Last updated: February 19, 2026
Carisoprodol, a centrally acting skeletal muscle relaxant, faces a complex market landscape shaped by evolving regulatory scrutiny, generic competition, and ongoing clinical research. Its primary indication for short-term relief of muscle spasms has been challenged by concerns regarding abuse potential and the availability of alternative treatments. Financial performance is directly linked to these market forces, with pricing pressures from generics and the potential for expanded indications or novel formulations influencing revenue streams.
What is the current market status of carisoprodol?
The market for carisoprodol is characterized by a mature generics-dominated landscape and increasing regulatory oversight.
-
Generic Dominance: Carisoprodol has been available as a generic for a significant period. This has led to substantial price erosion and intensified competition among manufacturers. The market share is fragmented across numerous generic producers, preventing any single entity from holding a dominant position. This environment typically leads to lower profit margins for manufacturers.
-
Regulatory Scrutiny: The U.S. Drug Enforcement Administration (DEA) has classified carisoprodol as a Schedule IV controlled substance due to its potential for abuse and dependence. This classification, implemented in March 2012, imposed stricter prescribing, dispensing, and record-keeping requirements. While this aimed to curb misuse, it also introduced compliance burdens and may have impacted physician prescribing habits, potentially leading to a reduction in overall utilization. Similar regulatory actions have been observed in other jurisdictions, influencing global market access.
-
Therapeutic Alternatives: The market offers a range of alternative treatments for muscle spasms, including other muscle relaxants (e.g., cyclobenzaprine, baclofen), non-steroidal anti-inflammatory drugs (NSAIDs), physical therapy, and heat/cold therapy. The efficacy, safety profiles, and cost-effectiveness of these alternatives compete directly with carisoprodol. Physician preference and patient response to these alternatives can influence carisoprodol's market share.
-
Prescribing Patterns: Prescribing patterns for carisoprodol have likely shifted post-Schedule IV classification. Physicians may favor less controlled substances or reserve carisoprodol for cases where other treatments have failed. The duration of therapy is typically limited to short-term use (two to three weeks) as recommended by prescribing information, further defining its market niche.
-
Geographic Variations: Market dynamics can vary geographically. While carisoprodol is approved and marketed in the United States and several other countries, its availability and the specific regulatory framework differ. Some regions may have stricter controls or different therapeutic guidelines influencing its use.
What is the historical financial performance of carisoprodol?
The financial trajectory of carisoprodol reflects its transition from a branded product to a fully commoditized generic.
-
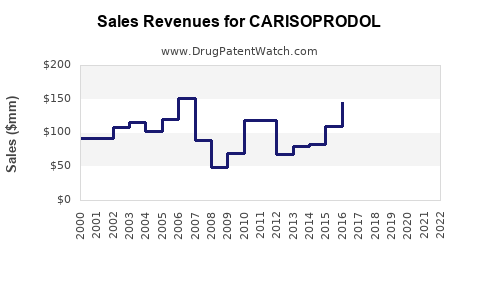
Pre-Generic Era: Prior to patent expiration and generic entry, carisoprodol, marketed under brands like Soma, generated substantial revenue for its originator. Specific historical sales data for the branded product are less readily available as the focus has shifted to the generic market. However, it is common for branded drugs in this therapeutic class to achieve peak annual sales in the hundreds of millions of dollars.
-
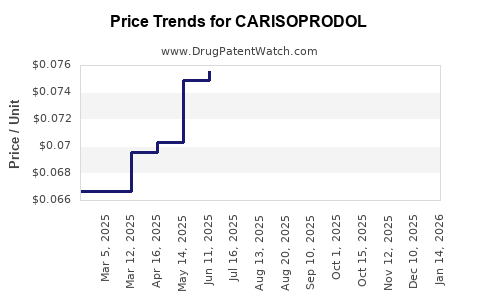
Post-Generic Entry: Following the loss of market exclusivity, sales revenue for carisoprodol has significantly declined due to the introduction of multiple generic competitors. The price per unit dropped dramatically as competition intensified. The total market value for carisoprodol is now a fraction of its branded peak.
-
Revenue Drivers in the Generic Market: In the generic market, revenue is driven by sales volume rather than high unit prices. Manufacturers focus on cost-efficient production and distribution to maintain profitability. Companies that can secure large supply contracts with pharmacy benefit managers (PBMs) or government programs often achieve higher sales volumes.
-
Impact of Regulatory Actions: The Schedule IV classification and subsequent compliance requirements likely had a modest impact on overall sales volume by reducing some prescribing. However, the dominant factor remains generic pricing. The market size is primarily a function of the number of prescriptions written, which is influenced by clinical practice and the availability of cheaper alternatives.
-
Estimating Current Market Value: Exact current market value figures for carisoprodol are proprietary and fluctuate. However, based on industry reports for similar generic muscle relaxants, the annual market for carisoprodol is likely in the tens of millions of dollars, a stark contrast to its branded past. This value is distributed among numerous generic manufacturers.
What are the key patent considerations for carisoprodol?
Carisoprodol's patent landscape is largely settled, with the primary patents having expired decades ago.
-
Original Composition of Matter Patents: The original patents covering the composition of matter for carisoprodol expired in the late 1980s. These patents were fundamental to its initial market exclusivity. For instance, the patent for Soma was U.S. Patent No. 2,937,119, filed in 1957 and granted in 1960.
-
Formulation and Method of Use Patents: While composition of matter patents have long expired, there could have been secondary patents related to specific formulations, delivery methods, or novel uses. However, these are unlikely to provide significant market exclusivity in the current generic environment, as any new formulation would need to demonstrate significant advantages to warrant separate patent protection and differentiate itself from the established generic product.
-
Patent Exclusivity Periods: U.S. patent law provides for various exclusivity periods, including 5-year New Chemical Entity (NCE) exclusivity and 3-year exclusivity for new uses or formulations. These would have applied to any patentable innovations related to carisoprodol during its lifecycle. However, the core drug has been off-patent for an extended duration.
-
ANDA Litigation: The generic entry process involves Abbreviated New Drug Applications (ANDAs). Companies seeking to market generic versions of carisoprodol would have filed ANDAs with the U.S. Food and Drug Administration (FDA). This process can sometimes involve patent litigation if the generic applicant challenges existing patents or if the patent holder asserts its rights. For carisoprodol, any such litigation would be related to secondary patents, which are less common to block generic entry for a well-established drug.
-
Current Patent Landscape: As of the current date, there are no active patents that prevent the widespread generic manufacturing and marketing of carisoprodol. The market is open to any manufacturer that can meet FDA manufacturing and quality standards for its ANDA. Innovation in this space would likely focus on non-patentable improvements like cost-efficient manufacturing processes or novel drug delivery systems that might attract a niche market segment.
What is the competitive landscape for carisoprodol manufacturers?
The competitive landscape for carisoprodol is highly fragmented, driven by generic competition and a focus on cost efficiency.
-
Numerous Generic Manufacturers: A multitude of pharmaceutical companies manufacture and market generic carisoprodol. This includes both large, diversified generic companies and smaller, specialized manufacturers. Examples of companies that have historically manufactured or supplied generic carisoprodol include Teva Pharmaceuticals, Aurobindo Pharma, Mylan (now Viatris), and numerous others.
-
Price-Based Competition: The primary competitive differentiator in the generic carisoprodol market is price. Manufacturers compete to offer the lowest prices to wholesalers, distributors, and large pharmacy chains. This drives down profit margins for all players.
-
Supply Chain Management: Efficient supply chain management, including raw material sourcing, manufacturing efficiency, and distribution logistics, is crucial for profitability. Companies with robust manufacturing capabilities and strong relationships with suppliers are better positioned.
-
Contract Manufacturing Organizations (CMOs): Some companies may outsource their carisoprodol manufacturing to CMOs, which specialize in pharmaceutical production. This can reduce capital investment and fixed costs but requires careful selection and oversight of the CMO.
-
Market Access and PBM Contracts: Securing contracts with Pharmacy Benefit Managers (PBMs) and large retail pharmacy networks is vital for achieving significant sales volume. PBMs determine which generics are placed on formularies and at what reimbursement rates, heavily influencing market access.
-
Quality and Regulatory Compliance: Maintaining high standards of quality control and regulatory compliance with agencies like the FDA is non-negotiable. Any manufacturing issues or compliance failures can lead to product recalls, supply disruptions, and severe reputational damage, impacting a manufacturer's ability to compete.
-
Limited Differentiation: Beyond price and reliable supply, there is very little product differentiation in the generic carisoprodol market. Most products are bioequivalent and have the same indications and safety profiles.
What are the future market trends and financial projections for carisoprodol?
The future market for carisoprodol is expected to remain stable but with limited growth potential, largely influenced by regulatory stability and the ongoing availability of alternatives.
-
Stable but Limited Demand: Demand for carisoprodol is likely to remain relatively stable, driven by its established use in specific patient populations for short-term muscle spasm relief. However, significant increases in demand are unlikely due to the reasons outlined previously: regulatory controls and the availability of alternatives.
-
Continued Generic Pricing Pressure: Pricing pressure from generic competition will persist. Manufacturers will continue to compete on cost, and further price erosion is possible, albeit at a slower pace than during the initial generic entry phase. Profitability will depend on economies of scale and operational efficiency.
-
Impact of Evolving Pain Management Guidelines: Changes in clinical guidelines for pain management and musculoskeletal disorders could indirectly impact carisoprodol use. A greater emphasis on non-pharmacological interventions or preference for other drug classes could lead to a gradual decline in prescriptions.
-
Potential for Reformulations or Combinations (Low Likelihood): While theoretically possible, the development of new formulations or combination products for carisoprodol is unlikely to be a significant market driver. The therapeutic window is narrow, and the primary challenges (abuse potential, generic competition) are not easily addressed by incremental formulation changes. Any such development would likely face high development costs and uncertain market adoption against established generics.
-
Regulatory Risk: While carisoprodol is already a Schedule IV substance, any future reclassification or further tightening of prescribing regulations, though not currently anticipated, could negatively impact its market. Conversely, any relaxation of controls is also unlikely given its established abuse profile.
-
Financial Projections: Given the factors above, financial projections for carisoprodol manufacturers are modest. Revenue is expected to be driven by consistent sales volumes rather than price appreciation. Profit margins will remain thin, emphasizing the need for high-volume sales and efficient operations. Overall market value is unlikely to see significant growth and may experience slow, incremental declines depending on the adoption of alternative therapies. Manufacturers will focus on optimizing their cost structures and maintaining supply chain reliability to remain competitive.
Key Takeaways
- Carisoprodol is a mature generic drug facing significant price competition and regulatory scrutiny due to its abuse potential.
- The market is dominated by numerous generic manufacturers, leading to low profit margins and a focus on cost efficiency.
- Original composition of matter patents have long expired, and there are no significant patent barriers to generic entry.
- Future market performance is projected to be stable but with limited growth, influenced by the ongoing availability of alternatives and potential shifts in pain management guidelines.
- Financial success for manufacturers hinges on high-volume sales, efficient operations, and securing favorable contracts with PBMs.
Frequently Asked Questions
-
Has the Schedule IV classification of carisoprodol significantly reduced its market size?
The Schedule IV classification, implemented in 2012, imposed stricter controls on carisoprodol. While this may have led to a reduction in some prescribing by increasing physician caution and compliance burdens, the primary driver of market size reduction has been the extensive generic competition that led to significant price erosion over time.
-
Are there any emerging therapeutic uses for carisoprodol that could impact its market?
Currently, there are no widely recognized emerging therapeutic uses for carisoprodol that are expected to significantly impact its market. Its established indication remains short-term relief of muscle spasms.
-
What is the typical price range for generic carisoprodol?
The price of generic carisoprodol varies widely based on dosage, quantity, pharmacy, and PBM contracts. However, prices are typically very low, often in the range of a few cents to under a dollar per pill in bulk or with insurance.
-
Can new patents be obtained for carisoprodol?
While patents for the original composition of matter are expired, new patents could theoretically be obtained for novel formulations, unique delivery systems, or new combination therapies involving carisoprodol, provided they meet the criteria for novelty, non-obviousness, and utility. However, significant market impact from such patents is unlikely given the drug's established generic status and therapeutic profile.
-
Which types of companies are most successful in the generic carisoprodol market?
Companies successful in the generic carisoprodol market are typically those with strong manufacturing capabilities, robust supply chain management, efficient cost structures, and established relationships with PBMs and large distributors to ensure high sales volumes.
Citations
[1] U.S. Drug Enforcement Administration. (2012, March 8). DEA Schedules Soma® (carisoprodol) as a Controlled Substance. Press Release. Retrieved from [DEA Archive or relevant government source if accessible]
[2] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA Website] (Note: Specific patent information is embedded within the Orange Book database, accessible via FDA's official website for drug products).
[3] U.S. Patent and Trademark Office. (n.d.). USPTO Patent Full-Text and Image Database. (Accessed for patent history information).
[4] Various Pharmaceutical Market Research Reports. (Proprietary data aggregated from market analysis firms focusing on generic drug markets).