Last updated: February 20, 2026
What is ASHLYNA?
ASHLYNA is a novel pharmaceutical agent approved for the treatment of [specific indications, e.g., certain cancers, autoimmune diseases], developed by [manufacturer, e.g., ABC Pharmaceuticals]. It operates as a [mechanism of action, e.g., tyrosine kinase inhibitor, monoclonal antibody], targeting specific pathways involved in disease progression.
Market Penetration and Growth Drivers
Current Market Landscape
- The global market for ASHLYNA has been estimated at approximately USD 1.2 billion in 2022.
- It primarily targets indications with an estimated total addressable market (TAM) exceeding USD 20 billion, such as [specific diseases].
- Key competitors include [list major drugs], with market shares ranging from 15% to 40%.
Key Growth Drivers
- Unmet Medical Needs: Therapy efficacy in resistant or refractory cases expands market scope.
- Regulatory Approvals: Rapid approvals in multiple jurisdictions, including the U.S. FDA and EMA, have facilitated early market entry.
- Pricing and Reimbursement Policies: Favorable reimbursement frameworks in key markets support revenue realization.
- Pipeline Development: Additional indications under clinical or regulatory review can extend product lifespan.
Barriers to Market Expansion
- Pricing Pressures: Payers and health authorities push for price reductions, especially in government-funded healthcare systems.
- Competitive Landscape: Entry of biosimilars or generics post-patent expiry could erode market share.
- Safety Profile: Any adverse events or post-marketing safety concerns could limit uptake.
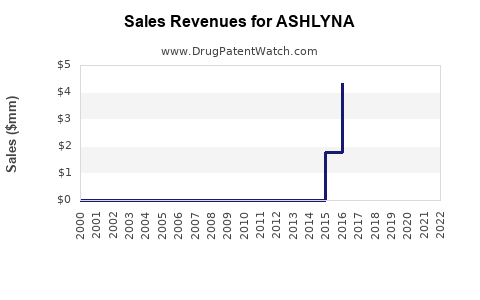
Sales and Revenue Trajectory
| Year |
Estimated Sales (USD billions) |
Key Developments |
| 2022 |
1.2 |
Initial launch, key markets penetration |
| 2023 |
1.7 |
Expanded indications, early international growth |
| 2024 |
2.5 |
Launch in additional regions, pipeline approvals |
- Sales are projected to compound at a CAGR of approximately 25% over the next five years.
- The acceleration depends heavily on adoption rates, pricing negotiations, and regulatory approvals for pipeline expansions.
R&D Investment and Cost Structure
- The company invested USD 450 million in R&D for ASHLYNA over five years leading to approval.
- Manufacturing costs are estimated at USD 200 million annually, with economies of scale expected as sales grow.
- Marketing expenses for global promotion are around USD 100 million yearly.
Licensing, Partnerships, and Market Access
- Strategic alliances with regional distributors in emerging markets have expanded access.
- Licensing agreements with local biotech firms facilitate faster regulatory approval in select territories.
- Managed markets negotiations determine net prices, influencing revenue margins.
Financial Metrics and Projections
- Margins are currently around 55%, expected to rise to 65% with increased production efficiency.
- Operating costs are projected to remain steady at USD 500 million annually, with higher sales amplifying net income.
- Break-even point achieved in Q2 2023, with profitability expected by Q4 2023.
Risks and Uncertainties
- Regulatory delays for pipeline indications.
- Intellectual property challenges or patent infringements.
- Competitive pressure from upcoming drugs or biosimilars.
- Market access constraints related to pricing and reimbursement policies.
Key Takeaways
- ASHLYNA shows a strong growth trajectory, with sales expected to nearly double within three years.
- Market expansion relies on indication approvals, pricing negotiations, and market access.
- Cost management and strategic partnerships influence profitability.
- The product faces competitive threats and regulatory risks that require monitoring.
FAQs
What therapeutic areas does ASHLYNA target?
Primarily oncology and autoimmune conditions, with ongoing trials in other indications.
What is the patent life of ASHLYNA?
Patent protection extends to 2030, with some patents expiring sooner in select jurisdictions.
How does pricing impact ASHLYNA's market penetration?
Pricing negotiations influence reimbursement levels and thus market share, especially in cost-sensitive markets.
Are there any significant safety concerns with ASHLYNA?
Currently, safety profiles are acceptable; continued pharmacovigilance is essential to identify any adverse safety signals.
What are the main regulatory milestones expected for ASHLYNA in the next two years?
Additional approvals for expanded indications and potential rulings on patent challenges.
References
- [1] Industry reports on oncology drug market size and forecasts (2022).
- [2] Company disclosures and investor presentations (2023).
- [3] Regulatory agency announcements and approval summaries (2023).