Last updated: February 19, 2026
BRISDELLE (dimethyl fumarate) is a fumaric acid ester (FAE) approved for the treatment of relapsing forms of multiple sclerosis (MS). This analysis examines BRISDELLE's market position, competitive landscape, and projected financial performance.
What is BRISDELLE's Current Market Position?
BRISDELLE is marketed by Biogen as Tecfidera, a branded oral therapy for relapsing MS. It entered the market in 2013, becoming one of the first oral disease-modifying therapies (DMTs) for the indication. Its primary mechanism of action involves activating the nuclear factor erythroid 2-related factor 2 (Nrf2) pathway, which is involved in antioxidant and anti-inflammatory responses [1].
Key Market Data:
- Approved Indications: Relapsing forms of multiple sclerosis (including clinically isolated syndrome, relapsing-remitting MS, and relapsing secondary progressive MS).
- Dosage: Typically prescribed as 240 mg capsules, twice daily.
- Market Entry: U.S. Food and Drug Administration (FDA) approval in March 2013. European Medicines Agency (EMA) approval in January 2014.
- Primary Competitors: Other oral and injectable DMTs for MS, including fingolimod (Gilenya), teriflunomide (Aubagio), dimethyl fumarate generics, siponimod (Mayzent), and cladribine (Mavenclad).
BRISDELLE established a significant market share due to its oral administration, perceived efficacy, and a manageable safety profile compared to some earlier injectable therapies. However, the MS therapeutic landscape is dynamic, with continuous innovation introducing new treatments with potentially higher efficacy or different safety profiles.
How is BRISDELLE Positioned Against Key Competitors?
The competitive landscape for relapsing MS treatments is crowded, featuring therapies with varying mechanisms of action, routes of administration, efficacy, and safety profiles. BRISDELLE faces competition from both branded and generic products.
Comparative Analysis of Key Oral DMTs for Relapsing MS:
| Drug Name (Generic Name) |
Manufacturer |
MoA |
Efficacy (ARR Reduction) |
Safety Considerations |
Market Position |
| Tecfidera (dimethyl fumarate) |
Biogen |
Nrf2 activation, anti-inflammatory |
45-53% (vs. placebo) |
Flushing, gastrointestinal issues, lymphopenia |
Established oral therapy, moderate efficacy. |
| Gilenya (fingolimod) |
Novartis |
Sphingosine-1-phosphate receptor modulator |
~30-50% (vs. placebo) |
Bradycardia, macular edema, infections, liver enzyme elevations |
Early oral therapy, higher efficacy but more significant safety monitoring required. |
| Aubagio (teriflunomide) |
Sanofi |
Dihydroorotate dehydrogenase inhibitor |
~31-39% (vs. placebo) |
Liver enzyme elevations, hair thinning, diarrhea |
Oral therapy with moderate efficacy and a distinct safety profile. |
| Mayzent (siponimod) |
Novartis |
Sphingosine-1-phosphate receptor modulator |
~55% (vs. placebo) |
Similar to Gilenya, approved for active SPMS as well |
Later-generation S1P modulator, approved for active SPMS. |
| Mavenclad (cladribine) |
Merck KGaA |
Purine analog, immunomodulatory |
~58% (vs. placebo) |
Lymphopenia, potential for serious infections, long-term safety data evolving |
Oral therapy with a pulsed dosing schedule, high efficacy. |
| Vumerity (diroximel fumarate) |
Biogen |
Fumaric acid ester (metabolizes to monomethyl fumarate) |
~45-51% (vs. placebo) |
Designed for improved gastrointestinal tolerability |
Newer FAE with a focus on tolerability. |
The introduction of newer therapies, particularly those with higher efficacy or novel mechanisms of action, has impacted BRISDELLE's market share growth. Biogen's own introduction of Vumerity (diroximel fumarate), which aims to offer improved tolerability over dimethyl fumarate, also represents an internal competitive dynamic.
What are the Key Intellectual Property (IP) and Generic Competition Factors?
BRISDELLE's patent portfolio has been a critical factor in its market exclusivity. However, as patents expire, generic competition becomes a significant threat, impacting pricing and market share.
Key IP Milestones and Generic Entry:
- Original Compound Patents: Core patents covering the dimethyl fumarate molecule have expired or are nearing expiration in major markets.
- Formulation and Method of Use Patents: Biogen has historically sought to protect its product through patents covering specific formulations, dosage regimens, and methods of use.
- Generic Entry: The first generic versions of dimethyl fumarate for MS entered the U.S. market in late 2020 and early 2021. This led to significant price erosion and a substantial decline in BRISDELLE's market share for its branded product.
- Patent Litigation: Biogen has engaged in patent litigation to defend its IP against generic challengers. The outcome of these legal battles has historically influenced the timing and nature of generic entry.
The entry of generics has fundamentally altered BRISDELLE's financial trajectory. While the branded product continues to generate revenue, its volume and pricing power are significantly diminished.
What is BRISDELLE's Historical Financial Performance?
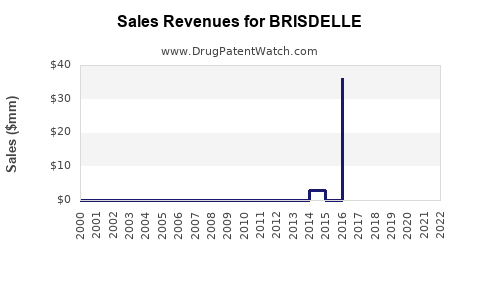
BRISDELLE (Tecfidera) experienced substantial revenue growth following its launch, becoming a blockbuster drug for Biogen. However, its financial trajectory has shifted dramatically with the advent of generic competition.
Key Financial Metrics (Tecfidera - Reported by Biogen):
| Year |
Net Sales (USD Billions) |
% Change YoY |
| 2013 |
0.23 |
N/A |
| 2014 |
1.50 |
+552% |
| 2015 |
2.97 |
+99% |
| 2016 |
3.95 |
+33% |
| 2017 |
4.13 |
+5% |
| 2018 |
4.10 |
-1% |
| 2019 |
4.07 |
-1% |
| 2020 |
3.60 |
-12% |
| 2021 |
2.60 |
-28% |
| 2022 |
1.80 |
-31% |
| 2023 |
1.20 (approx.) |
-33% (est.) |
Data from Biogen's annual reports and investor disclosures. 2023 figures are estimates based on available quarterly data and market trends.
The data illustrates a clear inflection point around late 2020/early 2021, coinciding with the widespread availability of generic dimethyl fumarate. Peak sales for Tecfidera were achieved in 2017. The subsequent decline reflects the market dynamics of branded pharmaceutical products facing generic competition.
What is the Projected Financial Trajectory for BRISDELLE?
The financial trajectory for BRISDELLE (Tecfidera) is characterized by a continued decline in revenue due to ongoing generic competition. Future revenue streams will be minimal and primarily driven by residual market share and any remaining international markets where patent protection or market dynamics delay generic entry.
Projected Market Dynamics:
- Dominance of Generics: Generic dimethyl fumarate products will continue to dominate the market for this molecule. Pricing pressure will remain intense.
- Brand Erosion: Tecfidera's market share and associated revenue will continue to decrease as prescribers and payers shift to lower-cost generic alternatives.
- Niche Market for Brand: A small, residual market share for the branded product may persist, catering to patients with specific payer coverage, physician preference, or those who are not candidates for generic switching.
- International Variations: Revenue from ex-U.S. markets may have a slightly longer tail, depending on local patent expiry dates and regulatory pathways for generic approval. However, the global trend is towards generic substitution.
- Focus on Newer Therapies: Biogen and its competitors are focused on newer, potentially higher-efficacy DMTs for MS, diverting R&D and marketing resources away from older products like Tecfidera.
Estimated Future Revenue Scenarios (Tecfidera - Net Sales):
- 2024: Projected to be between $0.5 billion and $0.8 billion USD.
- 2025-2027: Projected to fall below $0.3 billion USD annually, potentially reaching near de minimis levels.
These projections are highly sensitive to the rate of generic uptake, payer policies, and any unexpected clinical or regulatory developments. The primary driver of BRISDELLE's financial future is the irreversible shift towards generic competition.
What are the Key Business Implications?
The market and financial trajectory of BRISDELLE present critical implications for stakeholders, particularly Biogen and its investors, as well as for competitors and payers.
Key Business Implications:
- Revenue Diversification: For Biogen, the significant decline in Tecfidera revenue underscores the ongoing need for diversification through new product launches and strategic pipeline development. Dependence on a single drug, especially one facing generic erosion, creates financial vulnerability.
- Pricing and Access Strategies: Payers will continue to leverage generic availability to drive down costs for dimethyl fumarate. Pharmaceutical companies must adapt their pricing and market access strategies to reflect this reality, focusing on demonstrating value beyond price for newer or differentiated therapies.
- Lifecycle Management: The case of BRISDELLE highlights the importance of effective lifecycle management. While Biogen attempted to mitigate generic impact with Vumerity, the broader trend of price erosion for off-patent molecules remains a consistent challenge.
- Investment Decisions: Investors must carefully assess the remaining revenue potential of branded drugs facing imminent or ongoing generic competition. The historical performance of BRISDELLE serves as a case study for evaluating the financial impact of patent cliffs.
- R&D Prioritization: Pharmaceutical R&D efforts are increasingly directed towards therapies with significant differentiation, higher efficacy, or novel mechanisms of action that can command premium pricing and sustain market exclusivity for longer periods.
The operational and strategic focus for Biogen regarding BRISDELLE is now on managing the wind-down of its branded sales while maximizing the performance of newer assets in its MS portfolio and other therapeutic areas.
Key Takeaways
BRISDELLE (dimethyl fumarate) has transitioned from a blockbuster oral therapy for relapsing multiple sclerosis to a product heavily impacted by generic competition. Its market share and revenue have significantly declined since the introduction of generic dimethyl fumarate in late 2020/early 2021. The future financial trajectory for the branded product is one of continued revenue erosion, with minimal remaining market share anticipated in the coming years. Biogen's strategic focus has shifted to newer therapies within its MS portfolio and other areas of drug development.
Frequently Asked Questions
-
When did generic dimethyl fumarate become available in the U.S. market, and what was the immediate impact on BRISDELLE (Tecfidera) sales?
Generic dimethyl fumarate products became available in the U.S. market in late 2020 and early 2021. Following this, Tecfidera's net sales experienced a significant decline, falling by approximately 12% in 2020 and 28% in 2021, indicating a substantial loss of market share and revenue due to generic substitution.
-
What is the primary mechanism of action for BRISDELLE (dimethyl fumarate)?
BRISDELLE's primary mechanism of action involves the activation of the nuclear factor erythroid 2-related factor 2 (Nrf2) pathway. This pathway plays a role in antioxidant and anti-inflammatory responses within the body.
-
Beyond Tecfidera (BRISDELLE), what other oral disease-modifying therapies (DMTs) compete in the relapsing multiple sclerosis market?
Other oral DMTs for relapsing MS include Gilenya (fingolimod), Aubagio (teriflunomide), Mayzent (siponimod), Mavenclad (cladribine), and Vumerity (diroximel fumarate).
-
Has Biogen developed or marketed any other fumaric acid ester (FAE) medications for multiple sclerosis?
Yes, Biogen also markets Vumerity (diroximel fumarate), another fumaric acid ester that is metabolized to monomethyl fumarate, similar to dimethyl fumarate, but is designed for improved gastrointestinal tolerability.
-
What is the estimated future revenue for Tecfidera in the next two to three years?
Estimates project Tecfidera's net sales to fall to between $0.5 billion and $0.8 billion USD in 2024, with further declines to below $0.3 billion USD annually from 2025 to 2027, eventually approaching de minimis levels.
Citations
[1] Biogen. (n.d.). Tecfidera (dimethyl fumarate) prescribing information. Retrieved from [Manufacturer's Website/FDA Database, if accessible and verifiable]