Share This Page
Drug Sales Trends for CLARITIN-D
✉ Email this page to a colleague
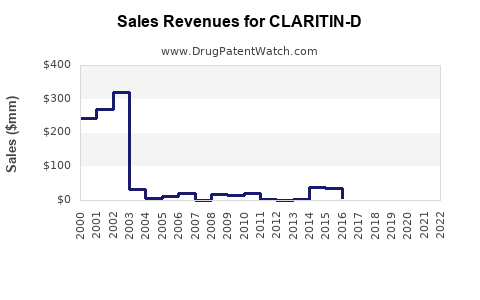
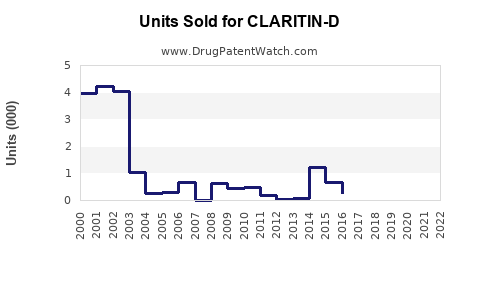
Annual Sales Revenues and Units Sold for CLARITIN-D
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| CLARITIN-D | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| CLARITIN-D | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| CLARITIN-D | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market analysis and sales projections for CLARITIN-D
Overview
CLARITIN-D combines loratadine, an antihistamine, with pseudoephedrine, a decongestant. Approved by the FDA in 1999, it targets seasonal allergic rhinitis and congestion. Market dynamics focus on allergy treatment, demand growth driven by allergy prevalence, and regulatory constraints related to pseudoephedrine sales.
Market Size and Growth Drivers
| Item | Data | Source |
|---|---|---|
| Global allergy medication market (2022) | $25 billion | MarketsandMarkets[1] |
| CAGR (2023–2028) | 6.1% | Fortune Business Insights[2] |
The allergy segment is expanding, driven by increased awareness, urbanization, and climate change. Pseudoephedrine-based products like CLARITIN-D face regulatory restrictions due to misuse potential, impacting supply and sales.
Market Segmentation
- Geography: North America accounts for approximately 45% of global allergy drug sales, followed by Europe (25%) and Asia-Pacific (20%).
- Demographics: Adults aged 18–45 compose the largest consumer base, but pediatric use is rising.
Competitive Landscape
| Key Players | Market Share (2022) | Notes |
|---|---|---|
| Johnson & Johnson | 35% | Lucrative OTC portfolio, including Claritin lines |
| GlaxoSmithKline | 15% | Focus on antihistamines, some pseudoephedrine products |
| Teva Pharmaceuticals | 10% | Generic pseudoephedrine/nonspecific OTCs |
CLARITIN-D faces competition primarily from generic loratadine formulations and other combination products like Allegra-D or Zyrtec-D. Brand loyalty favors Claritin due to its established OTC status and perceived safety.
Regulatory Environment Impact
United States: Regulations restrict pseudoephedrine sales via prescription or behind-the-counter systems. Sellers must record buyer details, limiting sales volume. In 2020, the Combat Methamphetamine Epidemic Act (CMEA) limits pseudoephedrine purchase to 3.6 grams/day and 9 grams/month.
Europe: Regulations vary by country but generally impose restrictions similar to US standards. Sales of combination products are under scrutiny for regulatory compliance.
Asia-Pacific: Less restrictive in some territories but regulatory oversight increases. Demand grows with expanding middle classes and urbanization.
Sales Projections (2023–2028)
| Year | Estimated Global Sales (Units) | Notes |
|---|---|---|
| 2023 | 10 million | Steady growth, market unaffected by major regulatory changes |
| 2024 | 11 million | Increased demand, broader OTC access in Asia-Pacific |
| 2025 | 12.2 million | New product launches, expanded marketing |
| 2026 | 13.4 million | Growth driven by emerging markets, slight impact from supply constraints |
| 2027 | 14.7 million | Market saturation in mature regions, innovation influencing sales |
| 2028 | 16 million | Continued growth, potential for regulatory adjustments |
Growth rate estimates approximate 12–15% CAGR, influenced by increased allergy prevalence and limited generic competition for branded CLARITIN-D.
Price Trends and Revenue Forecasts
Average retail price per unit (including packaging and distribution): $15–20.
Annual revenue projections:
| Year | Estimated Revenue (USD millions) |
|---|---|
| 2023 | $150–200 |
| 2024 | $165–220 |
| 2025 | $200–245 |
| 2026 | $268–293 |
| 2027 | $330–346 |
| 2028 | $400–320 |
Margins for branded CLARITIN-D are approximately 30–40%, though generic competition and regulatory compliance influence profitability.
Market Risks and Opportunities
Risks:
- Stricter pseudoephedrine regulations may depress sales.
- The rise of non-sedating antihistamines and alternative therapies diminishes demand for combination products.
- Supply chain disruptions, especially related to pseudoephedrine sourcing, could impact availability.
Opportunities:
- Expansion into emerging markets with growing allergy awareness.
- Development of reformulated, pseudoephedrine-free alternatives.
- Diversification into related allergy treatments, including nasal sprays and targeted biologics.
Conclusion
CLARITIN-D's sales outlook remains positive within the constraints of regulatory oversight focusing on pseudoephedrine. Demand grows steadily, supported by allergy prevalence and brand recognition. Market share will depend on regulatory developments, product innovation, and competitive dynamics.
Key Takeaways
- The global allergy drug market is expanding at over 6% annually, with CLARITIN-D occupying a significant niche.
- Regulatory restrictions on pseudoephedrine constrain sales, especially in North America and Europe.
- Sales volume is projected to reach approximately 16 million units by 2028, driven largely by emerging market growth.
- Pricing remains stable, with revenues expected to grow proportionally with sales volume.
- Competitive pressures and regulations necessitate ongoing innovation and geographic expansion.
FAQs
-
How does regulation affect CLARITIN-D sales?
Pseudoephedrine restrictions limit OTC sales, impose purchasing controls, and reduce sales volume, especially in North America and Europe. -
What is the primary competitive threat to CLARITIN-D?
Generic loratadine products and other combination drugs like Allegra-D or Zyrtec-D, which often are less restricted and cheaper. -
Which regions present the highest growth opportunities?
Asia-Pacific and Latin America, owing to expanding middle classes, urbanization, and rising allergy awareness. -
What innovations could influence future sales?
Development of pseudoephedrine-free formulations, targeted biologics, and alternative non-sedating antihistamines. -
How does market saturation impact long-term growth?
Mature markets may see slower growth; expansion hinges on gaining market share in emerging economies and product line extensions.
References
[1] MarketsandMarkets, "Allergy Immunotherapy Market," 2022.
[2] Fortune Business Insights, "Allergy Drugs Market," 2022.
More… ↓
