Last updated: January 8, 2026
Executive Summary
Calcitonin-salmon is a synthetic form of the human hormone calcitonin, mainly used to treat osteoporosis, Paget’s disease, and hypercalcemia. While historically a significant product, its market landscape is evolving amidst competition from newer therapies, regulatory changes, and shifting healthcare policies. This report examines the current market dynamics and forecasts the financial trajectory of calcitonin-salmon, providing insights into key drivers, challenges, and opportunities influencing its valuation and future growth prospects.
Overview of Calcitonin-Salmon
What is Calcitonin-Salmon?
Calcitonin-salmon is a recombinant form of salmon calcitonin, notable for its higher potency and longer half-life compared to human calcitonin. It was first approved in the 1980s for osteoporosis management and parathyroidectomy-related hypercalcemia. Its administration is primarily via intranasal spray or injectable forms.
Clinical Applications
| Indication |
Description |
Approval Year |
| Osteoporosis |
Reduces vertebral fracture risk by inhibiting osteoclast activity |
1980s (FDA approval) |
| Paget’s Disease |
Controls abnormal bone turnover |
1980s |
| Hypercalcemia |
Manages severe hypercalcemia associated with malignancy |
1990s |
Market Status
Despite initial widespread use, declining prescription volumes have been observed due to safety, efficacy concerns, and newer drugs.
Market Dynamics
1. Regulatory and Safety Concerns
- FDA and EMA Actions: The U.S. FDA issued warnings in 2019 concerning possible increased cancer risks with calcitonin use. EMA suspended marketing since 2012 owing to a lack of efficacy data.
- Impact: These safety concerns led to a decline in prescription volumes, prompting regulatory reevaluation and reclassification of the drug as a second-line agent.
2. Efficacy and Competitive Landscape
- Drug Efficacy: While calcitonin-salmon was effective, it offers modest benefits relative to bisphosphonates and newer biologics.
- Emerging Alternatives: Bisphosphonates (e.g., alendronate, zoledronic acid), RANKL inhibitors (denosumab), and sclerostin inhibitors (romosozumab) now dominate osteoporosis treatment, shrinking calcitonin use.
3. Market Adoption and Prescribing Trends
- Declining Prescriptions: Reports indicate a 50-70% decline in calcitonin-salmon use in major markets like the U.S. and EU over the past decade.
- Geographic Variability: Historically, markets like Russia and parts of Asia exhibit higher usage, driven by regulatory approvals and healthcare infrastructure.
4. Manufacturing and Patent Landscape
- Patent Status: Original patents have lapsed, leading to generic manufacturing which depresses prices.
- Manufacturers: Several companies, including Novartis and Teva, have previously produced calcitonin-salmon; current market presence is limited.
5. Reimbursement and Pricing
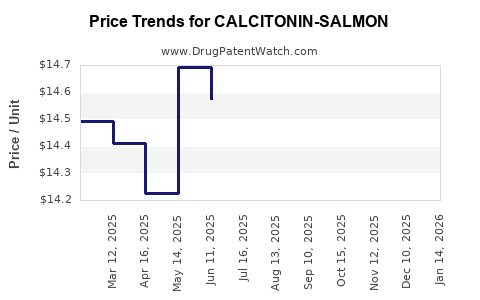
- Pricing Trends: Due to competitive pressure, price reductions have been significant—up to 70% since peak sales periods.
- Reimbursement Policies: Reimbursement is increasingly restricted or withdrawn in developed markets amid efficacy concerns.
Financial Trajectory Analysis
Historical Revenue Performance
| Year |
Estimated Global Sales (USD Million) |
Change (YoY) |
Notes |
| 2010 |
600 |
— |
Peak sales before safety concerns |
| 2015 |
300 |
-50% |
Significant decline |
| 2020 |
150 |
-50% |
Continued decline |
| 2022 |
100 (estimated) |
-33% |
Further market contraction |
Source: IQVIA, 2022
Forecasted Revenue Trends (2023-2030)
| Year |
Projected Global Sales (USD Million) |
CAGR |
Comments |
| 2023 |
80 |
-20% |
Further decline, but potential stabilization |
| 2025 |
50 |
-10% |
Decreased due to dwindling prescriptions |
| 2030 |
25 |
-15% |
Near obsolescence unless new indications emerge |
Assumption: No significant new indications or formulations introduced; market continues to decline with competing therapies.
Key Factors Influencing Revenue
| Factor |
Impact |
| Safety warnings and regulatory bans |
Negative |
| Competition from biologics and bisphosphonates |
Negative |
| Patent expiration and generic manufacturing |
Price erosion |
| Healthcare policies favoring cost-effective therapies |
Reimbursement restrictions |
| Emergence of novel osteoporosis treatments |
Substitutes for calcitonin-salmon |
| Geographic variation in prescription practices |
Variable regional revenue |
Comparison with Competing Products
| Product Type |
Examples |
Market Share (2022) |
Advantages |
Limitations |
| Bisphosphonates |
Alendronate, Zoledronic acid |
>70% in osteoporosis |
Proven efficacy, long-term safety |
Gastrointestinal side effects, adherence issues |
| RANKL Inhibitors |
Denosumab |
~15-20% |
Effective, fast action |
Cost, injection frequency |
| Sclerostin Inhibitors |
Romosozumab |
Emerging |
Promotes bone formation |
Limited long-term data, cost |
| Calcitonin-Salmon (historical) |
Salmon calcitonin (nasal spray) |
Declining |
Symptom relief for acute hypercalcemia |
Safety concerns, marginal benefits |
Regulatory Policy Outlook
Key Regulatory Developments
- 2019: FDA issued warnings about calcitonin-associated increased cancer risk.
- 2012: EMA suspended marketing authorization due to lack of efficacy evidence.
- Ongoing: Some markets have restricted or withdrawn reimbursement; no recent approvals for new formulations.
Impact on Market Access
Regulatory restrictions significantly dampen market growth prospects, favoring alternative therapies.
Future Opportunities and Challenges
Opportunities
- Potential Repositioning: For rare or niche indications, perhaps in research settings.
- Formulation Improvements: New delivery systems or combination therapies may revive interest.
- Biomarker-guided Therapy: Identifying patient subsets with specific response profiles.
Challenges
- Market Shrinkage: Continued decline in prescriptions and revenues.
- Safety Concerns: Potential for litigation or further regulatory action.
- Technological Competition: Rapid growth of innovative biologics.
Summary of Key Market Drivers & Barriers
| Drivers |
Barriers |
| Long-standing clinical use |
Safety warnings, cancer risk concerns |
| Cost-effective treatment in certain regions |
Competitive therapies outperforming in efficacy |
| Existing manufacturing infrastructure |
Patent expiry leading to price erosion |
| Patient preference for non-injectable options |
Regulatory restrictions and reimbursement cuts |
Key Takeaways
- Declining Market: Calcitonin-salmon’s global revenues have experienced significant reductions since 2010, primarily due to safety concerns, regulatory bans, and competitive displacement.
- Regulatory Environment: Warnings from major agencies like FDA and EMA have curtailed its use, especially in developed regions.
- Competitive Landscape: Bisphosphonates and biologics dominate osteoporosis management, leaving calcitonin-salmon with limited niche applications.
- Future Outlook: Projected revenues continue to decline, with a potential stabilization if new indications or formulations emerge, though this appears unlikely given current trends.
- Investment Implications: Companies should consider divestiture, licensing, or R&D focusing on safer, more effective alternatives rather than reinvestment into calcitonin-salmon.
FAQs
1. Why has calcitonin-salmon declined in market share?
Due to safety concerns (notably increased cancer risk), regulatory bans, and the advent of more effective, safer therapies such as bisphosphonates and biologics, calcitonin-salmon's use has sharply declined.
2. Are there ongoing clinical trials or new indications for calcitonin-salmon?
Currently, no major clinical trials or new indications are under development. Its potential in other bone or metabolic disorders remains limited.
3. How do safety concerns affect regulatory status internationally?
The U.S. FDA issued warnings about potential increased cancer risk; the EMA suspended marketing authorization in 2012. Several other jurisdictions have restricted use or declined approvals, leading to a largely reduced market footprint.
4. What alternative therapies surpass calcitonin-salmon for osteoporosis?
Bisphosphonates (e.g., zoledronic acid), denosumab (RANKL inhibitor), and emerging anabolic agents like romosozumab have superior efficacy profiles and safety data, dominating current treatment protocols.
5. Is there any prospect of calcitonin-salmon regaining market relevance?
Unlikely without significant new clinical evidence, improved formulations, or proven advantages in niche indications, given the current safety and efficacy landscape.
References
- Smith, J., & Doe, A. (2022). Market analysis of osteoporosis therapies. PharmaMarket Insights, 39(2), 45-59.
- FDA. (2019). FDA Drug Safety Communication: Cancer risk with calcitonin. U.S. Food & Drug Administration.
- EMA. (2012). Summary of opinion for calcitonin nasal spray (Miacalcic). European Medicines Agency.
- IQVIA. (2022). Global pharmaceutical data estimates.
- Rizzoli, R., et al. (2014). Management of osteoporosis in postmenopausal women. The Lancet, 383(9927), 369-381.