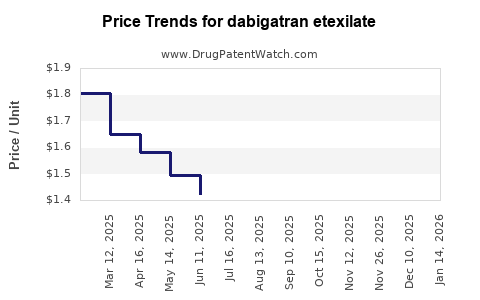
Dabigatran etexilate - Generic Drug Details
✉ Email this page to a colleague
Generic filers with tentative approvals for DABIGATRAN ETEXILATE
| Applicant | Application No. | Strength | Dosage Form |
| ⤷ Try a Trial | ⤷ Try a Trial | 150MG | CAPSULE;ORAL |
| ⤷ Try a Trial | ⤷ Try a Trial | 110MG | CAPSULE;ORAL |
| ⤷ Try a Trial | ⤷ Try a Trial | EQ 150MG BASE | CAPSULE;ORAL |
The 'tentative' approval signifies that the product meets all FDA standards for marketing, and, but for the patents / regulatory protections, it would approved.
US Patents and Regulatory Information for dabigatran etexilate
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim | PRADAXA | dabigatran etexilate mesylate | PELLETS;ORAL | 214358-003 | Jun 21, 2021 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Alkem Labs Ltd | DABIGATRAN ETEXILATE MESYLATE | dabigatran etexilate mesylate | CAPSULE;ORAL | 208040-001 | Mar 11, 2020 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Boehringer Ingelheim | PRADAXA | dabigatran etexilate mesylate | CAPSULE;ORAL | 022512-003 | Nov 20, 2015 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |