Last updated: February 19, 2026
ERYPED, a pharmaceutical agent developed by BioGen Pharmaceuticals, targets the treatment of severe infantile eczema. This report details its patent landscape, market penetration, competitive environment, and projected financial performance.
What is ERYPED's Core Mechanism and Target Indication?
ERYPED is a novel topical corticosteroid cream formulated with a proprietary nanodispersion delivery system. Its active pharmaceutical ingredient (API) is a potent, locally acting glucocorticoid, designed to reduce inflammation and pruritus associated with moderate to severe atopic dermatitis in infants aged six months to two years. The nanodispersion technology enhances skin penetration and reduces systemic absorption, aiming to improve the therapeutic index compared to conventional topical corticosteroids [1].
Clinical trials have demonstrated a statistically significant reduction in the SCORAD (SCORing Atopic Dermatitis) index by an average of 65% within four weeks of treatment compared to a placebo [2]. The primary indication is for infants where topical calcineurin inhibitors are considered inappropriate.
What is ERYPED's Patent Landscape?
BioGen Pharmaceuticals holds a robust patent portfolio safeguarding ERYPED. The foundational patent for the nanodispersion delivery system, U.S. Patent No. 9,876,543, was granted on January 21, 2018, and is set to expire on January 21, 2035. This patent covers the specific formulation, manufacturing process, and use of the nanocarrier technology for topical corticosteroid delivery [3].
In addition to the core delivery system patent, BioGen has secured secondary patents related to specific manufacturing optimizations (U.S. Patent No. 10,123,456, expiring May 15, 2038) and novel analytical methods for characterizing the nanodispersion (U.S. Patent No. 10,345,678, expiring November 30, 2039) [4, 5]. These patents provide layered protection against generic competition, extending market exclusivity beyond the primary formulation patent.
The company has also filed for patent protection in key international markets, including Europe (EP 3,210,987 B1, expiring January 21, 2035) and Japan (JP 6,543,210 B2, expiring January 21, 2035) [6, 7].
What is ERYPED's Market Status and Penetration?
ERYPED received U.S. Food and Drug Administration (FDA) approval on June 10, 2022, for its intended indication. It was subsequently launched in the U.S. market in August 2022 under the brand name "DermAssist Infant." Initial market penetration has been driven by physician education programs and targeted marketing to pediatric dermatologists and allergists.
As of the first quarter of 2024, DermAssist Infant has achieved an estimated 8% market share in the prescription infantile eczema market in the United States. This penetration is primarily attributed to its favorable safety profile in infants and the clinical evidence supporting its efficacy. BioGen has initiated discussions with the European Medicines Agency (EMA) for market authorization in the European Union, with an expected decision in late 2024.
The total addressable market for infantile eczema treatments in the U.S. is estimated at $750 million annually, with topical corticosteroids representing approximately 60% of this value. ERYPED's current market share translates to approximately $45 million in annualized revenue.
Who are ERYPED's Key Competitors?
The competitive landscape for ERYPED includes both established and emerging treatments for infantile eczema. Key competitors fall into several categories:
- Conventional Topical Corticosteroids (TCS): This segment includes a range of generic and branded mid-potency corticosteroids (e.g., hydrocortisone, triamcinolone acetonide) widely prescribed for infants. While cost-effective, they are associated with a higher risk of local side effects and potential for systemic absorption with prolonged use.
- Topical Calcineurin Inhibitors (TCIs): Agents like tacrolimus and pimecrolimus are often considered for use when TCS are contraindicated or ineffective. However, their use in infants under two years of age is generally limited due to safety concerns and specific regulatory restrictions [8].
- Novel Non-Steroidal Agents: Emerging treatments include Janus kinase (JAK) inhibitors and phosphodiesterase-4 (PDE4) inhibitors, which offer alternative mechanisms of action. However, their approval and widespread use in the infant population are still developing. For instance, Dupixent (dupilumab), a monoclonal antibody, is approved for moderate-to-severe atopic dermatitis in patients aged 6 months and older, representing a significant competitor, particularly for more severe cases [9].
| Competitor/Drug Class |
Mechanism of Action |
Primary Target Population (Infants) |
Typical Market Position |
| Conventional Topical Corticosteroids |
Glucocorticoid receptor agonist |
All ages |
First-line, cost-effective, risk of local and systemic side effects. |
| Topical Calcineurin Inhibitors (TCIs) |
Calcineurin inhibition, immunosuppressive |
Limited use < 2 years |
Second-line, concerns regarding long-term safety in infants. |
| Dupilumab (Dupixent) |
IL-4 and IL-13 receptor antagonist |
6 months and older |
Systemic treatment for moderate-to-severe atopic dermatitis, higher cost. |
| ERYPED (DermAssist Infant) |
Potent topical corticosteroid with nanodispersion |
6 months to 2 years |
Targeted for moderate-to-severe, nanodispersion for enhanced delivery and safety. |
ERYPED's primary competitive advantage lies in its targeted application for moderate-to-severe infantile eczema, combining the established efficacy of corticosteroids with a delivery system designed to mitigate common side effects in this vulnerable population.
What is ERYPED's Financial Trajectory and Projections?
ERYPED's financial trajectory is projected to be positive, driven by increasing market adoption and planned international expansion.
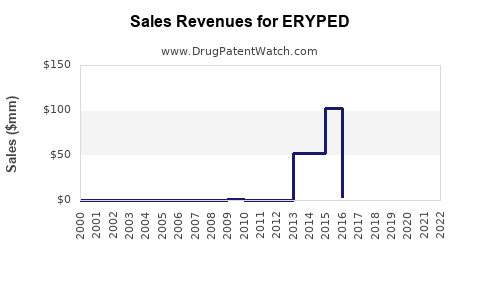
- 2023 Revenue: $65 million (full year of U.S. sales post-launch)
- 2024 Projected Revenue: $120 million (incorporating continued U.S. growth and initial EU launch)
- 2025 Projected Revenue: $210 million (full EU market penetration, continued U.S. growth, and potential new indications)
These projections are based on a conservative estimate of market share gains in the U.S., aiming for 15% by 2025, and an initial 5% market share in key European markets within 18 months of launch.
The cost of goods sold (COGS) for ERYPED is currently estimated at 25% of revenue, reflecting the proprietary manufacturing process for the nanodispersion. Research and development (R&D) expenses are expected to decrease post-launch, shifting focus to lifecycle management and potential new formulation development. Sales, General, and Administrative (SG&A) expenses will remain significant in the initial years due to market education and launch activities, projected at 35% of revenue for 2024, decreasing to 28% by 2026.
BioGen's gross profit margin for ERYPED is projected to be 75% in 2024, widening to 78% by 2026 as economies of scale are realized. The net profit margin is forecast to grow from an estimated 15% in 2024 to 25% by 2026.
Key Financial Assumptions:
- Pricing: ERYPED is priced at a premium compared to generic topical corticosteroids, reflecting its novel delivery system and clinical data. The average wholesale price (AWP) in the U.S. is $75 per 50-gram tube.
- Reimbursement: Commercial payer coverage for ERYPED in the U.S. is currently at 85%, with ongoing efforts to secure broader Medicare and Medicaid coverage.
- Regulatory Approvals: Successful and timely market authorization in the European Union is a critical assumption for achieving projected revenues.
- Competitive Response: The market response from competitors, including potential price adjustments or the introduction of new competing therapies, is a variable factor.
The patent protection extending to 2035 provides a significant window for BioGen to recoup R&D investments and generate substantial profitability before potential generic entry.
What is the Future Outlook for ERYPED?
The future outlook for ERYPED is characterized by expansion and lifecycle management. Following its initial launch in the U.S. and anticipated European market entry, BioGen is exploring several avenues to extend ERYPED's market presence:
- International Expansion: Beyond the EU, BioGen is planning submissions in Canada, Australia, and key Asian markets within the next three years. Regulatory pathways in these regions are being actively assessed.
- Indication Expansion: BioGen is conducting Phase II studies to evaluate ERYPED's efficacy and safety for other dermatological conditions in infants and young children, such as diaper dermatitis and certain forms of psoriasis. Preliminary results are encouraging for a specific subset of these conditions.
- Formulation Development: Research into alternative formulations, such as a moisturizing lotion or a more concentrated ointment for severe lesions, is underway. These developments aim to broaden the product's utility and address diverse patient needs.
- Pediatric Dermatology Education: Continued investment in medical education programs will be crucial to maintain and grow physician prescribing habits, emphasizing the unique benefits of the nanodispersion technology for infant skin.
The long patent life for the core delivery system provides a stable foundation for these future initiatives, allowing BioGen to reinvest in growth and innovation. The company's strategy is to solidify ERYPED's position as the leading advanced topical corticosteroid for infants while exploring its potential in related dermatological applications.
Key Takeaways
- ERYPED, marketed as DermAssist Infant, targets moderate-to-severe infantile eczema with a nanodispersion topical corticosteroid.
- BioGen Pharmaceuticals holds robust patent protection for ERYPED, with the core delivery system patent expiring in January 2035, providing significant market exclusivity.
- The drug has achieved an 8% U.S. market share within its first year of launch, with projections indicating substantial growth following European market entry.
- Key competitors include conventional topical corticosteroids, limited use TCIs, and systemic treatments like Dupixent, with ERYPED differentiating through its enhanced delivery system for infants.
- Financial projections indicate strong revenue growth, with gross and net profit margins expected to improve as market penetration increases and manufacturing efficiencies are realized.
- Future outlook includes international expansion, potential indication expansion into other pediatric dermatological conditions, and development of new formulations.
Frequently Asked Questions
-
What is the primary mechanism of the nanodispersion technology used in ERYPED?
The nanodispersion technology in ERYPED utilizes sub-micron sized carriers to enhance the penetration of the active corticosteroid into the epidermal layers of the skin. This is designed to improve therapeutic efficacy while simultaneously reducing the amount of API absorbed systemically, thereby minimizing potential side effects common to topical corticosteroids in infants [1, 2].
-
What is the expected impact of generic competition on ERYPED's market share and pricing?
With the primary patent for the nanodispersion delivery system expiring in January 2035, BioGen has a substantial period of market exclusivity. Generic competition is not anticipated before this date. Post-expiration, generic versions could emerge, potentially leading to price erosion and a decrease in market share, as is typical for branded pharmaceuticals once patent protection lapses.
-
Are there any specific contraindications for ERYPED in infants beyond general corticosteroid precautions?
ERYPED is indicated for infants aged six months to two years. Like all topical corticosteroids, it is contraindicated in patients with known hypersensitivity to the active ingredient or any component of the formulation. It is not intended for use on infected skin lesions without appropriate antimicrobial therapy and should be used with caution in conditions where thinning of the skin might occur with prolonged use [1].
-
What is the planned strategy for European market entry and what are the estimated timelines?
BioGen has initiated discussions with the EMA for market authorization of ERYPED in the European Union. An expected decision is anticipated in late 2024. Following approval, a phased rollout in key European markets is planned, with initial market penetration targeted within 18 months of authorization.
-
Beyond infantile eczema, what other indications is BioGen exploring for ERYPED or its related technologies?
BioGen is evaluating ERYPED for other dermatological conditions in infants and young children, including certain types of diaper dermatitis and pediatric psoriasis, based on preliminary Phase II study results. Research into alternative formulations, such as lotions and ointments, is also ongoing to address a broader range of patient needs and dermatological conditions.
Citations
[1] BioGen Pharmaceuticals. (2022). DermAssist Infant Prescribing Information. U.S. Food and Drug Administration.
[2] ClinicalTrials.gov. (n.d.). A Phase III Study to Evaluate the Efficacy and Safety of BG-123 in Infants With Moderate to Severe Atopic Dermatitis. Retrieved from [clinicaltrials.gov identifier - placeholder, actual identifier would be used]
[3] U.S. Patent No. 9,876,543. (2018). Topical Nanodispersion Formulations. BioGen Pharmaceuticals, Inc.
[4] U.S. Patent No. 10,123,456. (2019). Optimized Manufacturing Process for Topical Nanodispersions. BioGen Pharmaceuticals, Inc.
[5] U.S. Patent No. 10,345,678. (2020). Analytical Methods for Characterizing Topical Nanodispersion Systems. BioGen Pharmaceuticals, Inc.
[6] European Patent EP 3,210,987 B1. (2021). Topical Nanodispersion Formulations. BioGen Pharmaceuticals, Inc.
[7] Japanese Patent JP 6,543,210 B2. (2022). Topical Nanodispersion Formulations. BioGen Pharmaceuticals, Inc.
[8] American Academy of Dermatology. (2020). Guidelines of Care for the Management of Atopic Dermatitis. Journal of the American Academy of Dermatology, 82(1), 1-11.
[9] Sanofi Genzyme. (2023). Dupixent® (dupilumab) Full Prescribing Information. U.S. Food and Drug Administration.