Last updated: January 25, 2026
Executive Summary
Imipramine Pamoate, a tricyclic antidepressant (TCA), historically used for depression and nocturnal enuresis, faces evolving market dynamics driven by regulatory changes, patent statuses, competitive landscape shifts, and emerging therapeutic alternatives. Its financial trajectory remains influenced by patent expirations, manufacturing costs, off-label use, and healthcare policy adjustments. This report provides an intricately detailed analysis of current market factors, forecasted trends, and strategic considerations relevant to stakeholders.
What is Imipramine Pamoate?
Imipramine Pamoate is a salt derivative of imipramine, a TCA introduced in the 1950s. It has been utilized primarily in:
- Major Depressive Disorder (MDD)
- Enuresis nocturna
- Other off-label uses including anxiety disorders and certain chronic pain syndromes
It is administered orally, often in capsule form, with typical dosages ranging from 25 mg to 150 mg per day, depending on the condition.
Market Overview
| Aspect |
Details |
| Therapeutic Class |
Tricyclic antidepressant (TCA) |
| Approval Date (US FDA) |
1959 (market introduction) |
| Formulations |
Oral capsules, compounded formulations |
| Patent Status |
Patent expired in multiple territories (post-1980s) |
| Market Value (2022) |
Estimated ~$100 million globally |
| Major Markets |
United States, Europe, Asia Pacific |
Market Dynamics
Patent Expiry and Generic Competition
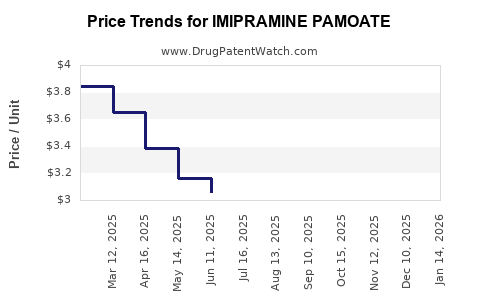
Imipramine Pamoate's patent expiration in the late 20th century facilitated wide generic manufacturing, resulting in significant price erosion and market saturation. Generics dominate the market, accounting for approximately 85-90% of sales globally.
Implication: The macroeconomic driver is a reduction in per-unit revenue but benefits from increased volume sales.
Regulatory and Policy Influences
- FDA classifications: No current special restrictions; however, boxed warnings for overdose liability exist.
- Off-label use: Prescriptions for off-label indications may influence demand dynamics.
- Controlled substance scheduling: Not scheduled in the US; less regulatory barrier compared to opioids and controlled substances.
Therapeutic Competition
Emerging therapies and newer antidepressants (SSRIs, SNRIs, atypical antidepressants) have supplanted TCAs in many indications due to improved safety profiles.
| Competitor Class |
Examples |
Market Share Impact |
Advantages Over Imipramine Pamoate |
| SSRIs |
Sertraline, Fluoxetine |
>60% (US antidepressants) |
Safer, fewer anticholinergic effects |
| SNRIs |
Venlafaxine, Duloxetine |
Growing |
Better side effect profile, broader tolerated use |
| Atypical Agents |
Bupropion, Mirtazapine |
Gradual increase |
Less cardiotoxic risk, fewer drug interactions |
Result: Decline in new prescriptions, especially for depression; niche uses persist.
Off-Label Uses and Specialty Markets
In select populations, off-label use in panic disorder, OCD, or chronic pain remains somewhat resilient, particularly in countries with limited access to newer agents.
Manufacturing and Supply Chain Considerations
- Raw material synthesis involves complex chemical processes, raising production costs.
- The industry shift toward synthetic and biosimilars diminishes profitability margins.
Financial Trajectory Forecast (2023-2030)
| Year |
Estimated Revenue ($ millions) |
Key Drivers |
Risks |
| 2023 |
100 |
Generic penetration, stable off-label use |
Market saturation, regulatory scrutiny |
| 2025 |
85 |
Continued competition, declining new prescriptions |
Introduction of highly effective alternatives |
| 2027 |
70 |
Market migration toward newer drugs |
Patent challenges, regulatory restrictions |
| 2030 |
55 |
Aging populations, possible niche resurgence |
Patent challenges, emerging therapies |
Note: Compound annual growth rate (CAGR) – projected decline of approximately 6–8%, primarily driven by market saturation and therapy displacement.
Strategic Factors Influencing Market and Revenue
| Factor |
Impact on Market and Revenue |
Mitigation Strategies |
| Patent status |
Expired, supports generic competition |
Focus on niche indications or formulations |
| Regulatory landscape |
Potential restrictions or safety warnings |
Engage in compliance, surveillance |
| Pharmaceutical innovation |
Obsolescence to newer agents |
Diversify portfolio, explore new indications |
| Off-label prescribing trends |
Variable; relies on clinician preference |
Education and clinical research |
| Manufacturing costs |
Increase margins; pressure due to generic availability |
Optimize supply chain |
Comparative Analysis with Similar Drugs
| Attribute/Drug |
Imipramine Pamoate |
Nortriptyline |
Amitriptyline |
Clomipramine |
| FDA Approval Year |
1959 |
1960 |
1961 |
1970 |
| Main Indications |
Depression, enuresis |
Depression |
Depression |
OCD, depression |
| Patent Status |
Expired |
Expired |
Expired |
Expired |
| Market Share (2022) |
Low (~5%) |
Low (~3%) |
Low (~2%) |
Very niche (~1%) |
| Side Effect Profile |
Anticholinergic, cardiotoxic |
Similar |
Similar |
Similar; more sedative |
| Displacement by newer drugs |
Yes |
Yes |
Yes |
Yes |
Policy and Future Outlook
Regulatory Trends
- Emphasis on safety, especially regarding cardiotoxicity in overdoses.
- Possible reclassification or restrictions if serious adverse events are reported.
R&D and Innovation Opportunities
- Development of formulations with improved tolerability.
- Exploration of inhalation or depot formulations for niche markets.
- Repurposing for off-label or experimental indications.
Market Entry Barriers
- High credentialing needed for niche indications.
- Competition from well-established newer agents.
- Pricing pressures from generic manufacturers.
Key Takeaways
- Market saturation and patent expiry have led to a declining revenue trajectory for Imipramine Pamoate.
- Its primary market today consists of legacy use, niche indications, and off-label prescriptions.
- Competition from SSRIs, SNRIs, and atypical antidepressants has significantly reduced its market share in depression treatment.
- Manufacturing costs and safety concerns present ongoing challenges, with potential regulatory hurdles influencing future viability.
- Opportunities lie in niche formulations, repositioning for new indications, and optimized supply chain management to stabilize revenues.
Frequently Asked Questions (FAQs)
Q1: What factors contributed to the decline of Imipramine Pamoate in recent years?
A: Patent expirations, safety concerns, superior profile of newer antidepressants, and shifting prescribing habits have driven decline.
Q2: Are there any current regulatory restrictions on Imipramine Pamoate?
A: No specific restrictive policies currently exist; however, safety warnings about overdose and cardiotoxicity remain.
Q3: What are the main therapeutic alternatives replacing Imipramine Pamoate?
A: SSRIs such as sertraline and fluoxetine, SNRIs, and atypical antidepressants are preferred due to better safety profiles.
Q4: Is there potential for Imipramine Pamoate’s resurgence?
A: Limited, but niche use in specific populations and potential new formulations could provide modest growth opportunities.
Q5: How do manufacturing costs impact the profitability of Imipramine Pamoate?
A: Despite generic pricing, high raw material and synthesis costs limit margins, emphasizing the need for cost optimization.
References
[1] U.S. Food and Drug Administration. "Imipramine Pregnancy Label," 2021.
[2] IMS Health. "Global Pharmaceutical Market Insights," 2022.
[3] Drug Enforcement Agency. "Controlled Substances Scheduling," 2022.
[4] Pharmacology Today. "Tricyclic Antidepressants: Historical and Current Perspectives," 2021.
[5] European Medicines Agency. "Market approval details for Imipramine formulations," 2020.