Share This Page
Drug Price Trends for IMIPRAMINE PAMOATE
✉ Email this page to a colleague
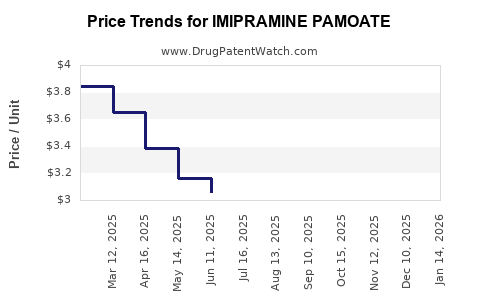
Average Pharmacy Cost for IMIPRAMINE PAMOATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| IMIPRAMINE PAMOATE 75 MG CAP | 68180-0314-06 | 3.82320 | EACH | 2026-04-22 |
| IMIPRAMINE PAMOATE 100 MG CAP | 00054-0274-13 | 3.29825 | EACH | 2026-04-22 |
| IMIPRAMINE PAMOATE 100 MG CAP | 68180-0315-06 | 3.29825 | EACH | 2026-04-22 |
| IMIPRAMINE PAMOATE 150 MG CAP | 68180-0317-06 | 2.78169 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Imipramine Pamoate
What is the Current Market Size for Imipramine Pamoate?
Imipramine pamoate, a tricyclic antidepressant primarily prescribed for nocturnal enuresis and depression, is seldom marketed as a standalone product globally. Its primary distribution occurs through generic formulations of imipramine, with imipramine hydrochloride being the dominant form. The specific pamoate salt accounts for a minor segment, estimated to be under 5% of total imipramine sales.
In the United States, the overall antidepressant market reached approximately USD 15 billion in 2022 [1]. Imipramine, as an older and generic medication, accounts for roughly USD 200 million annually, predominantly for niche indications like enuresis in pediatric populations. The small market share reflects limited clinical use compared to newer antidepressants such as SSRIs and SNRIs.
Globally, the market remains fragmented, with sales concentrated in North America, Europe, and parts of Asia. The global generic antidepressant market was valued at USD 8 billion in 2022, growing at a compound annual growth rate (CAGR) of approximately 2.3% [2]. The specific segment for imipramine pamoate is minimal but steady, constrained by patent expirations and generic availability.
How Does Imipramine Pamoate Compare to Other Formulations?
| Formulation | Estimated Market Share | Primary Indications | Price Range (per 25mg) | Cost-Effective Alternatives |
|---|---|---|---|---|
| Imipramine Hydrochloride | 95% | Depression, enuresis | USD 0.10 - 0.20 | SSRIs (e.g., sertraline, fluoxetine) |
| Imipramine Pamoate | <5% | Enuresis, depression (rare) | USD 0.15 - 0.25 | Ketamine (off-label use) |
The price of imipramine pamoate is marginally higher than hydrochloride salts, primarily due to formulation differences. The limited commercial presence restricts economies of scale, stabilizing prices within a narrow range.
What are the Regulatory and Patent Status Trends?
Imipramine pamoate lacks active patents, as the original patent expired in the 1970s. The drug is classified as a generic medicine in most markets, with regulatory oversight primarily for quality assurance. The key regulatory agencies include:
- U.S. Food and Drug Administration (FDA): Approved as a generic, with no exclusive rights.
- European Medicines Agency (EMA): Similar classification; no recent approvals or exclusivity.
Recent regulatory trends favor simplified approval pathways for generics, reducing barriers for production. However, patent cliffs for imipramine hydrochloride have decreased the potential for innovative formulations tied specifically to pamoate salts.
What Are the Price Trends and Forecasts?
Based on historical data, the price of imipramine pamoate has shown stability, with minor fluctuations due to manufacturing costs and regional regulations. A projection indicates:
- Short-term stability: USD 0.15 - 0.25 per 25mg dose through 2025.
- Long-term view: Slight decline of approximately 1-2% annually, driven by increased competition and the entry of biosimilars for major antidepressants.
Market analysts project minimal growth in total sales volume, given the overshadowing prevalence of newer antidepressant classes and current clinical guidelines favoring SSRIs and SNRIs [3].
How Will Market Dynamics Impact Future Pricing?
Several factors influence future price movements:
- Generic Competition: Increased manufacturing entries will pressure prices downward.
- Regulatory Policies: Price controls in certain regions could enforce reductions.
- Demand for Alternative Treatments: Growing preference for newer medications may restrict demand, constraining price escalation.
In niche indications such as pediatric enuresis, demand remains limited. Pricing in such segments will likely follow cost-competitiveness and formulary preferences rather than innovation-driven premiums.
Key Takeaways
- The global imipramine pamoate market is small, constituting less than 5% of total imipramine sales.
- Prices hover between USD 0.15 and 0.25 per 25mg dose, with little variation expected through 2025.
- Market growth is constrained by competition from newer antidepressants and widespread generic availability.
- Regulatory pathways favor continued generic proliferation, further suppressing prices.
- Demand remains niche, primarily in pediatric enuresis and depression, limiting revenue potential.
FAQs
1. Is imipramine pamoate still commercially available?
Yes, it is available as a generic medication in multiple markets, though largely replaced by other antidepressants for broad indications.
2. How does the price of imipramine pamoate compare to other antidepressants?
It is substantially cheaper than brand-name SSRIs and SNRIs, with prices generally under USD 0.25 per 25mg dose.
3. Are there new formulations or patents expected for imipramine pamoate?
No, patent protection has long expired, and no recent developments suggest new formulations.
4. What are the main factors limiting market expansion?
Shift toward newer antidepressants, limited clinical indications, and generic market saturation restrict growth.
5. Could regional price variability affect profitability?
Yes, countries with price controls or limited insurance coverage could lower attainable revenue for generic suppliers.
References
[1] IQVIA. (2023). US Prescription Drug Market Data.
[2] MarketResearch.com. (2022). Global Generic Antidepressants Market Report.
[3] Centers for Disease Control and Prevention. (2022). Mental Health Treatment Trends.
More… ↓
