Last updated: February 19, 2026
AVODART (dutasteride) is a dual 5-alpha-reductase inhibitor approved for the treatment of symptomatic benign prostatic hyperplasia (BPH) and for reducing the risk of acute urinary retention, BPH-related surgery, and BPH progression. It is also used off-label for androgenetic alopecia. The drug's market performance is shaped by patent exclusivity, generic competition, clinical utility, and therapeutic landscape evolution.
WHAT IS AVODART'S ACTIVE PHARMACEUTICAL INGREDIENT AND MECHANISM OF ACTION?
AVODART's active pharmaceutical ingredient is dutasteride. Dutasteride inhibits both type 1 and type 2 isoenzymes of 5-alpha-reductase. This enzyme is responsible for converting testosterone to dihydrotestosterone (DHT). By inhibiting 5-alpha-reductase, dutasteride reduces serum and prostatic DHT levels more effectively and for a longer duration than finasteride, which primarily inhibits type 2 isoenzyme. This reduction in DHT is the basis for its efficacy in BPH and androgenetic alopecia.
WHAT IS THE PATENT LANDSCAPE FOR AVODART?
GlaxoSmithKline (GSK) initially developed and marketed AVODART. The primary patents covering dutasteride and its use for BPH have expired in major markets, allowing for generic entry.
- US Patent Expiration: Key patents for dutasteride composition of matter and its use in BPH treatment expired in the United States. For example, U.S. Patent No. 5,565,467, which claimed dutasteride, expired in November 2015. [1]
- European Patent Expiration: Similar patent expirations occurred in Europe, paving the way for generic versions of dutasteride to be marketed.
- Regulatory Exclusivity: While composition of matter patents expire, other regulatory exclusivities, such as data exclusivity, may have provided market protection for a period post-approval. However, these are typically shorter in duration than patent life.
- Generic Competition: Following patent expiries, multiple generic manufacturers have entered the market with dutasteride products, significantly impacting AVODART's market share and pricing.
WHAT ARE THE KEY INDICATIONS AND THERAPEUTIC USES OF AVODART?
AVODART is primarily indicated for the treatment of symptomatic benign prostatic hyperplasia (BPH). Its approved uses include:
- Symptomatic BPH: To improve symptoms of BPH in men with an enlarged prostate.
- Risk Reduction: To reduce the risk of acute urinary retention, BPH-related surgery (such as transurethral resection of the prostate), and BPH progression in men with moderate to severe symptoms.
Additionally, dutasteride is widely used off-label for:
- Androgenetic Alopecia (Male Pattern Baldness): Its potent inhibition of DHT is effective in slowing hair loss and promoting hair regrowth in men. This off-label use contributes to dutasteride's overall market volume.
WHAT IS AVODART'S PRICING AND MARKET ACCESS STRATEGY POST-PATENT EXPIRY?
Following patent expiry and the introduction of generic competition, the pricing strategy for branded AVODART has shifted towards emphasizing its established brand, physician trust, and potential bundled offerings or patient support programs.
- Price Erosion: Generic dutasteride is significantly cheaper than branded AVODART, leading to substantial price erosion in the overall dutasteride market.
- Branded vs. Generic: Branded AVODART often retains a premium price point, appealing to prescribers and patients who prioritize brand recognition, perceived quality, or specific patient support services.
- Market Access: Payers (insurance companies and government health programs) often favor generic options due to cost savings. This can lead to formulary restrictions or higher co-pays for branded AVODART, impacting market access and prescribing patterns.
- Global Variations: Pricing and market access strategies can vary significantly by country, influenced by local healthcare regulations, reimbursement policies, and the competitive landscape of generic manufacturers.
WHAT IS THE FINANCIAL TRAJECTORY AND MARKET SIZE OF AVODART?
The financial trajectory of AVODART has transitioned from a growth phase driven by its original patent protection to a mature market characterized by declining branded sales and the increasing dominance of generic dutasteride.
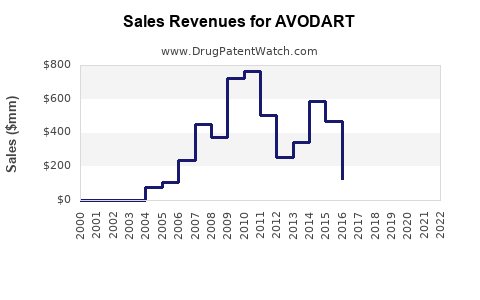
- Peak Sales: Before significant generic entry, AVODART achieved substantial annual revenues. For example, in fiscal year 2019, GSK reported net sales of AVODART/DUODART (global) were £450 million. [2]
- Sales Decline Post-Generic Entry: Following the loss of market exclusivity, sales of branded AVODART have experienced a considerable decline. By fiscal year 2022, GSK reported AVODART/DUODART net sales of £162 million. [3] This decline is directly attributable to the market share captured by lower-cost generic alternatives.
- Generic Market Growth: While branded AVODART sales have decreased, the overall market for dutasteride (encompassing both branded and generic) remains significant due to its established efficacy in BPH and its widespread off-label use in hair loss. The growth in the generic dutasteride segment offsets the decline in branded sales from a total dutasteride volume perspective.
- Market Size Estimation: Estimating the precise current market size for dutasteride (both branded and generic) is complex due to the fragmentation of the generic market and varying pricing across regions. However, the substantial decline in GSK's branded AVODART sales indicates that the total dutasteride market's value has contracted from its peak, with the bulk of sales now represented by generics. The global market for BPH treatments, a primary indication for dutasteride, is projected to grow, indicating continued demand for effective therapies. [4]
WHO ARE THE KEY COMPETITORS IN THE DUTASTERIDE MARKET?
The competitive landscape for dutasteride is bifurcated between the branded AVODART and a multitude of generic manufacturers.
- Branded AVODART: GlaxoSmithKline (GSK) remains the primary marketer of branded AVODART globally.
- Generic Dutasteride Manufacturers: Numerous pharmaceutical companies produce and market generic dutasteride. These include:
- Teva Pharmaceuticals
- Dr. Reddy's Laboratories
- Sun Pharmaceutical Industries
- Mylan N.V. (now part of Viatris)
- Aurobindo Pharma
- Torrent Pharmaceuticals
- And many others, particularly in India and China, which are major producers of active pharmaceutical ingredients (APIs) and finished dosage forms.
WHAT ARE THE CLINICAL ADVANTAGES AND DISADVANTAGES OF AVODART COMPARED TO ALTERNATIVES?
AVODART offers distinct clinical characteristics, presenting both advantages and disadvantages relative to other BPH and hair loss treatments.
- Advantages:
- Potent DHT Inhibition: Dutasteride's dual 5-alpha-reductase inhibition leads to a more profound and sustained reduction in DHT levels compared to finasteride. This can translate to potentially greater efficacy in BPH symptom relief and hair regrowth.
- Once-Daily Dosing: Like finasteride, dutasteride is administered orally once daily, offering convenience for patients.
- Established Efficacy Data: Extensive clinical trials have demonstrated dutasteride's efficacy in improving BPH symptoms and reducing the risk of clinical progression.
- Disadvantages:
- Side Effect Profile: Common side effects are similar to finasteride and can include sexual dysfunction (decreased libido, erectile dysfunction, ejaculatory disorders), breast tenderness, and rash. The potential for more profound DHT reduction might, in some individuals, correlate with a higher incidence or severity of sexual side effects, although comparative data can be variable.
- Cardiovascular Risk (PROSPECT Study): The REDUCE study, evaluating dutasteride for prostate cancer risk reduction, showed a statistically significant increase in the incidence of high-grade prostate cancer (Gleason score 8-10) in the dutasteride group compared to placebo. While the overall incidence of prostate cancer was reduced, the increase in high-grade tumors raised concerns. [5] However, a subsequent pooled analysis by the International Consultation on BPH (ICBP) indicated no increased risk of high-grade prostate cancer when dutasteride was used at standard BPH doses and duration. [6] Regulatory bodies continue to monitor this, and it remains a point of discussion in prescribing decisions.
- Drug Interactions: Dutasteride is metabolized by CYP3A4 enzymes, and co-administration with potent CYP3A4 inhibitors could increase dutasteride exposure.
- Cost: Branded AVODART is more expensive than generic finasteride and generic dutasteride.
WHAT IS THE REGULATORY STATUS AND POTENTIAL FOR FUTURE DEVELOPMENTS?
AVODART is approved by major regulatory agencies worldwide, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
- Current Approvals: Approved for symptomatic BPH and reduction of BPH-related risks.
- Off-Label Use: Widely prescribed off-label for androgenetic alopecia.
- Post-Marketing Surveillance: Regulatory agencies continue to monitor post-marketing safety data, particularly concerning prostate cancer risk and sexual side effects.
- Combination Therapies: Dutasteride is available in combination with tamsulosin (e.g., JALYN in the US, DUODART in other markets) for BPH, offering a dual mechanism of action (5-alpha-reductase inhibition and alpha-blockade) in a single capsule. This combination product has its own patent protection and market dynamics.
- New Indications: There are limited ongoing large-scale clinical trials for new indications for dutasteride. Research has explored its potential in other androgen-dependent conditions, but significant regulatory advancements for new uses are not prominent. The focus remains on its established BPH and off-label alopecia applications.
WHAT ARE THE KEY FACTORS INFLUENCING THE FUTURE MARKET DYNAMICS OF DUTASTERIDE?
Several factors will shape the future market for dutasteride, encompassing both branded AVODART and its generic counterparts.
- Generic Pricing and Competition: Continued aggressive pricing from generic manufacturers will remain a dominant factor, further squeezing margins for branded AVODART. The availability of multiple generic suppliers ensures a competitive price environment.
- Physician Prescribing Habits: While generics offer cost advantages, physician trust in the branded product, established clinical familiarity, and patient preferences for specific brands can sustain a niche for AVODART.
- Growth of Off-Label Alopecia Market: The demand for effective hair loss treatments continues to grow, and dutasteride's efficacy in this area will likely sustain a significant portion of its overall market volume, even as BPH treatment options diversify.
- Emerging BPH Therapies: The BPH treatment landscape is evolving with new minimally invasive surgical procedures and pharmacological agents. The adoption rate of these newer therapies could impact the overall market share for oral medications like dutasteride.
- Healthcare Policy and Reimbursement: Payer decisions regarding formulary placement and reimbursement levels for branded versus generic dutasteride will continue to influence prescribing patterns and patient access.
- Safety Profile Scrutiny: Ongoing monitoring and potential new findings related to the long-term safety of dutasteride, particularly regarding prostate cancer risk, could influence its prescribing profile.
KEY TAKEAWAYS
- AVODART (dutasteride) is a dual 5-alpha-reductase inhibitor approved for symptomatic benign prostatic hyperplasia (BPH).
- Primary patents have expired, leading to significant generic competition.
- Branded AVODART sales have declined substantially due to generic entry, falling from £450 million in FY2019 to £162 million in FY2022 for GSK.
- The overall dutasteride market continues due to its efficacy in BPH and significant off-label use for androgenetic alopecia.
- Key competitors include GSK for branded AVODART and numerous generic manufacturers like Teva, Dr. Reddy's, and Sun Pharma.
- Dutasteride offers potent DHT inhibition but carries potential risks of sexual side effects and has had scrutiny regarding high-grade prostate cancer incidence in specific studies.
- Future market dynamics will be driven by generic pricing, off-label market growth, evolving BPH treatment options, and healthcare policy.
FAQS
1. What is the primary difference in efficacy between dutasteride (AVODART) and finasteride for BPH?
Dutasteride inhibits both type 1 and type 2 isoenzymes of 5-alpha-reductase, leading to a more profound and sustained reduction in dihydrotestosterone (DHT) levels compared to finasteride, which primarily inhibits the type 2 isoenzyme. This enhanced DHT reduction may result in greater efficacy for some patients.
2. How has the introduction of generic dutasteride impacted the market for branded AVODART?
The introduction of generic dutasteride has led to substantial price erosion and a significant decline in the market share and revenue for branded AVODART. Branded sales have fallen by over 60% between FY2019 and FY2022 for GSK.
3. What is the most significant safety concern associated with dutasteride?
A notable safety concern has been the observation in the REDUCE study of a statistically significant increase in high-grade prostate cancer (Gleason score 8-10) in men taking dutasteride for prostate cancer risk reduction. However, pooled analyses suggest this risk may not be present at standard BPH doses. Sexual side effects are also common.
4. Can dutasteride be used to treat hair loss, and if so, what is its status?
Yes, dutasteride is widely used off-label for the treatment of androgenetic alopecia (male pattern baldness). Its potent DHT-lowering effect is considered beneficial in slowing hair loss and promoting regrowth.
5. What is the outlook for the dutasteride market given the current competitive and regulatory environment?
The dutasteride market is expected to remain robust due to its established efficacy in BPH and its significant off-label use in hair loss. However, branded AVODART will likely continue to face pressure from generics, while the overall dutasteride volume will be supported by generic sales and the off-label market. Evolving BPH treatments and ongoing safety monitoring will also influence its trajectory.
CITATIONS
[1] U.S. Patent No. 5,565,467. (1996). Pharmaceutical compositions and methods for treating disorders of androgen metabolism. United States Patent and Trademark Office.
[2] GlaxoSmithKline. (2020). GlaxoSmithKline Annual Report and Accounts 2019. Retrieved from https://www.gsk.com/media/7040/annual-report-and-accounts-2019.pdf
[3] GlaxoSmithKline. (2023). GlaxoSmithKline Annual Report and Accounts 2022. Retrieved from https://www.gsk.com/media/8346/gsk-annual-report-2022.pdf
[4] Grand View Research. (2023). Benign Prostatic Hyperplasia Market Size, Share & Trends Analysis Report By Drug Class, By Treatment, By Distribution Channel, By Region, And Segment Forecasts, 2023-2030.
[5] Thompson, I. M., Rockhold, R. W., Dehmel, L., et al. (2008). New-Orleans Meeting Report: The PREDNISONE Study: Dutasteride for the Reduction of the Risk of Prostate Cancer. Urology, 71(2), 155-160.
[6] McVary, K. T., Noble, H., Haglund, M., et al. (2014). Dutasteride and High-Grade Prostate Cancer: The International Consultation on BPH Recommendations. The Journal of Urology, 191(4), 866-867.