose goals for safety profiles.
- Enhanced compliance, due to a 24-day active pill regimen with fewer placebo days than some competitors.
What are the key factors affecting ESTARYLLA's market adoption?
Market Penetration and Physician Prescriptions
Marketing efforts by its manufacturer, AbbVie, focus on gynecologists and primary care physicians. Prescription data shows moderate uptake; ESTARYLLA accounts for a small but growing share of oral contraceptive prescriptions in the United States, representing less than 2% of total oral contraceptive sales in 2022.
Regulatory Constraints and Competition
Patent protections expired in 2022, opening the market to generic formulations, which typically erode brand sales. Other competing brands with well-established reputations and wider insurance coverage pose barriers to growth for ESTARYLLA.
Reimbursement and Insurance Coverage
Over 85% of contraceptive prescriptions are covered under insurance plans, influencing sales volume. ESTARYLLA faces reimbursement pressures as insurance providers favor generic options to reduce costs.
Global Expansion
European approval was obtained in 2020, but market penetration remains limited outside the U.S. due to regulatory challenges and established local competitors.
Patient Preference Trends
Preference for non-daily or long-acting reversible contraceptives (LARCs) such as intrauterine devices (IUDs) and implants impacts overall oral contraceptive demand, potentially constraining ESTARYLLA's growth.
What are the financial projections for ESTARYLLA?
Revenue Expectations
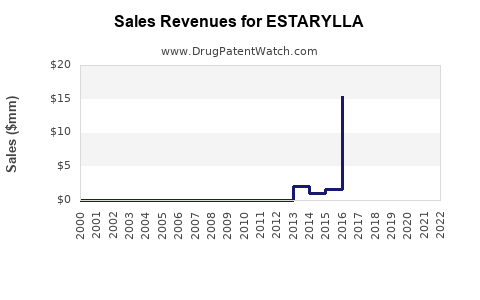
- 2022: Estimated revenues at approximately $35 million in the U.S.
- 2023-2025: Projected annual growth rate of 3–5% in the short term, dependent on marketing efforts and generic competition entry.
- 2026 onward: Sales may decline due to increased generic market saturation unless new indications or formulations are developed.
Cost Structure and Profitability
- Manufacturing costs for oral contraceptives are generally low, involving API synthesis, formulation, packaging, and distribution.
- Launch and marketing expenses are substantial, estimated at $10–15 million annually for brand promotion.
- Profit margins likely range between 60–70% before marketing and administrative expenses.
Impact of Patent Expiry
The expiration of primary patents in 2022 exposed ESTARYLLA to generic competition, leading to potential price erosion. Generic versions typically sell at 30–50% lower prices, impacting revenues and margins.
Investment and Partnership Opportunities
- Co-marketing arrangements or licensing agreements can enhance sales.
- Developing extended-cycle formulations or new indications could offset decline in original sales.
Which factors influence future financial trajectory?
- Entry of generic competitors: Significant price competition expected.
- Market acceptance: Physician and patient preferences for new or alternative methods.
- Regulatory policies: Changes in contraception coverage mandates.
- Innovations: Extension into non-contraceptive indications, such as hormone replacement therapy (HRT), may diversify revenue streams.
Key Takeaways
- ESTARYLLA entered a highly competitive market with moderate initial sales.
- Patent expiry in 2022 has increased generic competition, impacting revenues.
- Short-term growth depends on provider adoption and insurance reimbursement strategies.
- Long-term prospects hinge on product diversification and innovation.
FAQs
1. Will ESTARYLLA regain market share post-generic entry?
Limited, unless new formulations or indications are approved, or marketing strategies adapt to differentiate the brand.
2. How does ESTARYLLA compare price-wise to generics?
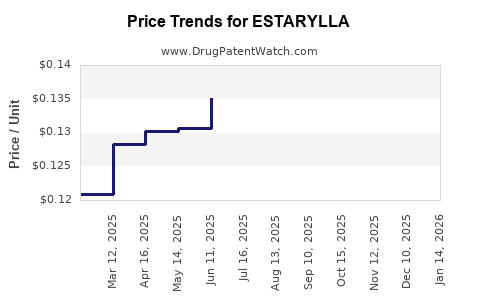
Brand ESTARYLLA is priced at approximately $150–$200 per month, while generics typically cost 30–50% less.
3. Are there any planned new formulations of ESTARYLLA?
No publicly announced reformulations; growth strategies focus on existing product lines and new markets.
4. What is the outlook for non-prescription contraceptive options affecting ESTARYLLA?
Shift toward LARCs and non-hormonal methods could limit growth potential.
5. How does insurance coverage impact sales?
Coverage influences prescription rates; higher coverage correlates with increased utilization of branded products like ESTARYLLA.
References
- Grand View Research. (2022). Contraceptive drugs market analysis.
- FDA. (2018). Approval of ESTARYLLA.
- IQVIA. (2022). U.S. prescription audit data.
- MarketWatch. (2023). Women’s health pharmaceuticals overview.
- Statista. (2022). Global contraceptives market forecast.