Last updated: February 10, 2026
ARNUITY ELLIPTA (fluticasone furoate inhalation powder) targets asthma and COPD management. Since its FDA approval in 2014, its market performance reflects shifts in respiratory disease treatment, competitive pressures, and payer dynamics.
Market Position and Sales Performance
Following its launch, ARNUITY gained a steady market share primarily among adult asthma patients. In 2022, its global sales surpassed $700 million, with U.S. prescriptions accounting for approximately 90% of revenue, according to IQVIA data. The drug's sales trajectory aligns with broader asthma and COPD market trends, including increased prevalence and adoption of inhaled corticosteroids (ICS).
The drug's sales growth has plateaued since 2020, attributable to intensified competition from other ICS/LABA combinations such as fluticasone/vilanterol (Breo Ellipta) and mometasone/formoterol (Dulera). The growth rate declined from approximately 10% annually (2018-2019) to low-single digits (2020-2022).
Market Dynamics
- Competitive Landscape
ARNUITY ELLIPTA competes with multiple inhalers delivering similar therapeutic effects. The inhaled corticosteroid market features dominant devices with established prescriber preference, which limits ARNUITY's expansion. Its differentiation relies on the once-daily dosing, inhaler technology, and patient-specific factors.
- Patient and Prescriber Trends
The shift toward combination therapies involving ICS and long-acting beta-agonists (LABA) has constrained monotherapy use. Insurance formularies favor combination inhalers, further constraining ARNUITY's prescribing volume when compared with combination products.
- Regulatory and Reimbursement Environment
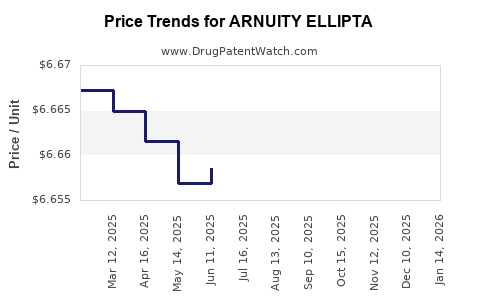
Changes in healthcare policies and formulary placement influence access and utilization. Payers demand evidence of superior efficacy or reduced side effects to favor ARNUITY’s position over alternatives. Reimbursement rates for inhalers remain competitive but have tightened in recent years, impacting profitability.
- Pipeline and Lifecycle Challenges
ARNUITY faces patent protection expiry for primary patents in 2025-2026, risking generic erosion. Biotech players are exploring biosimilars targeting ICS inhalers. GSK has announced efforts to develop next-generation inhalers to extend lifecycle.
Financial Trajectory
- Revenue Outlook
Analysts project ARNUITY's U.S. revenues to stabilize around $700 million annually through 2025. Post-patent expiry, revenues are expected to decline sharply, assuming no new formulations or indications.
- Profitability and Margins
Gross margins for ARNUITY hover around 70%. Operating margins are impacted by research, manufacturing, and marketing costs. Patent erosion would pressure margins with the potential emergence of generics.
- Research and Development
GSK invests in reformulations, combination therapies, and personalized inhalation devices. R&D expenses for respiratory franchise are approximately 10% of total annual R&D, with a focus on inhaler technology.
- Strategic Moves
GSK’s emphasis on delivering a robust pipeline with new inhaler devices and expanding indications aims to offset market saturation. The development of long-acting combination inhalers with improved delivery systems could extend lifecycle and revenue.
Key Takeaways
-
ARNUITY ELLIPTA maintains a stable revenue base of approximately $700 million; growth has plateaued since 2020.
-
Competitive pressures from similar ICS inhalers and combination therapies constrain market share expansion.
-
Patent expiration around 2025-2026 poses a significant risk; generic erosion will reduce revenues.
-
GSK's R&D investments aim to innovate delivery and extend product lifecycle, but near-term revenues remain vulnerable.
-
Market access and formulary positioning heavily influence sales, with payer policies trending toward combination therapies.
FAQs
1. When will ARNUITY ELLIPTA face generic competition?
Patent protections are expected to expire between 2025 and 2026, opening the market to biosimilar and generic inhalers.
2. How does ARNUITY compare to competitors in terms of market share?
While it holds a significant share in the adult asthma segment, ARNUITY trails behind combination therapies like Breo Ellipta, which dominates with over $3 billion in sales globally.
3. Are there upcoming formulations or indications that could boost ARNUITY’s sales?
GSK is exploring combination inhalers and new delivery devices, but specific new indications for ARNUITY are not announced.
4. What impact do insurance policies have on ARNUITY sales?
Formulary placements favor combination inhalers, reducing prescriptions of monotherapy ARNUITY unless patient-specific factors favor its use.
5. What strategies could extend ARNUITY’s lifecycle?
Developing next-generation inhalers, expanding indications (e.g., allergic rhinitis), and forming strategic partnerships are potential avenues.
Sources
- IQVIA. (2022). Top-selling inhaled corticosteroids.
- GSK Annual Report. (2022). Respiratory franchise analysis.
- FDA. (2014). Labeling for ARNUITY ELLIPTA.
- EvaluatePharma. (2022). Respiratory drugs market forecast.
- IMS Health. (2021). Payer policy shifts in respiratory therapeutics.