Last updated: April 24, 2026
Where is cefaclor in the drug lifecycle?
Cefaclor is an oral second-generation cephalosporin first approved in the US in 1975 and is now off-patent globally in most major markets, with limited contemporary commercial positioning versus newer oral and long-acting beta-lactam alternatives. Its market has shifted from growth to contraction, driven by (1) generic substitution, (2) formulary tightening for older cephalosporins, and (3) safety and resistance perceptions that favor newer agents with broader spectra, improved dosing convenience, or stronger stewardship positioning.
Key lifecycle milestones:
- US approval: 1975. (FDA Orange Book; approval history as reflected in FDA records for cefaclor) [1]
- Oral-only market presence historically; current access relies on generic supply and country-specific registrations.
- Patents: cefaclor itself is long expired; commercial outcomes are governed by generics, supply stability, and local reimbursement.
What drives cefaclor’s demand?
Cefaclor demand is fundamentally linked to community-acquired bacterial infection (CABSI) prescribing patterns for susceptible organisms, with substitution to other beta-lactams when they offer better value, lower resistance risk, or simpler regimens.
The main demand drivers:
- Clinical fit in older guidance: Cefaclor has historically been used for infections such as respiratory tract infections and otitis media when organisms were susceptible.
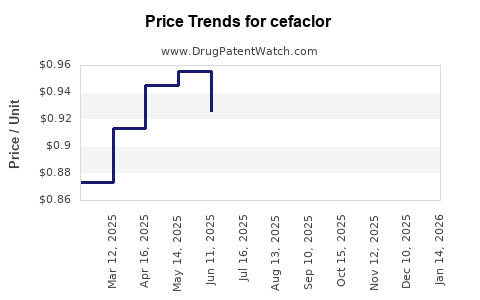
- Generic price pressure: As with other older antibiotics, cefaclor is subject to aggressive price competition among manufacturers.
- Formulary and stewardship constraints: Stewardship programs prefer narrower-spectrum agents where appropriate and newer options where evidence and local resistance support them.
- Switching to preferred comparators: In many formularies, other oral cephalosporins (and non-cephalosporin beta-lactams) capture share when they are stocked as first-line or are cheaper net-of contracts.
What is the competitive landscape?
Cefaclor competes primarily within:
- Oral second-generation cephalosporins (generic and branded legacy variants depending on geography)
- Other oral beta-lactams used for similar indications (penicillins, amoxicillin-clavulanate, and other cephalosporins where available)
- Non-beta-lactams where resistance patterns support them or where guidelines prioritize them
Competition is typically resolved through:
- Net pricing and pharmacy contracts
- Per-prescription preference at the primary care level
- Availability and stability of generic supply
How does resistance and safety perception affect market dynamics?
Cefaclor’s market has been shaped by antimicrobial stewardship trends and evolving susceptibility patterns for common community pathogens. Practical effects include:
- Reduced off-guideline use when clinicians perceive diminished expected benefit.
- Increased reliance on local antibiograms that may not support cefaclor in many regions for common empiric choices.
- Safety-driven substitution to alternatives when prescribers prefer agents with lower adverse-event perception in local experience.
Cefaclor’s safety and adverse reaction profile is documented in labeling and drug reference sources, which can influence clinician preference and payer decisions over time. Labelled adverse effects include hypersensitivity reactions and gastrointestinal effects, with the typical cephalosporin class considerations. (Drug monographs and labeling summaries) [2], [3]
What does the historical and current formulary reality suggest?
In practice, cefaclor functions as a low-cost oral generic rather than a growth product. Demand tends to be:
- Fragmented by country and by local antibiotic guideline alignment
- Concentrated among patients and prescribers where cefaclor is already established or where contracts keep it low cost relative to alternatives
- Suppressed in regions where other generics are preferentially tendered
Because cefaclor is older and off-patent, the financial trajectory largely reflects:
- Generic pricing declines
- Margin compression across manufacturers
- Supply chain volatility effects on short-term availability and wholesaler purchasing decisions
What is cefaclor’s financial trajectory profile?
Cefaclor’s financial trajectory is best characterized as mature-to-declining:
- Earlier era: Brand and initial generics supported a higher market value.
- Current era: Value declines as generic pricing falls and prescription volume shifts toward alternative agents with better guideline fit or pricing.
The economic model of off-patent antibiotics generally produces:
- Revenue growth only through unit volume expansion, which is hard for older molecules once guidelines move on.
- Revenue decline as price per unit falls and as prescribing shifts to other agents.
- Profit concentration among suppliers with cost advantages and stable manufacturing capacity.
What do regulatory and market-access signals imply for revenue?
Two regulatory signals matter for any financial trajectory call:
1) Continued US market eligibility
Cefaclor remains listed in core US regulatory databases for approval history and generic status, indicating ongoing regulatory presence, even if commercial scale is reduced. (FDA Orange Book listing and historical record) [1]
2) Legacy labeling constraints
Labeling content, including indications and safety language, restricts use to contexts where prescribers still find it clinically aligned. Labeling is documented in standard pharmacology and drug reference sources. [2], [3]
How does generic competition shape margins and profitability?
Cefaclor’s profitability is typically driven by generic dynamics:
- Price erosion from multiple approved manufacturers
- Contracting pressure from large wholesalers and pharmacy chains
- Batch availability and manufacturing cost control determining who sustains market share during tender rounds
In mature antibiotic generics, profit often shifts to:
- Lowest-cost producers
- Producers with stronger scale and lower rejection rates
- Suppliers positioned in markets with stable contracting
Market scenario table: likely financial outcomes
The following table frames financial trajectory by core market variable, consistent with mature generic antibiotic economics:
| Variable |
Typical direction for cefaclor |
Financial impact |
| Net price per unit |
Down vs historical brand era |
Revenue decline, margin compression |
| Unit volume |
Flat to down where guideline substitution occurs |
Revenue stability only if price holds |
| Formulary preference |
Lower over time as newer agents gain share |
Reduced steady-state demand |
| Generic supply |
Competitive; occasional disruptions possible |
Short-term volatility, longer-term margin pressure |
| Payer contracting |
Stronger for cheap generics |
Winners gain share via contracts, losers exit or shrink |
What are the key commercial inflection points to watch?
For cefaclor, the practical “inflection points” are not patent events but policy and market structure events:
- Formulary changes at national or insurer level that drop older cephalosporins in favor of contracted alternatives
- Local antibiogram shifts that move susceptibility patterns away from empiric cefaclor use
- Generic supply consolidation where only a small number of manufacturers remain active in certain markets
- Regulatory actions affecting specific product lots or manufacturers, which can temporarily shift demand to other generics
Where does cefaclor sit versus newer antibiotic classes?
Compared with newer oral agents and newer cephalosporin-like alternatives:
- Cefaclor has lower perceived differentiation
- It competes primarily on price rather than on dosing convenience or novel spectrum
- Prescribing is more likely to follow legacy habits and local tender economics than competitive clinical advantage
This typically results in a long runway of sales but limited upside.
Key Takeaways
- Cefaclor’s market is mature and largely off-patent, with commercial performance driven by generic pricing, contracting, and guideline fit rather than innovation.
- Demand is stable only where local formularies and susceptibility patterns support its use; otherwise, prescription shifts to alternative oral beta-lactams.
- Financial trajectory is revenue pressure and margin compression, with supply and contracting determining which manufacturers capture remaining share.
- Regulatory persistence in listings supports continued availability, but legacy labeling and class-level safety considerations constrain expansion.
FAQs
1) Is cefaclor currently patent-protected in major markets?
Cefaclor is an older molecule; commercial presence is dominated by generic dynamics with long-expired patent coverage in practice, as reflected by its legacy regulatory status. [1]
2) What indications historically drove cefaclor use?
Cefaclor has been used for bacterial infections where second-generation cephalosporins were clinically appropriate, with specific indications defined in labeling and drug references. [2], [3]
3) Does cefaclor face significant competitive substitution from other oral antibiotics?
Yes. Prescribing shifts toward other contracted oral beta-lactams and alternatives when guidelines and resistance patterns favor them. (Class and labeling context from drug references) [2], [3]
4) What determines cefaclor’s profitability most today?
Net pricing and contracting, plus manufacturing cost efficiency and supply stability across generic manufacturers. (Generic antibiotic market structure; regulatory continuity) [1]
5) Where are the main risks to cefaclor’s remaining demand?
Formulary down-trading of older cephalosporins, local antibiogram changes, and stewardship-driven narrowing of empiric choices. (Safety and usage constraints reflected in labeling and monographs) [2], [3]
References
[1] U.S. Food and Drug Administration (FDA). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. FDA database entry for cefaclor. https://www.accessdata.fda.gov/scripts/cder/daf/ (accessed via Orange Book records).
[2] DailyMed. Cefaclor (for oral use) prescribing information/labeling. U.S. National Library of Medicine. https://dailymed.nlm.nih.gov/
[3] Lexicomp / Drug reference monographs (as reflected in standard drug information summaries). Cefaclor: safety and adverse reactions; mechanism and clinical use notes.