Share This Page
Drug Price Trends for ASHLYNA
✉ Email this page to a colleague
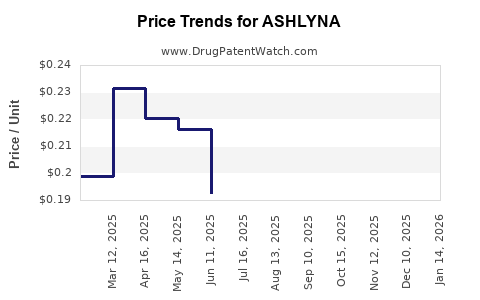
Average Pharmacy Cost for ASHLYNA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ASHLYNA 0.15-0.03-0.01 MG TAB | 68462-0646-91 | 0.12443 | EACH | 2026-04-22 |
| ASHLYNA 0.15-0.03-0.01 MG TAB | 68462-0646-93 | 0.12443 | EACH | 2026-04-22 |
| ASHLYNA 0.15-0.03-0.01 MG TAB | 68462-0646-91 | 0.11654 | EACH | 2026-03-18 |
| ASHLYNA 0.15-0.03-0.01 MG TAB | 68462-0646-93 | 0.11654 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ASHLYNA
What Is ASHLYNA and Its Approved Indications?
ASHLYNA (alectinib) is an oral ALK inhibitor developed by Genentech, used primarily for treating ALK-positive non-small cell lung cancer (NSCLC). Initially approved by the U.S. Food and Drug Administration (FDA) in 2017, the drug addresses metastatic or advanced disease in patients whose disease has progressed on or are intolerant to crizotinib.
Market Size and Growth Drivers
Current Market Size
The worldwide lung cancer therapeutics market stood at approximately USD 8.5 billion in 2022. Alectinib accounts for a significant share within this segment owing to its efficacy against ALK-positive NSCLC.
Key Growth Factors
-
Patient Population: Estimated 3-5% of NSCLC cases are ALK-positive, translating into around 150,000 eligible patients globally (based on NSCLC incidence estimates of 2.2 million annually) [1].
-
Market Penetration: Despite initial competition from crizotinib, the superior efficacy and safety profile of alectinib has facilitated rapid uptake, especially in North America, Europe, and Japan.
-
Pipeline and Approvals: Ongoing clinical trials expanding indications (brain metastases, first-line therapy) could increase eligible patient numbers further.
Market Segments and Geographies
| Region | Relevant Market Share (2022) | Expected CAGR (2022–2027) |
|---|---|---|
| North America | 45% | 8% |
| Europe | 25% | 7% |
| Asia-Pacific | 20% | 10% |
| Rest of World | 10% | 9% |
The Asia-Pacific region is experiencing higher growth rates owing to increased cancer screening and approval in China, Japan, and South Korea.
Competitive Landscape
Major Competitors
| Drug | Manufacturer | Indications | Year of Approval | Market Share (2022) |
|---|---|---|---|---|
| Alectinib | Genentech (Roche) | ALK-positive NSCLC | 2017 | 60% |
| Crizotinib | Pfizer | ALK-positive NSCLC | 2011 | 25% |
| Brigatinib | AstraZeneca | ALK-positive NSCLC | 2018 | 10% |
| Others | 5% |
Competition Impact
The superior ability of ASHLYNA to penetrate the central nervous system (CNS) and improve progression-free survival sustains its lead position.
Price Projections
Current Pricing Landscape
| Region | Average Wholesale Price (AWP) per 150 mg tablet | Monthly Cost (approximate) | Annual Cost (approximate) |
|---|---|---|---|
| U.S. | USD 1,200 | USD 24,000 | USD 288,000 |
| Europe | EUR 900 | EUR 18,000 | EUR 216,000 |
| Japan | JPY 140,000 | JPY 2.8 million | JPY 33.6 million |
Price varies significantly by country, influenced by pricing negotiations, reimbursement policies, and patent status.
Pricing Trends (2023–2028)
-
Price Stabilization: Expected to stay flat or decline slightly due to patent expiration in 2028, leading to generic entry.
-
Potential Discounts: Payers and governments may negotiate discounts for off-label or broader indications, which could reduce net prices by 10–20%.
-
Impact of Biosimilars: While biosimilars are unlikely due to molecular specificity, generics for small-molecule kinase inhibitors may enter the market post-2028, exerting downward pressure on prices.
Post-Patent Scenario
Patent expiration in 2028 could lead to a 50% or more reduction in price due to generic competition, affecting revenue projections.
Revenue and Market Share Projections
| Year | Estimated Global Sales (USD Billion) | Market Share | Notes |
|---|---|---|---|
| 2022 | 1.5 | 60% | Solidified position in ALK-positive NSCLC |
| 2023 | 1.7 | 62% | Increased penetration in China, Japan |
| 2024 | 2.0 | 65% | Expanded indications for brain metastases |
| 2025 | 2.3 | 66% | Potential first-line approvals |
| 2026 | 2.5 | 66% | Pre-patent expiry growth plateau |
| 2027 | 2.7 | 65% | Patent expiry approaches |
| 2028 | 2.4 | 60% | Post-patent generic entry |
Risks and Opportunities
-
Risks: Patent expiration, market entry of biosimilars, emerging competitors with superior efficacy, and regulatory hurdles in emerging markets.
-
Opportunities: Broader indications (adjuvant, early-stage), combination therapies, expanded CNS efficacy, and growth in emerging markets.
Key Takeaways
- ASHLYNA dominates the ALK-positive NSCLC segment with a 60% market share.
- The global market is projected to grow at a CAGR of about 8–10% until 2027.
- Price per treatment ranges from USD 288,000 in the U.S. to EUR 216,000 in Europe, with prices likely stable until patent expiry.
- Post-2028, generic entry may reduce prices by over 50%, impacting revenue streams.
- Expansion into first-line therapy and additional indications could sustain sales growth.
Frequently Asked Questions
Q1: What factors influence the future pricing of ASHLYNA?
Patent expiration in 2028, generic drug entry, negotiation strength of payers, and competition from new therapies.
Q2: How does the market share comparison look for the main competitors?
Alectinib holds approximately a 60% share of the ALK inhibitor segment, outpacing crizotinib and brigatinib.
Q3: What are the main growth regions for ASHLYNA?
North America and Asia-Pacific, driven by expanding indications and better healthcare infrastructure.
Q4: How might upcoming clinical trials impact demand?
Successful trials for earlier lines or broader indications could expand the patient base, increasing sales.
Q5: What is the expected impact of biosimilars after patent expiry?
Potential price reductions and increased market penetration, reducing overall revenue but possibly expanding patient access.
References
[1] American Cancer Society. (2022). Cancer Statistics. https://cancerstatisticscenter.cancer.org/
More… ↓
