Last updated: April 25, 2026
CYTOMEL (liothyronine sodium) is a mature, generic-access product with limited brand-level upside outside the U.S. label scope and routine thyroid-therapy demand. The financial trajectory is driven by (1) steady prescription volume tied to hypothyroidism and thyroid-suppression use, (2) competitive generic pricing, (3) payer formulary placement for liothyronine products, and (4) episodic reimbursement and supply dynamics affecting endocrine therapy switching.
What drives CYTOMEL demand in current markets?
1) Clinical use is stable and concentrated
CYTOMEL is used for hypothyroidism (including cases requiring T3) and for thyroid suppression. Demand is therefore tethered to incidence and long-term management of thyroid disorders rather than episodic seasonal demand.
Primary therapeutic value drivers
- Chronic use patterns in hypothyroidism management and replacement adjustments
- Use in thyroid suppression regimens where T3 is clinically selected
- Long-lived patient cohorts that keep baseline demand anchored
2) Brand value is structurally capped by generics
Liothyronine sodium is widely genericized in the U.S. and many markets. That compresses pricing power and pushes financial outcomes toward volume rather than net price.
Typical market effect on CYTOMEL
- Less pricing latitude versus patented, high-differentiation drugs
- Higher share sensitivity to pharmacy benefit manager (PBM) contracting and interchangeability
- Faster market penetration by lowest acquisition cost (LAC) competitors
3) Payer behavior shifts from coverage to cost-control
For mature thyroid therapies, payers tend to optimize around:
- Formulary tier placement (preferred vs non-preferred)
- Step edits or quantity limits in some cases
- PBM contracting that can swing net revenue based on discounts and rebates
Net result
CYTOMEL’s financial trajectory tracks reimbursement mechanics more than clinical differentiation.
4) Competitive interchange is a recurring headwind
When multiple liothyronine products are therapeutically equivalent, switches occur based on price and availability. That increases revenue volatility even when overall disease prevalence remains stable.
How do competitors shape CYTOMEL’s pricing and share profile?
Competition set
- Generic liothyronine sodium products (primary competitive pressure)
- Multi-product thyroid portfolios among endocrine brands and generics (secondary influence via therapy choice, not direct chemical competition)
Pricing dynamics
Generic competition forces:
- Net price compression across the class
- Higher dependence on contracting terms (rebates/discounts) to defend share
- Lower probability of sustained premium pricing for the brand
Market behavior
- If generic prices fall further, CYTOMEL typically faces incremental share leakage.
- If supply disruptions occur for certain generic lots, brand share can temporarily stabilize, but this rarely changes the structural economics.
What is the financial trajectory profile for CYTOMEL?
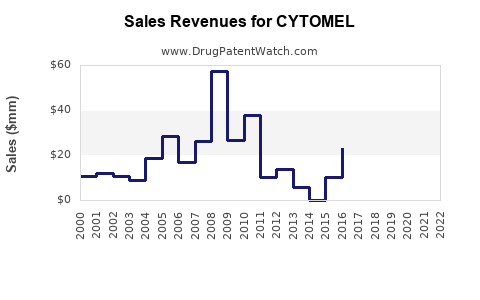
1) Mature product economics: volume-led, price-capped
CYTOMEL behaves like a classic mature commodity specialty:
- Revenue follows prescription demand and persistence in treated populations
- Gross-to-net margins reflect payer rebates and contract terms
- Operating performance is constrained by generic pricing pressure
2) Likely revenue sensitivity points
CYTOMEL’s revenue is most sensitive to:
- PBM formulary actions (preferred status changes)
- Wholesale acquisition cost (WAC) and discounting dynamics among generics
- Supply availability and substitution patterns at the pharmacy level
- Product mix: shifts between T4-heavy management versus T3-specified therapy choices
3) Financial trajectory: what investors typically see with this profile
For mature, genericized therapies, the observable pattern is usually:
- Limited growth after baseline demand saturation
- Intermittent declines as generics deepen cost advantage
- Periodic stabilization when payer contracting or supply issues temporarily reduce substitution speed
What does the U.S. regulatory label imply for market growth limits?
CYTOMEL is governed by established dosing and safety labeling for liothyronine use. In practice, that means clinical adoption is less about expansion into new indications and more about maintenance of existing prescribing channels.
Regulatory and clinical constraints that matter commercially
- Indications are established thyroid-management categories rather than new treatment paradigms
- Prescribing patterns are influenced by guideline-driven thyroid care pathways and clinician familiarity
- Label-level innovation is unlikely to create step-change demand in a generic-heavy category
How does competitive switching affect pharmacy and channel economics?
Retail and channel mechanics
Pharmacies and PBMs optimize for:
- Net acquisition cost and contract terms
- Availability and fulfillment reliability
- Claims adjudication outcomes tied to formulary status
Switching effects
- When a lower-cost generic gains preferred status, share migration accelerates.
- When a competitor has supply issues, CYTOMEL share can rise temporarily, but reverses when supply normalizes.
Consequence for financial trajectory
Revenue can show uneven month-to-month results even if epidemiology-driven demand remains stable.
What market-level indicators should track CYTOMEL’s trajectory going forward?
1) Prescription and claim volume
Track:
- Filled prescriptions for liothyronine sodium across key channels
- Claims counts by setting (retail vs specialty channels if any relevant shifts occur)
- Persistence metrics where available (patient-level continuity)
2) Net price and gross-to-net margin
Track:
- Net revenue per unit changes versus WAC
- Rebate and discount rate evolution under PBM contracting changes
- Channel inventory dynamics and returns exposure
3) Formulary and contracting actions
Track:
- PBM policy changes impacting formulary tiering
- Step edit or utilization management changes by major payers
- State Medicaid formulary updates that can shift pharmacy selection
4) Generic competition landscape
Track:
- New entrants and price cuts among generic liothyronine suppliers
- Product availability and lot-level supply issues
- Contracting changes that affect pharmacy substitution speed
Key Takeaways
- CYTOMEL’s market is stable-demand, but brand economics are constrained by generic liothyronine competition.
- The product’s financial trajectory is volume-led with net price pressure driven by PBM contracting and generic LAC dynamics.
- Near-term revenue path is most sensitive to formulary tier placement, rebate/discount terms, and substitution speed, not to clinical expansion.
- Expect muted growth and periodic volatility from channel and supply factors rather than sustained premium pricing.
FAQs
-
Is CYTOMEL growth primarily driven by new patient adoption?
No. Demand is anchored to established thyroid-treatment cohorts; growth depends more on prescribing persistence and formulary access than new clinical adoption.
-
What most directly compresses CYTOMEL net revenue?
Generic liothyronine pricing and PBM contracting that shifts purchasing toward lowest-cost equivalents.
-
Can CYTOMEL rebound during generic supply constraints?
It can temporarily stabilize share when alternative lots are unavailable, but that typically reverses when supply normalizes.
-
What payer levers matter most for CYTOMEL?
Formulary tier placement, rebate structures, and any utilization management actions that affect substitution and prescribing.
-
Where is CYTOMEL’s upside most limited?
In markets where liothyronine sodium is generic-dense and where clinician choice does not expand beyond established thyroid use cases.
References
[1] U.S. Food and Drug Administration. (n.d.). CYTOMEL (liothyronine sodium) prescribing information. FDA. https://www.accessdata.fda.gov/
[2] DailyMed. (n.d.). CYTOMEL (liothyronine sodium) tablet, for oral use. U.S. National Library of Medicine. https://dailymed.nlm.nih.gov/
[3] National Library of Medicine. (n.d.). Liothyronine sodium (T3) drug information and references. MedlinePlus. https://medlineplus.gov/