Last updated: February 19, 2026
Claritin-D, a combination non-drowsy antihistamine and nasal decongestant, has maintained a significant presence in the over-the-counter (OTC) allergy and sinus relief market. Its market position is shaped by patent expirations, generic competition, and evolving consumer preferences for allergy management.
What is the Active Pharmaceutical Ingredient (API) Composition of Claritin-D?
Claritin-D combines loratadine, a second-generation H1 antihistamine, with pseudoephedrine, a sympathomimetic amine acting as a nasal decongestant.
- Loratadine: Blocks the action of histamine, a substance in the body that causes allergic symptoms. It is known for its non-sedating properties.
- Pseudoephedrine: Constricts blood vessels in the nasal passages, reducing swelling and congestion.
This dual-action formulation targets multiple allergy symptoms, including sneezing, runny nose, itchy throat, and nasal congestion.
What is the Patent and Exclusivity History of Claritin-D?
The patent and exclusivity landscape for Claritin-D has been instrumental in its market trajectory.
- Loratadine (Claritin) Patents: The original patents for loratadine, the active ingredient in Claritin, expired in the early 2000s. Key patents included U.S. Patent No. 4,282,231, which covered loratadine.
- Combination Product Patents: Patents specifically covering the combination of loratadine and pseudoephedrine (as found in Claritin-D) also expired. These patents aimed to protect the formulation and its specific therapeutic benefit.
- Regulatory Exclusivity: Following patent expiration, generic manufacturers sought and obtained U.S. Food and Drug Administration (FDA) approval for their own versions of loratadine and the loratadine/pseudoephedrine combination.
- Controlled Release Formulations: Extended-release formulations of loratadine and pseudoephedrine, often marketed as Claritin-D 12-Hour and Claritin-D 24-Hour, had their own patent protection periods. These patents typically focused on the drug delivery system to ensure sustained release of the APIs. For example, patents related to extended-release formulations often describe specific matrix systems or coating technologies designed to control the rate of drug dissolution.
The expiration of these patents paved the way for significant generic competition, impacting the pricing and market share of branded Claritin-D.
Who are the Key Manufacturers and Competitors in the Claritin-D Market?
The market for Claritin-D is characterized by the presence of the originator brand and a robust generic sector.
- Originator Brand: Bayer HealthCare (and previously Schering-Plough) markets the branded Claritin-D.
- Generic Manufacturers: Numerous pharmaceutical companies produce generic versions of loratadine and pseudoephedrine combination products. These include, but are not limited to:
- Teva Pharmaceuticals
- Sandoz (a Novartis company)
- Apotex
- Wockhardt
- Various other companies holding Abbreviated New Drug Applications (ANDAs) for the product.
- Direct Competitors (Other OTC Allergy Medications):
- Single-Ingredient Antihistamines: Zyrtec (cetirizine HCl), Allegra (fexofenadine HCl) – both also have branded and generic versions.
- Other Combination Products: Products containing cetirizine/pseudoephedrine or fexofenadine/pseudoephedrine.
- Nasal Steroids: Flonase (fluticasone propionate), Nasacort (triamcinolone acetonide) – these are also available OTC and compete for the same patient population, offering a different mechanism of action.
- Oral Decongestants: Sudafed (phenylephrine HCl, though pseudoephedrine is often preferred for efficacy and is sold behind the counter in many regions).
The competitive landscape is intense, driven by price sensitivity and brand loyalty.
What is the Regulatory Status of Pseudoephedrine?
The regulatory status of pseudoephedrine is a critical factor influencing the availability and sale of products like Claritin-D.
- Combat Methamphetamine Epidemic Act of 2005 (CMEA): This U.S. federal law mandates that pseudoephedrine and products containing it be stored behind the counter of pharmacies.
- Purchase Limits: CMEA also imposes limits on the quantity of pseudoephedrine that can be purchased by an individual within a specified period.
- Daily limit: 3.6 grams
- 30-day limit: 7.5 grams
- Record Keeping: Retailers are required to maintain logs of pseudoephedrine sales, including customer identification and dates of purchase.
- State-Level Regulations: Some states have enacted additional restrictions, such as requiring a prescription for all pseudoephedrine-containing products, further impacting market access.
- International Regulations: Regulations concerning pseudoephedrine vary significantly by country due to its potential for illicit methamphetamine production.
These regulations affect consumer access and can shift market preference towards alternative decongestant ingredients or single-ingredient antihistamines not subject to these controls.
What are the Sales and Market Share Trends for Claritin-D?
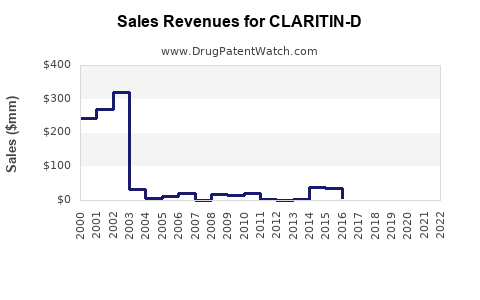
The sales and market share trajectory of branded Claritin-D has been significantly impacted by genericization.
- Pre-Generic Era: Branded Claritin-D commanded premium pricing and a substantial market share in the allergy and sinus relief category.
- Post-Generic Entry: Following the expiration of key patents and the introduction of generic loratadine/pseudoephedrine products, the market share for branded Claritin-D has declined.
- Generic loratadine products are typically priced at a significant discount compared to the branded equivalent.
- This price differential drives consumer choice towards generics, particularly for price-sensitive consumers.
- Market Size Data: While specific, real-time sales figures for branded Claritin-D are proprietary and fluctuate, the overall OTC allergy and cold market is a multi-billion dollar industry. Within this, combination allergy products represent a significant segment. Data from market research firms often shows that while branded products maintain some loyalty, the volume share in the allergy segment is heavily influenced by generics, especially for older, off-patent molecules like loratadine.
- Recent Trends:
- The market continues to see pressure from newer, branded antihistamines (e.g., Allegra, Zyrtec) and their generic equivalents, as well as the growing popularity of nasal steroid sprays.
- Claritin-D's market share is sustained by its established brand recognition and the combination of a non-drowsy antihistamine with a decongestant, a formulation still preferred by many for moderate to severe congestion.
- The extended-release formulations (12-hour and 24-hour) continue to hold a niche, offering convenience and sustained symptom relief.
The financial trajectory of branded Claritin-D is characterized by a mature product lifecycle, with sales revenue primarily driven by brand loyalty and specific market segments, while the overall volume share in the combination allergy category is distributed among numerous generic competitors.
What are the R&D and Future Market Outlook Considerations?
The future market outlook for Claritin-D is influenced by several factors, including regulatory changes, therapeutic innovation, and consumer behavior.
- Continued Generic Competition: The robust generic market for loratadine and pseudoephedrine will continue to exert downward pressure on the pricing of branded Claritin-D.
- Regulatory Scrutiny of Pseudoephedrine: Ongoing concerns regarding the diversion of pseudoephedrine for illicit drug manufacturing may lead to further regulatory restrictions, potentially impacting sales volumes or necessitating product reformulations.
- Emergence of New Allergens and Resistance: Climate change and environmental factors may lead to increased allergen exposure or novel allergic responses, creating demand for new or improved allergy treatments.
- Innovation in Allergy Treatment:
- Development of novel antihistamines with improved efficacy or side-effect profiles.
- Advancements in immunotherapy (allergy shots and sublingual tablets) offering long-term relief.
- Research into biologics for severe allergic conditions.
- Combination products with novel ingredient pairings to address multifaceted allergy symptoms.
- Consumer Preference Shifts:
- Growing awareness and adoption of nasal steroid sprays as a first-line treatment for allergic rhinitis, due to their anti-inflammatory action.
- Increasing demand for natural or homeopathic allergy remedies, though clinical efficacy is often debated.
- Preference for single-ingredient products to target specific symptoms.
- Claritin-D's Position: Claritin-D, as an established combination product, will likely maintain a segment of the market, particularly among consumers seeking the specific dual action of a non-drowsy antihistamine and a decongestant. However, its growth potential is limited by its mature product status and the competitive pressures. Future R&D related to Claritin-D would likely focus on:
- Improved Formulations: Development of novel drug delivery systems to enhance patient compliance or efficacy, or to mitigate side effects.
- Combination Therapies: Investigating the synergy of loratadine/pseudoephedrine with other therapeutic agents for specific allergic conditions, though this would likely necessitate prescription status.
- Cost-Effective Manufacturing: Optimizing production processes to maintain competitiveness in the generic-dominated market.
The outlook suggests a stable but likely declining market share for branded Claritin-D, with the broader allergy market continuing to evolve with new therapeutic options and shifting consumer preferences.
What are the Financial Performance Indicators for Claritin-D?
Directly reporting current financial performance for a specific branded OTC product like Claritin-D is challenging due to proprietary reporting by parent companies. However, general indicators and historical context can be inferred.
- Revenue Trend: Branded Claritin-D's revenue has likely experienced a decline from its peak in the pre-patent expiration era. Post-expiration, revenues are primarily driven by:
- Price Premiums: A modest premium over generic equivalents, justified by brand trust and formulation consistency.
- Market Share Maintenance: Efforts to retain a portion of the market through marketing and brand loyalty programs.
- Extended-Release Versions: Sales of 12-hour and 24-hour formulations contribute a significant portion of branded revenue due to their specific value proposition.
- Profitability: While gross margins on established OTC drugs can be high, net profitability is impacted by:
- Marketing and Advertising Expenses: Continued investment is required to maintain brand visibility against generic competition and newer branded alternatives.
- Research and Development (R&D): Investment in line extensions or lifecycle management.
- Distribution and Sales Costs: Standard costs associated with retail distribution.
- Comparison to Generics: Generic versions of loratadine/pseudoephedrine generate substantial revenue volume for manufacturers due to their lower price point and widespread availability. The overall market for these products is large, but individual generic manufacturers compete on thin margins.
- Parent Company Performance: Bayer HealthCare, the current marketer, integrates Claritin-D's performance within its broader Consumer Health division. Financial reports for this division provide a broader view of the performance of brands like Claritin, Coppertone, and Alka-Seltzer, rather than granular data for individual products like Claritin-D. For example, Bayer’s annual reports detail the performance of its Consumer Health segment, often citing growth or decline in specific therapeutic areas like allergy relief, without isolating a single SKU's financials.
- Market Share Value: Despite declining unit volume for the branded product, the dollar value of Claritin-D sales, especially for extended-release versions, can remain significant due to its premium pricing compared to generics. However, the total market value for loratadine/pseudoephedrine combinations is now dominated by the aggregate sales of generic manufacturers.
The financial trajectory of branded Claritin-D reflects a mature product in a highly competitive OTC market, relying on brand equity and specific formulation benefits to maintain revenue, while the broader loratadine/pseudoephedrine market is primarily driven by generic volume.
Key Takeaways
Claritin-D, combining loratadine and pseudoephedrine, operates in a mature OTC allergy market. Patent expirations have led to significant generic competition, reducing the market share and pricing power of the branded product. Regulatory controls on pseudoephedrine due to its diversion potential for illicit methamphetamine production impose restrictions on sales and distribution. The market outlook for Claritin-D is shaped by ongoing generic pressure, evolving consumer preferences towards alternative treatments like nasal steroids, and the potential for further regulatory actions. While branded Claritin-D maintains a segment through brand recognition and extended-release formulations, the overall financial performance of the loratadine/pseudoephedrine combination market is now largely driven by the volume and pricing dynamics of generic manufacturers.
Frequently Asked Questions
1. What are the primary symptoms Claritin-D is designed to treat?
Claritin-D is formulated to treat symptoms associated with seasonal and year-round allergies, including sneezing, runny nose, itchy throat or nose, and nasal congestion.
2. Why is pseudoephedrine sold behind the counter?
Pseudoephedrine is sold behind the counter in many jurisdictions, including the United States under the Combat Methamphetamine Epidemic Act of 2005, because it can be used as a precursor in the illegal manufacture of methamphetamine.
3. Are there any significant side effects associated with Claritin-D?
Common side effects can include nervousness, sleeplessness, dizziness, headache, and nausea. Due to pseudoephedrine, individuals with certain medical conditions, such as heart disease, high blood pressure, or thyroid disease, should consult a healthcare professional before use.
4. How does Claritin-D compare to single-ingredient loratadine?
Single-ingredient loratadine (e.g., Claritin) targets only allergy symptoms driven by histamine, such as sneezing and runny nose, without addressing nasal congestion. Claritin-D adds pseudoephedrine to relieve nasal congestion.
5. What are the alternatives to Claritin-D for managing allergy symptoms?
Alternatives include single-ingredient antihistamines (loratadine, cetirizine, fexofenadine), nasal corticosteroid sprays (fluticasone, triamcinolone), decongestant nasal sprays (oxymetazoline, phenylephrine), and various combination products with different active ingredients.
Citations
[1] U.S. Food and Drug Administration. (n.d.). New Drug Approvals. Retrieved from https://www.fda.gov/drugs/new-drugs-fda-approves/new-drug-approvals
[2] United States Congress. (2005). Combat Methamphetamine Epidemic Act of 2005. Public Law 109-162.
[3] Bayer AG. (Annual Reports). Annual Reports. Retrieved from https://www.bayer.com/en/investors/reports-publications
[4] U.S. Patent and Trademark Office. (n.d.). Patent Search. Retrieved from https://patft.uspto.gov/