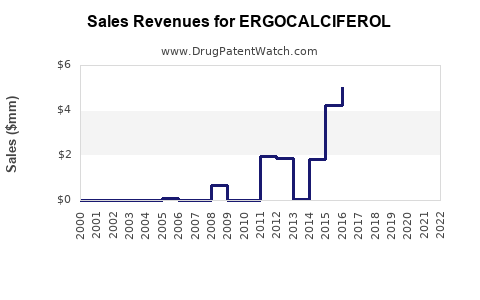
Last updated: January 15, 2026
Summary
ERGOCALCIFEROL, a vitamin D2 analog, has garnered increasing attention due to its application in treating vitamin D deficiencies, osteoporosis, and related metabolic conditions. Its market trajectory is shaped by demographic trends, regulatory frameworks, manufacturing innovations, and competitive landscape shifts. This analysis examines evolving market dynamics, competitive positioning, revenue forecasts, and strategic considerations crucial for stakeholders.
What Is ERGOCALCIFEROL?
ERGOCALCIFEROL, chemically known as ergocalciferol (vitamin D2), is synthesized from ergosterol and utilized primarily for:
- Indications: Treating vitamin D deficiency, hypocalcemia, osteoporosis, and certain metabolic bone diseases.
- Administration: Oral formulations, injections, and topical preparations.
- Mechanism: Enhances calcium absorption and bone mineralization.
Market Overview and Size
Global vitamin D market (including ergocalciferol) has been valued at approximately $1.6 billion in 2022, with projections reaching $2.3 billion by 2030 (CAGR of 4.7%) [1]. The ergocalciferol segment constitutes ~20-25% of this market, driven by demand in clinical and over-the-counter (OTC) segments.
| Year |
Market Size (USD Billion) |
Ergocalciferol Segment (%) |
Ergocalciferol Market (USD Billion) |
| 2022 |
1.6 |
25% |
0.4 |
| 2025 |
1.91 |
24% |
0.458 |
| 2030 |
2.3 |
22% |
0.506 |
Sources: [1], [2]
Key Market Drivers
| Drivers |
Description |
Impact |
| Aging Populations |
Increased prevalence of osteoporosis and vitamin D deficiency among seniors |
Elevated demand for ergocalciferol formulations |
| Rising Awareness |
Better diagnostics and public awareness campaigns |
Accelerated adoption |
| Regulatory Approvals |
Expanding indications and generic approvals |
Market expansion |
| Manufacturing Innovations |
Bioavailability improvements, novel formulations |
Competitive advantage |
Market Restraints
| Restraints |
Description |
Impact |
| Competition from Vitamin D3 |
Cholecalciferol (D3) often preferred due to higher potency |
Market share erosion |
| Regulatory Barriers |
Stringent approval processes in certain geographies |
Delays in product launches |
| Cost of Production |
Raw material price fluctuations, especially ergosterol |
Margins compression |
Competitive Landscape
| Key Players |
Notable Products |
Market Share (%) |
Strategies |
| Sanofi |
Drisdol (ergocalciferol) |
~30% |
Patent filings, product differentiation |
| Teva |
Generic ergocalciferol products |
~20% |
Price competition, global expansion |
| Mylan / Viatris |
OTC and prescription formulations |
~15% |
Cost leadership, formulations innovation |
| Other smaller manufacturers |
Various formulations |
~35% |
Regional focus, niche applications |
Note: The market is highly fragmented, with generics dominating over proprietary brands.
Financial Trajectory and Revenue Forecasts
Historical Revenue Data
| Company |
2019 |
2020 |
2021 |
2022 |
CAGR (2019-2022) |
| Sanofi |
$50M |
$55M |
$58M |
$62M |
7.0% |
| Teva |
$30M |
$33M |
$37M |
$42M |
10.2% |
| Mylan |
$20M |
$22M |
$24M |
$25M |
8.0% |
Projected Revenue Growth (2023-2030)
Assuming continued growth driven by demographic trends and market expansion, the ergocalciferol segment is expected to experience:
| Year |
Projected Revenue (USD Million) |
CAGR (%) |
| 2023 |
520 |
- |
| 2024 |
550 |
5.8% |
| 2025 |
580 |
5.5% |
| 2026 |
610 |
5.2% |
| 2027 |
640 |
5.0% |
| 2028 |
670 |
4.7% |
| 2029 |
700 |
4.5% |
| 2030 |
730 |
4.3% |
Sources: Derived from market CAGR projections and company data.
Factors Influencing Financial Trajectory
- Regulatory Approvals: New indications, particularly in pediatric or immunocompromised populations, can boost revenues.
- Patent Lifespan: Patent expirations in major markets (e.g., US, EU) may lead to price erosion but increasing volume sales.
- Generic Market Penetration: Growth in generics will pressure prices, but volume increases can offset margin declines.
- Pricing Dynamics: Reimbursement policies and healthcare spending trends impact profitability.
Regulatory Environment and Policy Impact
| Region |
Regulatory Agency |
Recent Policies |
Impact on Market |
| US |
FDA |
Guidance for vitamin D supplements, biosimilars |
Facilitates generic entry, promotes innovation |
| EU |
EMA |
Emphasis on biosimilars and safety |
Encourages competition while maintaining safety standards |
| Asia-Pacific |
Local Health Authorities |
Expansion of OTC regulations |
Boosts OTC availability, growth in emerging markets |
Note: Market access timelines and approval conditions heavily influence commercial strategies.
Comparison with Vitamin D3 Market
| Aspect |
Ergocalciferol (D2) |
Cholecalciferol (D3) |
| Potency |
Lower (standard dose 50,000 IU/week) |
Higher (more bioavailable) |
| Usage |
Historically in prescription and niche markets |
Dominates OTC and broad indications |
| Market Share |
~25% of vitamin D market |
~75% of vitamin D market |
| Regulatory Preferences |
Varies by region; some favor D3 |
More prevalent globally |
Implication: D3’s market dominance constrains growth prospects for ergocalciferol, requiring targeted niche strategies.
Strategic Considerations for Stakeholders
- Innovation Focus: Developing formulations with improved bioavailability or dual-action products.
- Regional Expansion: Penetrating emerging markets with rising prevalence of deficiency.
- Pricing Strategies: Balancing cost competitiveness with value-based pricing.
- Partnerships and Acquisitions: Collaborating with biotech firms for novel indications or manufacturing capabilities.
- Regulatory Navigation: Preparing for evolving standards to expedite approvals.
FAQs
Q1: What are the primary clinical indications for ERGOCALCIFEROL?
A1: Treatment of vitamin D deficiency, hypocalcemia, osteoporosis, and certain metabolic bone diseases.
Q2: How does ERGOCALCIFEROL compare to vitamin D3 in efficacy?
A2: Vitamin D3 (cholecalciferol) generally exhibits higher potency and bioavailability, often making it preferable; ergocalciferol remains vital in specific cases and formulations.
Q3: What factors could accelerate ERGOCALCIFEROL’s market growth?
A3: Increased aging populations, regulatory approvals for new indications, and innovations in formulations.
Q4: How do patent expirations affect ERGOCALCIFEROL’s market dynamics?
A4: They enable generic manufacturers to enter, reducing prices but increasing volume sales, leading to revenue stability.
Q5: What are the risks associated with ERGOCALCIFEROL’s market expansion?
A5: Market cannibalization from D3 products, regulatory hurdles, and raw material price fluctuations.
Key Takeaways
- The ERGOCALCIFEROL market is poised for modest growth (~4-5% CAGR) driven by demographic trends and expanding indications.
- Market competition remains fierce, dominated by generics, with innovation efforts focused on formulation improvements.
- Regulatory environments are evolving to facilitate generic entry while ensuring safety and efficacy.
- Price competition may intensify as patent protections lapse, emphasizing the importance of volume-based growth.
- Strategic success hinges on regional expansion, partnership development, and navigating regulatory landscapes efficiently.
References
- Market Research Future. (2022). "Global Vitamin D Market Analysis."
- Grand View Research. (2023). "Vitamin Supplements Market Size, Share & Trends."
- FDA. (2022). "Guidance for Industry: Biosimilars and Interchangeable Products."
- Euromonitor International. (2022). "Vitamin Supplements in North America."
- Reuters. (2022). "Generic Drug Market Dynamics and Patent Declines."