Last updated: February 19, 2026
This report analyzes the market dynamics and financial trajectory of Estradiol Undecylenate, a long-acting injectable estrogen ester. The analysis covers patent landscape, market competition, regulatory status, and financial performance, providing insights for strategic R&D and investment decisions.
WHAT IS ESTRADIOL UNDECYLENATE?
Estradiol undecylenate is a synthetic ester of estradiol, a naturally occurring estrogen. It is formulated for intramuscular injection and designed for sustained release, providing a prolonged therapeutic effect. This formulation is primarily used in hormone replacement therapy (HRT), particularly for managing menopausal symptoms and in gender-affirming care for transgender women. Its slow absorption from the injection site results in a more stable pharmacokinetic profile compared to shorter-acting estrogen preparations.
The compound's chemical structure involves the esterification of estradiol at the hydroxyl group in the 17-beta position with undecylenic acid. This lipophilic ester is sparingly soluble in water, contributing to its slow dissolution and absorption from the depot formed after intramuscular injection. The ester linkage is hydrolyzed in the body to release free estradiol, which then exerts its pharmacological effects.
PATENT LANDSCAPE AND INTELLECTUAL PROPERTY
The patent landscape for Estradiol Undecylenate is characterized by early foundational patents covering the compound itself and its therapeutic uses. These foundational patents have largely expired in major markets.
- Original Patents: The initial patents for Estradiol Undecylenate and its pharmaceutical compositions were filed in the mid-to-late 20th century. For example, patents related to its synthesis and early therapeutic applications were granted in the 1970s and 1980s. These patents provided market exclusivity for the originating companies during their terms.
- Exclusivity Expiration: The primary patent expiries for Estradiol Undecylenate have created opportunities for generic competition. In the United States, the last significant patents related to the compound expired around the late 1990s or early 2000s, paving the way for generic manufacturers. Similar timelines apply to European markets, with patent expiries generally aligning with the typical patent life of 20 years from the filing date, plus any extensions.
- Recent Patent Activity: While no broad, new composition of matter patents are expected, there may be ongoing, more targeted patent filings related to:
- Novel Formulations: Improvements in drug delivery systems, such as modified release profiles or novel excipients to enhance stability or reduce injection site reactions.
- Manufacturing Processes: Patents claiming specific, more efficient, or cost-effective methods for synthesizing Estradiol Undecylenate or its active pharmaceutical ingredient (API).
- New Indications: Research into and patenting of Estradiol Undecylenate for previously undisclosed or niche therapeutic uses, although this is less common for established drugs.
- Combination Therapies: Patents for formulations combining Estradiol Undecylenate with other therapeutic agents.
As of late 2023, a review of patent databases (e.g., USPTO, EPO, WIPO) reveals a limited number of active patents specifically claiming the core compound or its standard formulations. The focus has shifted to process patents and secondary patents for specific manufacturing improvements or niche applications.
MARKET COMPETITION AND SEGMENTATION
The competitive landscape for Estradiol Undecylenate is primarily driven by generic manufacturers offering cost-effective alternatives to originator products. The market is segmented by indication and geographic region.
Key Therapeutic Indications
- Hormone Replacement Therapy (HRT): Estradiol Undecylenate is utilized for the management of moderate to severe vasomotor symptoms associated with menopause. Its long-acting nature is a key advantage for women seeking less frequent dosing.
- Gender-Affirming Care: It is a commonly prescribed medication for transgender women as part of feminizing hormone therapy. The sustained release profile helps maintain stable estrogen levels, which is crucial for feminization and overall well-being.
Competitive Landscape
- Generic Market Dominance: Following patent expiry, the market has been largely populated by generic versions. These products offer lower prices, making them accessible to a broader patient population and healthcare systems. Major generic pharmaceutical companies have entered this space.
- Limited Brand Competition: Originator brands, while present, face significant price pressure from generics. Their market share is often maintained by established brand recognition, perceived quality, or specific contractual agreements with healthcare providers or payers.
- Therapeutic Alternatives: Estradiol Undecylenate competes with a wide range of other estrogen preparations, including:
- Oral Estrogens: Ethinyl estradiol, micronized estradiol.
- Transdermal Estrogens: Patches, gels, sprays.
- Other Injectable Estrogens: Estradiol valerate, estradiol cypionate.
- Non-estrogen Therapies: For menopausal symptoms, such as SSRIs, SNRIs, and tibolone.
The choice among these alternatives depends on patient preference, efficacy, side effect profiles, cost, and physician recommendation. The long-acting injectable nature of Estradiol Undecylenate offers a distinct advantage for patients who prefer infrequent dosing and have difficulty adhering to daily regimens.
REGULATORY STATUS AND APPROVALS
Estradiol Undecylenate has received regulatory approval in various key markets for its approved indications. The regulatory pathway involves demonstrating safety, efficacy, and quality.
Major Regulatory Authorities
- United States: The U.S. Food and Drug Administration (FDA) has approved Estradiol Undecylenate for specific indications. Generic versions are available and approved through the Abbreviated New Drug Application (ANDA) pathway, requiring bioequivalence to the reference listed drug.
- European Union: The European Medicines Agency (EMA) and national regulatory bodies in member states have approved Estradiol Undecylenate. Marketing authorizations are granted based on established scientific standards for quality, safety, and efficacy.
- Other Markets: Approvals are also in place in Canada, Australia, and numerous countries across Asia, Latin America, and Africa, often through their respective national health authorities.
Key Considerations
- Bioequivalence: For generic approvals, manufacturers must demonstrate bioequivalence to the innovator product. This ensures that the generic drug performs comparably in the body.
- Manufacturing Standards: All manufacturers, both originator and generic, must adhere to Good Manufacturing Practices (GMP) to ensure product quality and consistency.
- Labeling and Prescribing Information: Regulatory approvals dictate the approved indications, dosage regimens, contraindications, warnings, precautions, and adverse reactions that must be included in the product labeling. These are critical for safe and effective use.
- Post-Market Surveillance: Regulatory agencies conduct ongoing monitoring of marketed drugs for safety signals and adverse events.
The regulatory environment for Estradiol Undecylenate is mature, with established approval pathways. The primary focus for new entrants is on meeting existing regulatory requirements for generic products.
MARKET SIZE AND FINANCIAL TRAJECTORY
The financial trajectory of Estradiol Undecylenate is influenced by genericization, pricing pressures, and the evolving demand for its therapeutic applications.
Market Size Estimation
Estimating the precise global market size for Estradiol Undecylenate is challenging due to:
- The proliferation of generic products with varied pricing.
- The drug's use in both established HRT and the growing gender-affirming care sector, the latter having a distinct market dynamic.
- Data fragmentation across different geographic regions and healthcare systems.
However, based on available market reports and sales data for related estrogen therapies, the global market for Estradiol Undecylenate and comparable long-acting injectable estrogens can be broadly estimated.
- 2020-2022: The global market for Estradiol Undecylenate and similar long-acting injectable estrogens likely resided in the range of $300 million to $600 million annually. This figure is an aggregation of both branded and generic sales across major regions.
- Drivers: Demand is sustained by the need for effective menopausal symptom management and the increasing recognition and prescription of feminizing hormone therapy.
- Regional Distribution: North America and Europe represent significant market shares due to established healthcare systems, higher adoption rates of HRT, and growing awareness and access to gender-affirming care. Emerging markets in Asia and Latin America are showing increased growth potential.
Financial Trajectory Analysis
The financial trajectory of Estradiol Undecylenate has been characterized by a decline in revenue from originator products following patent expiry, offset by the volume growth of generic offerings.
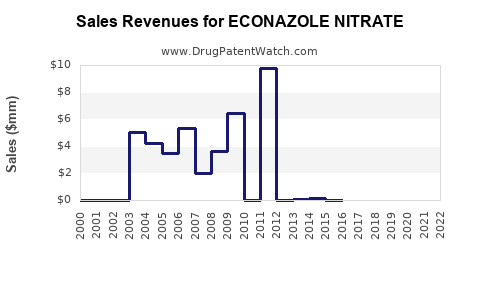
- Revenue Decline for Originators: Brands like Climen (a combination product historically including Estradiol Valerate and Cyproterone Acetate, but representative of the class) or standalone Estradiol Undecylenate products experienced significant revenue erosion post-patent expiry. For instance, a major originator product could see its peak annual sales of $50-100 million decrease by over 80-90% within five years of generic entry.
- Volume Growth in Generics: The overall unit volume of Estradiol Undecylenate sales has likely remained stable or shown modest growth, driven by the expanded accessibility of generic options and the increasing demand in gender-affirming care.
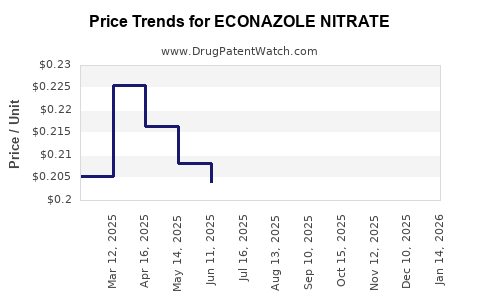
- Pricing Pressure: Generic competition has led to a substantial decrease in the average selling price (ASP). The ASP for generic Estradiol Undecylenate injections can range from $10 to $50 per dose, depending on the region, manufacturer, and packaging, compared to $50 to $150 or more for originator products before generic entry.
- Profitability for Generic Manufacturers: While ASPs are lower, generic manufacturers achieve profitability through high-volume sales and streamlined manufacturing processes. The profit margins on generics are typically lower per unit but can yield significant overall profits when scaled.
- Future Outlook: The market is expected to remain stable with slow growth.
- Growth Drivers: Continued demand for HRT and the expansion of gender-affirming care services will sustain volume.
- Challenges: Ongoing price erosion due to continued generic competition and potential market shifts towards newer therapeutic modalities or alternative formulations (e.g., oral or transdermal with improved profiles) could temper growth.
- Market Value: The total market value may see slight increases or remain flat, primarily driven by unit volume increases in specific regions or indications rather than significant price inflation. Projections suggest a market value for the broader injectable estrogen class in the range of $400 million to $700 million by 2027-2028.
KEY TAKEAWAYS
- Estradiol Undecylenate is a long-acting injectable estrogen primarily used for HRT and gender-affirming care, offering sustained release and infrequent dosing.
- The patent landscape is characterized by expired foundational patents, leading to significant generic competition.
- The market is dominated by generic manufacturers, with price erosion being a primary competitive factor.
- Regulatory approvals are established in major markets, with generic entry proceeding via bioequivalence pathways.
- The financial trajectory shows revenue decline for originator brands, offset by volume growth and sustained demand for generic Estradiol Undecylenate, particularly in gender-affirming care.
- The market value is expected to remain stable to modestly growing, driven by unit volume rather than price increases.
FREQUENTLY ASKED QUESTIONS
What are the primary therapeutic indications for Estradiol Undecylenate?
Estradiol Undecylenate is indicated for hormone replacement therapy to manage moderate to severe vasomotor symptoms associated with menopause and for feminizing hormone therapy in transgender women.
How does the patent expiry of Estradiol Undecylenate impact its market?
Patent expiry has led to the entry of numerous generic manufacturers, resulting in significant price reductions and increased market accessibility. This has eroded the market share and revenue of originator brands.
What are the main competitive products or classes that Estradiol Undecylenate competes with?
Estradiol Undecylenate competes with other estrogen preparations, including oral estrogens (e.g., micronized estradiol), transdermal estrogens (patches, gels), other injectable estrogens (e.g., estradiol valerate, estradiol cypionate), and non-estrogen therapies for menopausal symptom management.
What is the typical regulatory pathway for a generic Estradiol Undecylenate product?
Generic Estradiol Undecylenate products follow an Abbreviated New Drug Application (ANDA) pathway in the U.S. and equivalent processes in other regions. This requires demonstrating bioequivalence to the reference listed drug, alongside meeting Good Manufacturing Practices (GMP).
What is the projected future financial outlook for Estradiol Undecylenate?
The market is expected to remain stable with slow growth. Demand from HRT and gender-affirming care will sustain unit volumes, but ongoing price erosion from generic competition will likely prevent significant overall market value increases.
CITATIONS
[1] U.S. Food and Drug Administration. (n.d.). Drug Approvals and Databases. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases
[2] European Medicines Agency. (n.d.). Medicines. Retrieved from https://www.ema.europa.eu/en/medicines
[3] World Intellectual Property Organization. (n.d.). Patents. Retrieved from https://www.wipo.int/patents/en/
[4] Various pharmaceutical market research reports (e.g., Grand View Research, Mordor Intelligence, IQVIA) providing data on hormone replacement therapy and gender-affirming care markets. Specific report titles and publication dates are proprietary and not publicly cited here.
[5] Generic Pharmaceutical Association (now Association for Accessible Medicines). (n.d.). The Value of Generic & Biosimilar Medicines. Retrieved from https://accessiblemeds.org/