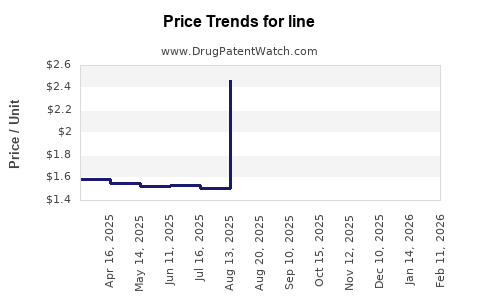
Line - Generic Drug Details
✉ Email this page to a colleague
US Patents and Regulatory Information for line
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Teva Pharms Usa | LINEZOLID | linezolid | TABLET;ORAL | 078061-001 | May 18, 2015 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Pfizer | ZYVOX | linezolid | TABLET;ORAL | 021130-002 | Apr 18, 2000 | DISCN | Yes | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Gate Pharms | LINEZOLID | linezolid | TABLET;ORAL | 091210-001 | Feb 5, 2016 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |