ALYFTREK Drug Patent Profile
✉ Email this page to a colleague
When do Alyftrek patents expire, and what generic alternatives are available?
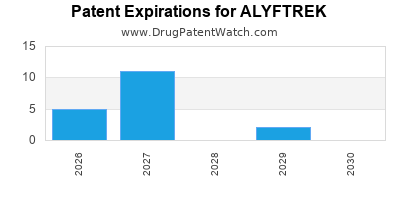
Alyftrek is a drug marketed by Vertex Pharms Inc and is included in one NDA. There are thirty-three patents protecting this drug.
This drug has five hundred and fifty-seven patent family members in fifty-two countries.
The generic ingredient in ALYFTREK is deutivacaftor; tezacaftor; vanzacaftor calcium. One supplier is listed for this compound. Additional details are available on the deutivacaftor; tezacaftor; vanzacaftor calcium profile page.
DrugPatentWatch® Generic Entry Outlook for Alyftrek
Alyftrek will be eligible for patent challenges on December 20, 2028. This date may extended up to six months if a pediatric exclusivity extension is applied to the drug's patents.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be May 17, 2032. This may change due to patent challenges or generic licensing.
There have been nineteen patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for ALYFTREK?
- What are the global sales for ALYFTREK?
- What is Average Wholesale Price for ALYFTREK?
Summary for ALYFTREK
| International Patents: | 557 |
| US Patents: | 33 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for ALYFTREK |
| What excipients (inactive ingredients) are in ALYFTREK? | ALYFTREK excipients list |
| DailyMed Link: | ALYFTREK at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for ALYFTREK
Generic Entry Date for ALYFTREK*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Pharmacology for ALYFTREK
US Patents and Regulatory Information for ALYFTREK
ALYFTREK is protected by thirty-three US patents and one FDA Regulatory Exclusivity.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of ALYFTREK is ⤷ Start Trial.
This potential generic entry date is based on patent 10,047,053.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Vertex Pharms Inc | ALYFTREK | deutivacaftor; tezacaftor; vanzacaftor calcium | TABLET;ORAL | 218730-002 | Dec 20, 2024 | RX | Yes | Yes | 10,206,877 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Vertex Pharms Inc | ALYFTREK | deutivacaftor; tezacaftor; vanzacaftor calcium | TABLET;ORAL | 218730-002 | Dec 20, 2024 | RX | Yes | Yes | 9,974,781 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Vertex Pharms Inc | ALYFTREK | deutivacaftor; tezacaftor; vanzacaftor calcium | TABLET;ORAL | 218730-002 | Dec 20, 2024 | RX | Yes | Yes | 9,512,079 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Vertex Pharms Inc | ALYFTREK | deutivacaftor; tezacaftor; vanzacaftor calcium | TABLET;ORAL | 218730-002 | Dec 20, 2024 | RX | Yes | Yes | 8,410,274 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for ALYFTREK
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Vertex Pharms Inc | ALYFTREK | deutivacaftor; tezacaftor; vanzacaftor calcium | TABLET;ORAL | 218730-001 | Dec 20, 2024 | 8,629,162 | ⤷ Start Trial |
| Vertex Pharms Inc | ALYFTREK | deutivacaftor; tezacaftor; vanzacaftor calcium | TABLET;ORAL | 218730-002 | Dec 20, 2024 | 8,629,162 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for ALYFTREK
When does loss-of-exclusivity occur for ALYFTREK?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 12255711
Estimated Expiration: ⤷ Start Trial
Patent: 17208313
Patent: Deuterated derivatives of ivacftor
Estimated Expiration: ⤷ Start Trial
Patent: 19222862
Patent: Deuterated derivatives of ivacaftor
Estimated Expiration: ⤷ Start Trial
Patent: 21200970
Patent: Deuterated derivatives of ivacaftor
Estimated Expiration: ⤷ Start Trial
Patent: 21203786
Patent: Deuterated derivatives of ivacaftor
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 2013029240
Patent: derivados deuterados do ivacaftor
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 34574
Patent: DERIVES DEUTERES DE L'IVACAFTOR (DEUTERATED DERIVATIVES OF IVACAFTOR)
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 15001358
Patent: Potenciadores del cftr deuterados
Estimated Expiration: ⤷ Start Trial
Croatia
Patent: 0170754
Estimated Expiration: ⤷ Start Trial
Patent: 0192114
Estimated Expiration: ⤷ Start Trial
Cyprus
Patent: 19090
Estimated Expiration: ⤷ Start Trial
Patent: 22498
Estimated Expiration: ⤷ Start Trial
Denmark
Patent: 09986
Estimated Expiration: ⤷ Start Trial
Patent: 35812
Estimated Expiration: ⤷ Start Trial
Eurasian Patent Organization
Patent: 8378
Patent: ДЕЙТЕРИРОВАННЫЕ ПРОИЗВОДНЫЕ ИВАКАФТОРА (DEUTERATED DERIVATIVES OF IVACAFTOR)
Estimated Expiration: ⤷ Start Trial
Patent: 1391615
Patent: ДЕЙТЕРИРОВАННЫЕ ПОТЕНЦИАТОРЫ CFTR
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 09986
Patent: DÉRIVÉS DEUTÉRÉS DE L'IVACAFTOR (DEUTERATED DERIVATIVES OF IVACAFTOR)
Estimated Expiration: ⤷ Start Trial
Patent: 35812
Patent: DÉRIVÉS DEUTÉRÉS D'IVACAFTOR (DEUTERATED DERIVATIVES OF IVACAFTOR)
Estimated Expiration: ⤷ Start Trial
Hong Kong
Patent: 44183
Patent: IVACAFTOR的氘化衍生物 (DEUTERATED DERIVATIVES OF IVACAFTOR)
Estimated Expiration: ⤷ Start Trial
Hungary
Patent: 32771
Estimated Expiration: ⤷ Start Trial
Patent: 47354
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 63455
Estimated Expiration: ⤷ Start Trial
Patent: 14515351
Estimated Expiration: ⤷ Start Trial
Patent: 17078082
Patent: 重水素化されたイバカフトルの誘導体 (DEUTERATED DERIVATIVES OF IVACAFTOR)
Estimated Expiration: ⤷ Start Trial
Lithuania
Patent: 09986
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 8621
Patent: DEUTERATED CFTR POTENTIATORS
Estimated Expiration: ⤷ Start Trial
Patent: 3582
Patent: DEUTERATED CFTR POTENTIATORS
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 9159
Patent: DERIVADOS DEUTERADOS DE IVACAFTOR. (DEUTERATED DERIVATIVES OF IVACAFTOR.)
Estimated Expiration: ⤷ Start Trial
Patent: 13013450
Patent: DERIVADOS DEUTERADOS DE IVACAFTOR. (DEUTERATED DERIVATIVES OF IVACAFTOR.)
Estimated Expiration: ⤷ Start Trial
Montenegro
Patent: 749
Patent: DEUTERISANI DERIVATI IVAKAFTORA (DEUTERATED DERIVATIVES OF IVACAFTOR)
Estimated Expiration: ⤷ Start Trial
Patent: 652
Patent: DEUTERISANI DERIVATI IVAKAFTORA (DEUTERATED DERIVATIVES OF IVACAFTOR)
Estimated Expiration: ⤷ Start Trial
Poland
Patent: 09986
Estimated Expiration: ⤷ Start Trial
Patent: 35812
Estimated Expiration: ⤷ Start Trial
Portugal
Patent: 09986
Estimated Expiration: ⤷ Start Trial
Patent: 35812
Estimated Expiration: ⤷ Start Trial
San Marino
Patent: 01700302
Estimated Expiration: ⤷ Start Trial
Patent: 01900679
Estimated Expiration: ⤷ Start Trial
Serbia
Patent: 096
Patent: DEUTERISANI DERIVATI IVAKAFTORA (DEUTERATED DERIVATIVES OF IVACAFTOR)
Estimated Expiration: ⤷ Start Trial
Patent: 744
Patent: DEUTERISANI DERIVATI IVAKAFTORA (DEUTERATED DERIVATIVES OF IVACAFTOR)
Estimated Expiration: ⤷ Start Trial
Slovenia
Patent: 09986
Estimated Expiration: ⤷ Start Trial
Patent: 35812
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 28465
Estimated Expiration: ⤷ Start Trial
Patent: 58028
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering ALYFTREK around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| San Marino | T201700302 | ⤷ Start Trial | |
| Japan | 2018048207 | ⤷ Start Trial | |
| Hong Kong | 1209318 | -二氟苯 二惡茂- -基 -二羥基 -氟代- -羥基- -甲基丙烷- -基 氫-吲哚- -基 環丙烷甲酰胺的藥劑學組合物和其應用 (PHARMACEUTICAL COMPOSITIONS OF (R)-1-(2,2- DIFLURORBENZO[D][1,3]DIOXOL-5-YL)-N-(1-(2,3-DIHYDROXYPROPYL)-6- FLUORO-2-(1-HYDROXY-2-METHYLPROPAN-2-YL)-1H-INDOL-5-YL) CYCLOPROPANECARBOXAMIDE AND ADMINISTRATION THEREOF (R)-1-(22-[D][13]-5-)-N-(1-(23-)-6--2- (1--2--2-)-1 --5-)) | ⤷ Start Trial |
| New Zealand | 611485 | ⤷ Start Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ALYFTREK
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 3752510 | 122025000060 | Germany | ⤷ Start Trial | PRODUCT NAME: VANZACAFTOR, ODER EIN PHARMAZEUTISCH UNBEDENKLICHES SALZ DAVON, BEVORZUGT EIN CALCIUMSALZ DAVON; REGISTRATION NO/DATE: EU/1/25/1943 20250630 |
| 2826776 | CR 2021 00013 | Denmark | ⤷ Start Trial | PRODUCT NAME: EN KOMBINATION AF (A) (R)-1-(2,2-DIFLUOROBENZO(D)(1,3)DIOXOL-5-YL)-N-(1-(2,3-DIHYDROXYPROPYL)-6-FLUORO-2-(1-HYDROXY-2-METHYLPROPAN-2-YL)-1H-INDOL-5-YL)CYCLOPROPANECARBOXAMID ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF OG (B) ....; REG. NO/DATE: EU/1/18/1306 20181106 |
| 3752510 | PA2025542 | Lithuania | ⤷ Start Trial | PRODUCT NAME: VANZACAFTOR, OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, PREFERABLY ACALCIUM SALT THEREOF; REGISTRATION NO/DATE: EU/1/25/1943 20250630 |
| 1773816 | PA2015028 | Lithuania | ⤷ Start Trial | PRODUCT NAME: IVACAFTORUM; REGISTRATION NO/DATE: EU/1/12/782/001 - EU/1/12/782/002 20120723 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for ALYFTREK
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.