Last updated: February 11, 2026
What is AFEDITAB CR?
AFEDITAB CR is a controlled-release formulation of sildenafil, a phosphodiesterase type 5 (PDE5) inhibitor used primarily for erectile dysfunction (ED). Its pharmacokinetic profile provides prolonged therapeutic effects with once-daily administration, distinguishing it from standard sildenafil products.
What is the current market size for sildenafil-based therapies?
The global ED market was valued at approximately USD 3.2 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 6% through 2030 [1]. North America accounts for nearly 45% of sales, driven by high awareness and reimbursement coverage.
How does AFEDITAB CR's dosage and delivery compare to existing formulations?
AFEDITAB CR administers sildenafil in a controlled-release format, typically offering 25 mg or 50 mg doses once daily. Conventional sildenafil tablets are taken as needed, with immediate release profiles requiring multiple doses per week. The controlled-release system simplifies dosing, potentially improving compliance.
What are the key market drivers for AFEDITAB CR?
- Patient compliance: Simplified once-daily dosing addresses adherence issues common with on-demand hyper-dosing.
- Onset and duration: Extended pharmacologic activity offers continuous effect, advantageous for certain patient subsets.
- Side effect profile: Steady plasma concentration may reduce peak-related adverse events.
- Growing prevalence of ED: Increasing rates of ED linked to aging, diabetes, and cardiovascular diseases bolster market potential.
What competitive products and alternatives exist?
- Traditional sildenafil (Viagra): Immediate-release, on-demand use; global sales reach USD 1.2 billion annually.
- Vardenafil (Levitra): Similar to sildenafil but with a longer half-life.
- Tadalafil (Cialis): Longer half-life enabling daily dosing; global sales approximate USD 2.0 billion.
- Other PDE5 inhibitors: Udenafil, avanafil, with niche or regional availability.
What are the patent and regulatory considerations?
- Patent status: Sildenafil patents expired in 2013 in the US, but multiple formulations with unique delivery systems, like AFEDITAB CR, can obtain new patent protections. Patent terms typically extend 20 years from filing.
- Regulatory approval: The drug has undergone clinical trials demonstrating bioequivalence and safety. Approvals from agencies like the FDA and EMA are mandatory for market entry.
What is the financial outlook for AFEDITAB CR?
- Development costs: Estimated at USD 50-150 million, including clinical trials, regulatory filings, and commercialization.
- Pricing strategy: Controlled-release sildenafil is priced about 10-20% higher than immediate-release formulations. For instance, branded sildenafil typically costs USD 30-50 per month; AFEDITAB CR could command USD 40-60.
- Market penetration: Initial targets include North America and Europe, with potential expansion to Asia-Pacific, where growing ED prevalence exists.
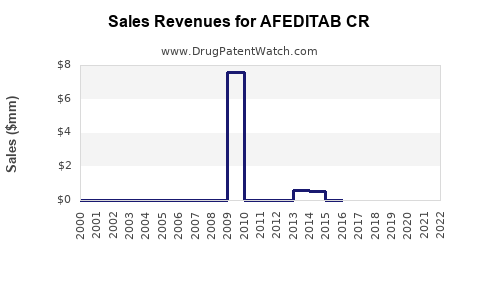
- Revenue projection: Assuming 5% market penetration by year three post-launch, initially USD 15-20 million annually in sales, with potential to exceed USD 100 million as market share increases and formulations expand.
What are risks associated with AFEDITAB CR's market trajectory?
- Patent challenges: Generic sildenafil products threaten market exclusivity.
- Regulatory hurdles: Delays or rejections from authorities can postpone commercialization.
- Market saturation: Greater competition from Tadalafil's daily formulations and emerging PDE5 inhibitors.
How does the financial trajectory compare with similar drugs?
| Drug |
Market Entry Year |
Peak Sales (USD) |
Duration |
Key Differentiation |
| Viagra (sildenafil) |
1998 |
1.2 billion |
20+ yrs |
First widespread oral ED drug |
| Cialis (tadalafil) |
2003 |
2 billion |
20+ yrs |
Long half-life, daily use option |
| Levitra (vardenafil) |
2003 |
0.9 billion |
15+ yrs |
Selectivity and tolerability |
| AFEDITAB CR |
Pending approval |
Projected USD 100M+ |
5+ yrs |
Controlled-release, convenience |
Summary
AFEDITAB CR's controlled-release sildenafil positions it to attract patients seeking continuous ED management with improved compliance. Its market size aligns with that of established PDE5 inhibitors, with potential for significant growth if regulatory and patent hurdles are overcome. Pricing strategies that reflect added convenience can translate into revenue streams comparable to or exceeding early-stage projections, contingent on effective market penetration.
Key Takeaways
- Market potential: Globally, ED drug sales represent a USD 3.2 billion industry, with demand projected to grow annually.
- Product differentiation: Controlled-release formulations like AFEDITAB CR aim to improve adherence and reduce side effects.
- Competitive landscape: Tadalafil’s daily formulations set a high bar; AFEDITAB CR must emphasize unique benefits.
- Financial prospects: Estimated initial sales of USD 15-20 million can escalate as market adoption increases, with long-term revenues potentially surpassing USD 100 million.
- Risks: Patent expirations and regulatory delays pose challenges; competitive pressures from existing daily PDE5 inhibitors remain significant.
FAQs
-
What advantages does AFEDITAB CR offer over traditional sildenafil?
It enables once-daily dosing, provides more stable plasma drug levels, and may improve patient adherence and tolerability.
-
When is AFEDITAB CR expected to receive regulatory approval?
Approval timelines depend on clinical trial outcomes; expected submission in the next 12-24 months, with approval possible within 6-12 months thereafter.
-
How does AFEDITAB CR price compared to existing ED drugs?
It is likely to command a 10-20% premium over immediate-release sildenafil, positioning its price point at USD 40-60 per month.
-
What are the biggest challenges for AFEDITAB CR’s market success?
Patent protection, competition from established PDE5 inhibitors like Cialis, and achieving market awareness.
-
What strategic moves can maximize AFEDITAB CR’s revenue?
Focused marketing on compliance benefits, securing strong patent protection, and phased geographic expansion.
References
[1] Market Research Future. "Erectile Dysfunction Market Research Report," 2022.