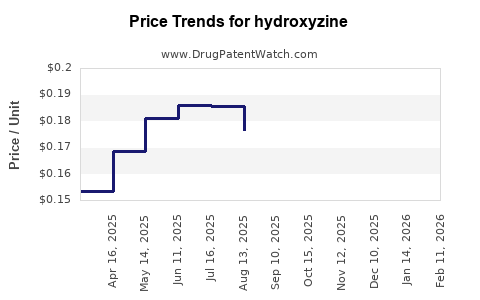
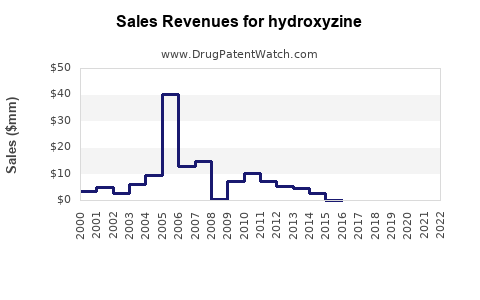
Hydroxyzine - Generic Drug Details
✉ Email this page to a colleague
Recent Clinical Trials for hydroxyzine
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| MATClinics | PHASE4 |
| Friends Research Institute, Inc. | PHASE4 |
| Sultan Qaboos University | Phase 4 |
US Patents and Regulatory Information for hydroxyzine
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Usl Pharma | HYDROXYZINE HYDROCHLORIDE | hydroxyzine hydrochloride | TABLET;ORAL | 089121-001 | Mar 20, 1986 | DISCN | No | No | ⤷ Get Started Free | ⤷ Get Started Free | ⤷ Get Started Free | ||||
| Watson Labs | HYDROXYZINE HYDROCHLORIDE | hydroxyzine hydrochloride | TABLET;ORAL | 081151-001 | Mar 18, 1994 | DISCN | No | No | ⤷ Get Started Free | ⤷ Get Started Free | ⤷ Get Started Free | ||||
| Solopak | HYDROXYZINE HYDROCHLORIDE | hydroxyzine hydrochloride | INJECTABLE;INJECTION | 087593-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Get Started Free | ⤷ Get Started Free | ⤷ Get Started Free | ||||
| Apozeal Pharms | HYDROXYZINE HYDROCHLORIDE | hydroxyzine hydrochloride | SYRUP;ORAL | 210634-001 | Feb 26, 2019 | AA | RX | No | No | ⤷ Get Started Free | ⤷ Get Started Free | ⤷ Get Started Free | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |