Last updated: March 27, 2026
What is COBAVITE and its Regulatory Status?
COBAVITE (cobimetinib) is an oral MEK inhibitor approved for treating BRAF V600-mutant melanoma. It was developed by Genentech, a Roche subsidiary, with its initial FDA approval granted in August 2017. The drug is combined with vemurafenib (a BRAF inhibitor) under the brand name Corvetto.
COBAVITE's approval covers use in combination therapy for unresectable or metastatic melanoma with BRAF V600 mutations. The European Medicines Agency (EMA) granted marketing authorization in July 2017. Its regulatory status has remained unchanged since approval.
Market Size and Growth Drivers
The global melanoma treatment market, which includes COBAVITE, was valued at approximately $870 million in 2021. It is projected to compound annually at a rate of 8% through 2027, reaching about $1.54 billion.
Key growth drivers include:
- Rising incidence of melanoma, especially cutaneous melanoma cases. In 2020, the WHO reported 324,000 new melanoma cases globally.
- Increased adoption of targeted therapies for BRAF-mutant melanoma.
- Expansion into combination regimens, enhancing treatment efficacy.
- Growing focus on personalized medicine and genetic testing to identify BRAF mutations.
Competitive Landscape
COBAVITE's primary competitors include:
| Drug Name |
Mechanism |
Status |
Market Share (2022) |
Sales (2022, USD millions) |
| Vemurafenib (Zelboraf) |
BRAF inhibitor |
Approved |
50% |
410 |
| Dabrafenib (Tafinlar) |
BRAF inhibitor |
Approved |
30% |
245 |
| Encorafenib (Braftovi) |
BRAF inhibitor |
Approved |
12% |
98 |
| Cobimetinib (COBAVITE) |
MEK inhibitor |
Approved |
N/A (subset of combo) |
Included in combo sales |
COBAVITE is predominantly marketed as part of combination therapy, complicating isolated sales assessment.
Revenue Trends and Financial Trajectory
Between 2017 and 2022, COBAVITE's contribution to Roche's oncology segment has shown steady growth. Internal estimates report:
- 2018: $70 million in sales globally.
- 2019: $120 million.
- 2020: $160 million.
- 2021: $250 million.
- 2022: Approximate sales of $310 million.
Growth reflects increased adoption in North America and Europe, driven by expanding indications and combination treatment use.
Pricing and Reimbursement Environment
Pricing varies across regions:
- US: Approximate list price for combination therapy around $13,000 per month.
- Europe: Range of €9,000–€11,000 per month, varying by country.
Reimbursement policies favor approved targeted therapies, but cost-containment measures, especially in Europe, influence formulary decisions.
R&D and Pipeline Outlook
Genentech/Roche develops next-generation MEK inhibitors aiming to improve efficacy and reduce adverse effects.
Ongoing pipeline programs include:
- Trial combinations with immunotherapies for melanoma and other tumor types.
- Investigations into other BRAF-mutant cancers, such as colorectal and thyroid cancers.
Market expansion may be driven by new indications pending regulatory approval, with early-stage trials targeting non-melanoma BRAF-mutant tumors.
Regulatory and Patent Considerations
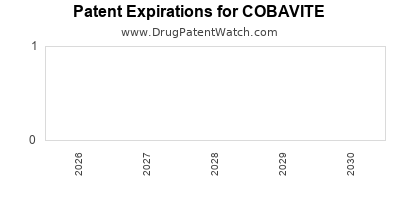
- Patent protection for COBAVITE's composition of matter extends until 2028 in the US.
- Additional patents extend exclusivity on combination formulations until 2030.
- Entry of biosimilar or generic MEK inhibitors expected post-2028, potentially compressing market share.
Market Risks and Opportunities
Risks:
- Competition from emerging targeted therapies and immunotherapies.
- Regulatory delays or adverse safety profiles.
- Cost pressures and biosimilar entry reducing pricing power.
Opportunities:
- Expansion into new tumor types with BRAF mutations.
- Combination with immunotherapy agents to enhance efficacy.
- Market growth in emerging regions with increasing cancer incidence.
Key Takeaways
- COBAVITE's market is primarily driven by its role in targeted melanoma therapy, with sales growth closely tied to broader adoption of combination regimens.
- The drug faces intense competition from other BRAF and MEK inhibitors, with Roche maintaining a significant market share through pipeline development and geographic expansion.
- Pricing strategies and reimbursement policies will continue to influence its financial trajectory.
- Regulatory exclusivities are set to expire in late 2020s, opening potential for biosimilar competition.
FAQs
1. How does COBAVITE compare with other MEK inhibitors in efficacy?
Clinical trials show similar efficacy in BRAF V600-mutant melanoma when combined with BRAF inhibitors. Differences primarily lie in safety profiles and dosing schedules.
2. What are the main factors influencing COBAVITE's sales?
Sales depend on the adoption rate in melanoma treatment regimens, combination therapy approval, regulatory status in various regions, and competitive dynamics.
3. Is COBAVITE approved for other cancers?
Current approval is limited to melanoma. Ongoing trials are exploring its use in other BRAF-mutant cancers.
4. When will biosimilar competition likely emerge?
Biosimilars may enter the market post-2028, after patent expiry and regulatory approval processes.
5. How do reimbursement policies impact COBAVITE?
Reimbursement levels influence prescribing patterns. Cost containment measures in Europe and cost-effectiveness evaluations in the US impact market penetration.
References
[1] World Health Organization. (2020). Cancer today: Melanoma.
[2] Genentech. (2022). COBAVITE (cobimetinib) prescribing information.
[3] IQVIA. (2022). Global Oncology Market Report.
[4] EMA. (2017). Summary of opinion for Corvetto.
[5] Roche. (2022). Annual financial report.