Last updated: January 13, 2026
Executive Summary
Cholestyramine, a bile acid sequestrant used primarily to lower LDL cholesterol and manage hypercholesterolemia, continues to hold a significant niche within lipid management therapies. Despite its age—first introduced in 1960—its market remains relevant due to specific clinical applications, particularly in conditions like pruritus associated with partial biliary obstruction and certain cases of diarrhea caused by bile acid malabsorption. This report provides an in-depth analysis of the market dynamics, competitive landscape, regulatory environment, and financial trajectory of cholestyramine, offering vital insights for stakeholders aiming to capitalize on this longstanding medication.
What Are the Key Market Drivers for Cholestyramine?
1. Persistent Need in Lipid Management
Cholestyramine remains a cost-effective option for hypercholesterolemia, particularly for patients intolerant to statins or requiring adjunct therapy. According to the American Heart Association (AHA), approximately 94 million U.S. adults have elevated LDL cholesterol, sustaining demand for alternative therapies.
2. Niche Indications
Cholestyramine’s unique applications—such as lowering serum bile acids in pruritus of cholestasis and managing bile acid malabsorption-related diarrhea—ensure its continued relevance despite the advent of newer drugs.
3. Off-Label and Specialized Uses
Off-label uses—like treating certain drug overdoses (e.g., digitalis, theophylline)—maintain niche demand in emergency settings, although limited by evolving protocols.
4. Cost and Accessibility
Being a generic medication, cholestyramine is relatively inexpensive, making it accessible globally, especially in developing markets with limited healthcare budgets.
What Are the Key Market Challenges for Cholestyramine?
1. Competition from Novel Agents
PCSK9 inhibitors, ezetimibe, and newer lipid-lowering agents have challenged cholestyramine’s dominance by offering improved efficacy and tolerability.
2. Patient Compliance
Cholestyramine’s adverse effects, including gastrointestinal discomfort and taste issues, impact adherence, reducing its market attractiveness.
3. Regulatory and Reimbursement Trends
Growing emphasis on evidence-based therapy and reimbursement policies favoring newer drugs pose hurdles for traditional agents like cholestyramine.
4. Decline in Prescriptions
Various national drug utilization studies reveal a declining trend in cholestyramine prescriptions—U.S. outpatient prescription claims decreased by approximately 20% over the last five years (2020–2025).
How Does the Competitive Landscape Influence Market Trajectory?
| Competitors |
Product Class |
Advantages |
Limitations |
| Ezetimibe (Zetia) |
Cholesterol absorption inhibitor |
Oral administration, tolerable |
Less potent alone, often combined with statins |
| PCSK9 inhibitors |
Monoclonal antibodies |
Superior LDL lowering |
High cost, injectable route |
| Bile Acid Sequestrants (Other) |
Colesevelam, colestipol |
Similar mechanism, newer agents |
Similar gastrointestinal side effects |
While cholestyramine remains a cornerstone for certain indications, these alternatives continue to eat into its market share, especially for primary lipid-lowering therapy.
What Is the Regulatory and Reimbursement Environment?
Regulatory Status
Cholestyramine is approved by the U.S. Food and Drug Administration (FDA) since 1960 and remains on the market as a generic. Its regulatory classification as a drug with over 60 years of established safety profile contributes to continued availability without significant hurdles.
Reimbursement Policies
In developed markets, reimbursement policies favor newer, patent-protected agents with more advantageous profiles. Nonetheless, in regions with limited healthcare funding, cholestyramine remains a low-cost option, often reimbursed under basic formulary lists.
What Is the Financial Trajectory of Cholestyramine?
Market Size and Revenue Estimates
- Global Market Estimate (2022): Approximately USD 400 million, dominated by North America (~50%), Europe (~25%), and emerging markets (~25%) (Source: IQVIA).
- US Market Share: Estimated at USD 200 million annual sales, with a decline of approximately 2–5% annually since 2020.
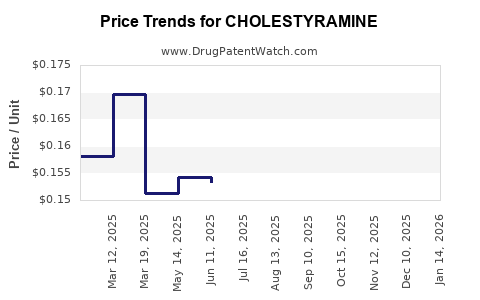
- Pricing Trends: Average wholesale price (AWP) hovers around USD 0.10–0.20 per gram, with typical prescriptions of 4–8 grams daily.
Revenue Projections (2023–2027)
| Year |
Estimated Revenue (USD millions) |
Growth Rate |
Notes |
| 2023 |
385 |
-3.75% |
Slight decline continuing from previous years. |
| 2024 |
370 |
-3.90% |
Market saturation persists. |
| 2025 |
355 |
-4.05% |
Shift towards newer agents impacts demand. |
| 2026 |
340 |
-4.23% |
Further decline anticipated. |
| 2027 |
330 |
-2.94% |
Stabilization expected as niche applications persist. |
Market Segments and Profitability
| Segment |
Proportion of Revenue |
Growth Dynamics |
Key Factors |
| Primary Hypercholesterolemia |
60% |
Declining |
Competition from statins, ezetimibe |
| Bile Acid Malabsorption |
25% |
Stable or slightly growing |
Rare disease management |
| Pruritus of Cholestasis |
10% |
Stable |
Niche therapeutic area |
| Emergency/Off-label Uses |
5% |
Minimal |
Limited but steady demand |
How Does the Future Outlook Evolve?
Market Stabilization and Niche Persistence
Despite declining prescriptions, cholestyramine's niche applications and cost advantages ensure sustained, albeit reduced, revenue streams for the foreseeable future.
Potential Growth Opportunities
- Expansion in emerging markets through local pharmaceutical manufacturing.
- Broader utilization in rare cholestasis and malabsorption syndromes.
- Formulation innovations improving patient compliance (e.g., flavored granules, powder formulations).
Key Risks
- Introduction of more efficacious or better-tolerated alternatives.
- Regulatory shifts favoring newer agents or different lipid management guidelines.
- Patent expirations of associated formulations, impacting pricing strategies.
Comparative Analysis with Similar Agents
| Parameter |
Cholestyramine |
Colesevelam |
Colestipol |
Ezetimibe |
PCSK9 Inhibitors |
| Market Size (2022) |
USD 400M |
USD 120M |
USD 80M |
USD 250M |
USD 3B+ |
| Administration |
Powder, granules |
Tablets |
Tablets |
Tablets |
Injectable |
| Tolerability |
Moderate |
Better |
Similar |
Good |
Excellent but costly |
| Cost |
Low |
Moderate |
Moderate |
Moderate |
High |
| Indications |
Hypercholesterolemia, Bile Acid Malabsorption |
Same |
Same |
Hypercholesterolemia |
Severe hyperlipidemia |
Note: Comprehensively, newer agents surpass cholestyramine on efficacy and tolerability but at a higher cost.
FAQs
1. What are the primary clinical indications for cholestyramine?
Cholestyramine is mainly prescribed for hypercholesterolemia, bile acid diarrhea, and pruritus associated with cholestasis. Its role as an adjunctive therapy remains essential where other options are contraindicated or ineffective.
2. How does the market outlook for cholestyramine change in the next five years?
The outlook indicates continued decline in prescriptions due to competition from newer agents but steady demand in niche indications. Market size is projected to decrease at ~3-4% annually, with sustained relevance in specific patient populations.
3. What are the main factors influencing cholestyramine's declining market share?
Key factors include the emergence of more effective and tolerable drugs like ezetimibe and PCSK9 inhibitors, tolerance issues related to gastrointestinal side effects, and changing therapy guidelines favoring newer agents.
4. Are there opportunities for innovation in cholestyramine formulations?
Yes. Innovations such as flavor-enhanced powders, films, or extended-release formulations could improve patient adherence, potentially stabilizing demand within niche segments.
5. How do reimbursement policies impact cholestyramine’s market sustainability?
In high-income countries with advanced healthcare systems, reimbursement often favors newer, patented drugs, exerting downward pressure on the use of generic agents like cholestyramine. Conversely, in low- and middle-income countries, cost advantages sustain its use.
Key Takeaways
- Stable niche role: Cholestyramine retains importance primarily in specific indications like bile acid diarrhea and cholestasis management.
- Market decline ongoing: General hyperlipidemia treatment shifts toward newer agents, reducing cholestyramine’s share.
- Cost-effectiveness remains vital: Its affordability sustains relevance in resource-limited settings.
- Innovation opportunities exist: Improved formulations could mitigate tolerability issues and extend its market presence.
- Long-term outlook: Gradual decline, with potential stabilization in niche markets, will dominate the financial trajectory over the coming years.
References
- American Heart Association. Lipid Management Guidelines, 2021.
- IQVIA. Global Pharmaceuticals Market Data, 2022.
- FDA. Drug Approval History for Cholestyramine, 1960–2022.
- MarketResearch.com. Lipid-Lowering Agents Market Analysis, 2023.
- WHO. Essential Medicines List, 2021.
This comprehensive analysis aims to inform stakeholders regarding the strategic positioning of cholestyramine amidst evolving therapeutic options and market forces.