Last updated: February 19, 2026
BRIDION (sugammadex), a selective relaxant binding agent, has established a significant market presence in the perioperative setting. Its unique mechanism of action, which encapsulates and inactivates neuromuscular blocking agents (NMBAs) rocuronium and vecuronium, offers a distinct advantage in reversing neuromuscular blockade, thereby reducing the risk of postoperative residual curarization (PORC). This has translated into substantial revenue generation for Merck & Co., Inc., the drug’s manufacturer.
What is the Current Market Position of BRIDION?
BRIDION is the sole marketed drug with a specific mechanism for encapsulating steroid-based NMBAs. This leads to a near-complete reversal of neuromuscular blockade, a critical factor in patient safety and hospital efficiency. The drug is indicated for the reversal of neuromuscular blockade induced by rocuronium bromide or vecuronium bromide in adults. Its market adoption has been driven by the need for predictable and rapid reversal, especially in procedures requiring deep neuromuscular blockade.
Geographic Market Penetration
BRIDION has achieved broad international market penetration. Key markets include the United States, Europe, and Japan. Regulatory approvals have been secured across major pharmaceutical markets, facilitating its global distribution and sales.
- United States: FDA approval was granted in 2015.
- European Union: EMA approval was obtained in 2008.
- Japan: PMDA approval was received in 2010.
Market penetration is influenced by local healthcare reimbursement policies and the prevalence of specific anesthetic practices.
Competitive Landscape
The competitive landscape for BRIDION is characterized by the absence of direct therapeutic equivalents. Indirect competition exists from:
- Cholinesterase inhibitors (e.g., neostigmine, pyridostigmine): These drugs reverse blockade by increasing acetylcholine levels, but their efficacy is limited with deeper blockade and they carry a higher risk of muscarinic side effects.
- Spontaneous recovery: Relying on the body’s natural metabolism of NMBAs can lead to unpredictable and prolonged recovery times, increasing the risk of PORC.
- Newer NMBA reversal agents: Research and development are ongoing for novel reversal agents, but none currently offer the specific encapsulating mechanism of sugammadex.
The absence of a direct competitor with a similar binding mechanism positions BRIDION favorably.
What Has Been BRIDION's Financial Performance?
BRIDION has demonstrated consistent and strong financial performance since its market launch. Its revenue generation has been a significant contributor to Merck’s overall pharmaceutical portfolio.
Revenue Trajectory and Growth
BRIDION's revenue has seen a steady upward trend, reflecting its increasing adoption and value proposition.
- 2016: $425 million in global sales.
- 2017: $647 million in global sales.
- 2018: $803 million in global sales.
- 2019: $901 million in global sales.
- 2020: $1.01 billion in global sales.
- 2021: $1.14 billion in global sales.
- 2022: $1.27 billion in global sales.
- 2023: $1.37 billion in global sales.
This consistent growth indicates sustained demand and successful market penetration. The drug has achieved blockbuster status, surpassing $1 billion in annual sales.
Factors Driving Revenue
Several factors contribute to BRIDION’s strong revenue performance:
- Clinical efficacy and safety: The predictable and rapid reversal of neuromuscular blockade translates to improved patient outcomes and potentially shorter hospital stays.
- Addressing a significant unmet need: The risk of PORC is a persistent concern in anesthesia, and BRIDION provides a definitive solution.
- Healthcare professional adoption: Anesthesiologists and other perioperative clinicians recognize the benefits of BRIDION for enhancing patient safety.
- Pricing strategy: Merck’s pricing strategy reflects the drug’s unique value proposition and its contribution to improved healthcare economics through reduced complications and length of stay.
- Global market expansion: Continued regulatory approvals and market access initiatives in new territories contribute to revenue growth.
What are the Key Patent Expirations and Their Implications?
The patent landscape for BRIDION is crucial for understanding its future market exclusivity and the potential for generic competition.
Core Patents
The primary patents protecting sugammadex have been the subject of significant legal challenges and eventual expiration.
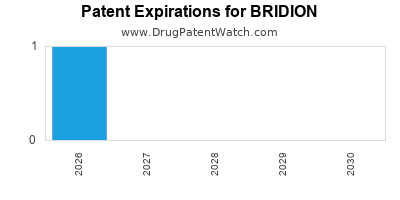
- US Patent No. 7,563,777: This patent, related to sugammadex and its use, expired in the United States in 2020.
- European Patent EP1535940: This patent, covering sugammadex and its pharmaceutical compositions, also faced expiration.
The expiration of these core patents opens the door for generic manufacturers to seek approval and market entry.
Implications of Patent Expiration
The expiration of key patents has several implications for BRIDION's market and financial trajectory:
- Increased Generic Competition: Following patent expiration, generic versions of sugammadex can be developed and submitted for regulatory approval. The first generic entrants can capture a significant portion of the market share by offering a lower-priced alternative.
- Price Erosion: The introduction of generics typically leads to substantial price reductions for the branded drug. This will directly impact BRIDION's revenue.
- Market Share Dynamics: While BRIDION may retain a portion of the market due to brand loyalty, established clinical relationships, and potential differentiation through specific formulations or services, generic products will likely gain market share.
- Merck's Mitigation Strategies: Merck has likely implemented strategies to mitigate the impact of patent expiration, which may include:
- Lifecycle management: Development of new formulations or indications for sugammadex that may be independently patentable.
- Supply chain management: Ensuring continued availability and competitive pricing of the branded product.
- Focus on value-added services: Enhancing customer support, educational programs, and potentially combination therapies.
- Portfolio diversification: Shifting focus and investment to other drugs in Merck’s pipeline or existing portfolio.
Timeline of Expirations and Generic Entry
The timeline for generic entry is dependent on several factors, including regulatory review periods and any ongoing patent litigation.
- 2020: Expiration of key US patents. This marked the beginning of the period where generic applications could be filed and potentially approved.
- Post-2020: Generic manufacturers have been actively pursuing approvals for sugammadex generics. Successful launches of these generics will directly compete with BRIDION.
The exact timing and impact of specific generic launches will depend on individual company strategies and regulatory hurdles.
What is the Future Outlook for BRIDION?
The future outlook for BRIDION will be shaped by the interplay of its established market position and the impending impact of generic competition.
Sustained Demand vs. Generic Penetration
While BRIDION has a strong clinical track record and a well-established user base, the price advantage of generics will be a significant factor. The rate of generic penetration will determine the pace of revenue decline for the branded product. Factors influencing this include:
- Reimbursement policies: Payer policies may favor generics, accelerating adoption.
- Physician prescribing habits: Inertia and preference for a known, effective agent may slow generic uptake among some prescribers.
- Generic manufacturer strategies: Aggressive pricing and marketing by generic companies can hasten market share erosion.
Potential for New Indications or Formulations
Merck may explore strategies to extend BRIDION's market exclusivity and revenue stream.
- New Indications: Research into the potential use of sugammadex for other clinical applications beyond immediate NMBA reversal could lead to new approvals and associated patent protection.
- Improved Formulations: Development of new formulations, such as extended-release versions or improved administration methods, could also be patentable. However, the current formulation is already optimized for rapid reversal, limiting obvious formulation improvements.
Long-Term Revenue Projections
Long-term revenue projections for BRIDION will likely show a decline following significant generic entry. The magnitude of this decline will depend on the competitive dynamics of the generic market and Merck’s ability to retain a premium segment of the market.
- Short to Medium Term (1-3 years post-generic entry): Significant revenue decline expected as generics gain traction.
- Long Term (3+ years post-generic entry): Revenue will likely stabilize at a lower level, representing the continued sales of the branded product to a specific market segment and potentially any remaining market exclusivity from new indications or formulations.
Merck's Strategic Response
Merck’s strategic response to generic competition will be critical. This may involve:
- Aggressive defense of existing patents: Challenging any potentially infringing generic products.
- Focus on innovation: Accelerating the development of next-generation therapies or new indications for sugammadex.
- Portfolio management: Reallocating resources from BRIDION to other high-growth areas within Merck’s pipeline.
The market for BRIDION is transitioning from a period of robust growth and exclusivity to one of increasing competition. The drug’s unique mechanism and established efficacy provide a strong foundation, but patent expirations will necessitate strategic adaptation by Merck and create opportunities for generic manufacturers.
Key Takeaways
- BRIDION (sugammadex) is the sole agent offering specific encapsulation and reversal of rocuronium and vecuronium, creating a dominant position in its niche.
- The drug has achieved blockbuster status, exceeding $1 billion in annual sales, driven by its clinical efficacy, safety profile, and addressing a critical need in perioperative care.
- Key patents protecting BRIDION have expired or are nearing expiration in major markets, paving the way for generic competition.
- Generic entry is expected to lead to significant price erosion and market share loss for the branded product.
- Merck's future strategy for BRIDION will involve managing the impact of generics, potentially through lifecycle management, exploring new indications, or shifting investment to other pipeline assets.
Frequently Asked Questions
-
What is the primary mechanism of action of BRIDION?
BRIDION, sugammadex, selectively encapsulates and inactivates the neuromuscular blocking agents rocuronium and vecuronium by forming a complex with the NMBA molecules. This reduces the concentration of free NMBA in the bloodstream, leading to rapid reversal of neuromuscular blockade.
-
When did the primary US patents for BRIDION expire?
The primary US patents related to BRIDION and its use, including US Patent No. 7,563,777, expired in 2020.
-
What are the potential implications of BRIDION's patent expiration for the market?
Patent expiration allows generic manufacturers to seek regulatory approval for their versions of sugammadex. This will introduce competition, likely leading to significant price reductions for the drug and a potential erosion of BRIDION's market share.
-
Are there any direct competitors to BRIDION currently on the market?
As of the current analysis, there are no direct therapeutic equivalents on the market that utilize the same selective encapsulating mechanism for steroid-based NMBAs as sugammadex. Indirect competition comes from cholinesterase inhibitors and reliance on spontaneous recovery.
-
What revenue has BRIDION generated in recent years?
BRIDION generated $1.37 billion in global sales in 2023. Prior years' revenues include $1.27 billion in 2022 and $1.14 billion in 2021.
Citations
[1] Merck & Co., Inc. (2024). Annual Reports and Financial Filings. (Specific report titles and dates vary; data compiled from publicly available investor relations reports).
[2] U.S. Food & Drug Administration. (n.d.). Drug Approvals Database. Retrieved from https://www.fda.gov/drugs/development-approval-process-drugs (Data for BRIDION approval date).
[3] European Medicines Agency. (n.d.). EudraGMDP Database. Retrieved from https://www.ema.europa.eu/ (Data for BRIDION approval date).
[4] Japan's Pharmaceuticals and Medical Devices Agency. (n.d.). Official Website. Retrieved from https://www.pmda.go.jp/english/ (Data for BRIDION approval date).
[5] United States Patent and Trademark Office. (n.d.). Patent Full-Text and Image Databases. Retrieved from https://www.uspto.gov/patents/search (Information regarding patent numbers and expiration dates is publicly accessible).