BRIVIACT Drug Patent Profile
✉ Email this page to a colleague
When do Briviact patents expire, and what generic alternatives are available?
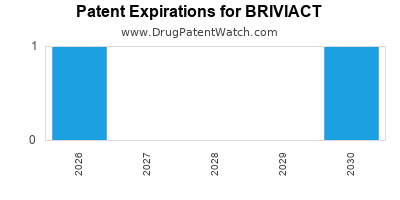
Briviact is a drug marketed by Ucb Inc and is included in three NDAs. There are two patents protecting this drug and three Paragraph IV challenges.
This drug has one hundred and seventy-three patent family members in forty-five countries.
The generic ingredient in BRIVIACT is brivaracetam. Fifteen suppliers are listed for this compound. Additional details are available on the brivaracetam profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Briviact
A generic version of BRIVIACT was approved as brivaracetam by SUNSHINE on June 9th, 2022.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for BRIVIACT?
- What are the global sales for BRIVIACT?
- What is Average Wholesale Price for BRIVIACT?
Summary for BRIVIACT
| International Patents: | 173 |
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 3 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 41 |
| Clinical Trials: | 15 |
| Patent Applications: | 1,094 |
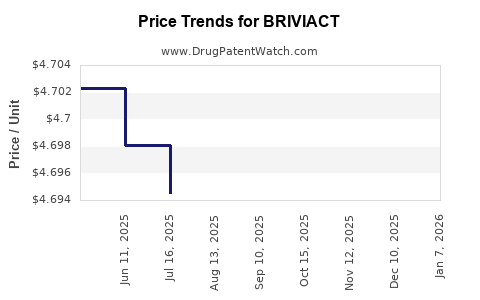
| Drug Prices: | Drug price information for BRIVIACT |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for BRIVIACT |
| What excipients (inactive ingredients) are in BRIVIACT? | BRIVIACT excipients list |
| DailyMed Link: | BRIVIACT at DailyMed |
Recent Clinical Trials for BRIVIACT
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| The Affiliated Nanjing Drum Tower Hospital of Nanjing University Medical School | PHASE1 |
| Jiangsu Chia Tai Fenghai Pharmaceutical Co., Ltd. | PHASE1 |
| University of California, Los Angeles | Phase 1/Phase 2 |
Pharmacology for BRIVIACT
| Mechanism of Action | Epoxide Hydrolase Inhibitors |
Paragraph IV (Patent) Challenges for BRIVIACT
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| BRIVIACT | Tablets | brivaracetam | 10 mg, 25 mg, 50 mg, 75 mg and 100 mg | 205836 | 7 | 2020-05-12 |
| BRIVIACT | Injection | brivaracetam | 50 mg/5 mL | 205837 | 2 | 2020-05-12 |
| BRIVIACT | Oral Solution | brivaracetam | 10 mg/mL | 205838 | 1 | 2020-05-12 |
US Patents and Regulatory Information for BRIVIACT
BRIVIACT is protected by four US patents.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ucb Inc | BRIVIACT | brivaracetam | SOLUTION;INTRAVENOUS | 205837-001 | May 12, 2016 | AP | RX | Yes | Yes | 6,911,461 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | |
| Ucb Inc | BRIVIACT | brivaracetam | TABLET;ORAL | 205836-002 | May 12, 2016 | AB | RX | Yes | No | 10,729,653 | ⤷ Start Trial | Y | ⤷ Start Trial | ||
| Ucb Inc | BRIVIACT | brivaracetam | TABLET;ORAL | 205836-001 | May 12, 2016 | AB | RX | Yes | No | 6,911,461 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | |
| Ucb Inc | BRIVIACT | brivaracetam | TABLET;ORAL | 205836-005 | May 12, 2016 | AB | RX | Yes | Yes | 6,911,461 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | |
| Ucb Inc | BRIVIACT | brivaracetam | TABLET;ORAL | 205836-003 | May 12, 2016 | AB | RX | Yes | No | 6,911,461 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for BRIVIACT
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Ucb Inc | BRIVIACT | brivaracetam | TABLET;ORAL | 205836-005 | May 12, 2016 | 8,492,416 | ⤷ Start Trial |
| Ucb Inc | BRIVIACT | brivaracetam | SOLUTION;ORAL | 205838-001 | May 12, 2016 | 6,784,197 | ⤷ Start Trial |
| Ucb Inc | BRIVIACT | brivaracetam | SOLUTION;INTRAVENOUS | 205837-001 | May 12, 2016 | 8,492,416 | ⤷ Start Trial |
| Ucb Inc | BRIVIACT | brivaracetam | SOLUTION;INTRAVENOUS | 205837-001 | May 12, 2016 | 6,784,197 | ⤷ Start Trial |
| Ucb Inc | BRIVIACT | brivaracetam | TABLET;ORAL | 205836-001 | May 12, 2016 | 6,784,197 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for BRIVIACT
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| UCB Pharma SA | Briviact (in Italy: Nubriveo) | brivaracetam | EMEA/H/C/003898Briviact is indicated as adjunctive therapy in the treatment of partial-onset seizures with or without secondary generalisation in adult and adolescent patients from 16 years of age with epilepsy. | Authorised | no | no | no | 2016-01-13 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for BRIVIACT
When does loss-of-exclusivity occur for BRIVIACT?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 10215646
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 1007161
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 47395
Estimated Expiration: ⤷ Start Trial
China
Patent: 2292071
Estimated Expiration: ⤷ Start Trial
Patent: 4083328
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0141006
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 15673
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 91349
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 2057
Patent: ФАРМАЦЕВТИЧЕСКИЕ КОМПОЗИЦИИ, ВКЛЮЧАЮЩИЕ БРИВАРАЦЕТАМ (PHARMACEUTICAL COMPOSITIONS COMPRISING BRIVARACETAM)
Estimated Expiration: ⤷ Start Trial
Patent: 1101116
Patent: ФАРМАЦЕВТИЧЕСКИЕ КОМПОЗИЦИИ, ВКЛЮЧАЮЩИЕ ПРОИЗВОДНЫЕ 2-ОКСО-1-ПИРРОЛИДИНА
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 91349
Patent: Compositions pharmaceutiques comprenant des dérivés de 2-oxo-1-pyrrolidine (PHARMACEUTICAL COMPOSITIONS COMPRISING 2-OXO-1-PYRROLIDINE DERIVATIVES)
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 61988
Patent: 包含 -氧代- -吡咯烷衍生物的藥物組合物 (PHARMACEUTICAL COMPOSITIONS COMPRISING 2-OXO-1-PYRROLIDINE DERIVATIVES 2--1-)
Estimated Expiration: ⤷ Start Trial
Patent: 98287
Patent: 包含 -氧代- -吡咯烷衍生物的藥物組合物 (PHARMACEUTICAL COMPOSITIONS COMPRISING 2-OXO-1-PYRROLIDINE DERIVATIVES 2--1-)
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 3545
Patent: תכשירי רוקחות המכילים תולדות 2-אוקסו-1-פירולידין (Pharmaceutical compositions comprising 2-oxo-1-pyrrolidine derivatives)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 76401
Estimated Expiration: ⤷ Start Trial
Patent: 12516302
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 11007267
Patent: COMPOSICIONES FARMACEUTICAS QUE COMPRENDEN DERIVADOS DE 2-OXO-1-PIRROLIDINA. (PHARMACEUTICAL COMPOSITIONS COMPRISING 2-OXO-1-PYRROLIDINE DERIVATIVES.)
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 927
Patent: FARMACEUTSKE KOMPOZICIJE KOJE SADRZE 2-OKSO-1-PIROLIDIN DERIVATE (PHARMACEUTICAL COMPOSITIONS COMPRISING 2-OXO-1-PYRROLIDINE DERIVATIVES)
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 91349
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 91349
Estimated Expiration: ⤷ Start Trial
San Marino
Patent: 01400155
Patent: Composizioni farmaceutiche comprendenti derivati di 2-osso-1-pirrolidina
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 554
Patent: FARMACEUTSKE KOMPOZICIJE KOJE SADRŽE 2-OKSO-1-PIROLIDIN DERIVATE (PHARMACEUTICAL COMPOSITIONS COMPRISING 2-OXO-1-PYRROLIDINE DERIVATIVES)
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 91349
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1640164
Estimated Expiration: ⤷ Start Trial
Patent: 120008022
Patent: PHARMACEUTICAL COMPOSITIONS COMPRISING 2-OXO-1-PYRROLIDINE DERIVATIVES
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 11047
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering BRIVIACT around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Czech Republic | 304420 | 2-Oxo-1-pyrrolidinové deriváty, způsob jejich přípravy a jejich použití (2-Oxo-1-pyrrolidine derivatives, process of their preparation and their use) | ⤷ Start Trial |
| Russian Federation | 2005125569 | ⤷ Start Trial | |
| Australia | 5214401 | ⤷ Start Trial | |
| Iceland | 7919 | ⤷ Start Trial | |
| Poland | 365159 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for BRIVIACT
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1452524 | C01452524/01 | Switzerland | ⤷ Start Trial | PRODUCT NAME: BRIVARACETAM; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 65830 06.10.2016 |
| 2391349 | C20160006 | Estonia | ⤷ Start Trial | PRODUCT NAME: BRIVARATSETAAM; NATIONAL AUTHORISATION NUMBER: EMA/CHMP/1199/2023 13.03.2023 |
| 1452524 | 1690009-4 | Sweden | ⤷ Start Trial | PRODUCT NAME: BRIVARACETAM; REG. NO/DATE: EU/1/15/1073 20150114 |
| 1265862 | PA2016013 | Lithuania | ⤷ Start Trial | PRODUCT NAME: BRIVARACETAMAS; REGISTRATION NO/DATE: EU/1/15/1073 20160114 |
| 1452524 | 2016/011 | Ireland | ⤷ Start Trial | PRODUCT NAME: BRIVARACETAM; REGISTRATION NO/DATE: EU/1/15/1073 20160114 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for BRIVIACT
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.