Last updated: February 19, 2026
Cefdinir is a third-generation oral cephalosporin antibiotic used to treat bacterial infections. The market for cefdinir is mature, with significant generic competition. Its financial trajectory is characterized by declining revenues due to price erosion, although volume may remain stable or see modest growth in certain emerging markets. Key drivers influencing its future include antibiotic resistance patterns, the availability of newer broad-spectrum antibiotics, and healthcare policy reforms impacting drug pricing and access.
What is Cefdinir's Primary Indication and Mechanism of Action?
Cefdinir is indicated for the treatment of mild to moderate infections caused by susceptible bacteria. These indications include acute bacterial exacerbations of chronic bronchitis, acute otitis media (ear infections), pharyngitis/tonsillitis, and uncomplicated skin and skin structure infections. Its spectrum of activity covers many Gram-positive and Gram-negative bacteria.
The mechanism of action for cefdinir, like other cephalosporins, involves the inhibition of bacterial cell wall synthesis. It binds to penicillin-binding proteins (PBPs) located in the bacterial cell wall. These PBPs are enzymes responsible for the final stages of peptidoglycan synthesis. By binding to PBPs, cefdinir prevents the cross-linking of peptidoglycan chains, leading to a weakened cell wall and subsequent bacterial lysis.
What is the Current Market Size and Growth Outlook for Cefdinir?
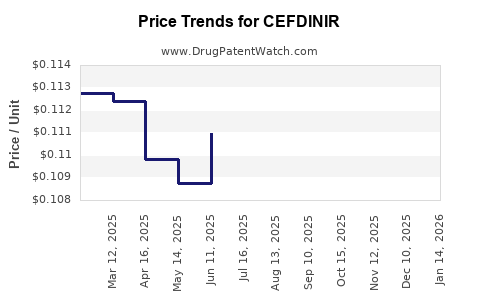
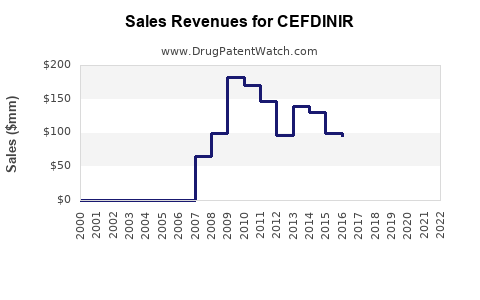
The global market for cefdinir is estimated to be in the range of $300 million to $400 million annually. This figure reflects the mature nature of the drug, which has been available in generic form for several years. The market has experienced a significant decline from its peak during its patent-protected life.
The growth outlook for cefdinir is generally stagnant to slightly negative in developed markets due to intense generic competition and the availability of alternative treatments. Unit sales may see modest increases in emerging economies as access to essential medicines expands. However, these volume gains are unlikely to offset the price erosion driven by competitive pressures and healthcare cost containment efforts. Forecasts suggest a compound annual growth rate (CAGR) of -1% to 0% over the next five years.
Key factors impacting the market size and growth include:
- Generic Competition: Cefdinir has been off-patent for over a decade, leading to a proliferation of generic manufacturers and a significant reduction in average selling prices.
- Antibiotic Resistance: The increasing prevalence of antibiotic-resistant bacteria can limit the efficacy of cefdinir for certain infections, prompting a shift to alternative agents.
- Newer Antibiotic Development: The introduction of novel antibiotics with broader spectrums or improved resistance profiles can displace older drugs like cefdinir.
- Healthcare Reimbursement Policies: Government and private payer policies, particularly in the U.S. and Europe, can influence prescribing patterns and drug prices.
- Emerging Markets: Growth opportunities exist in regions where access to broad-spectrum antibiotics is expanding, but pricing remains a critical factor.
Who are the Key Manufacturers and Competitors in the Cefdinir Market?
The cefdinir market is characterized by a fragmented landscape of generic manufacturers. Major players include both large multinational pharmaceutical companies with generic divisions and specialized generic drug producers.
Key Manufacturers (Representative List, not exhaustive):
- Teva Pharmaceutical Industries
- Sandoz (a Novartis division)
- Mylan (now Viatris)
- Aurobindo Pharma
- Dr. Reddy's Laboratories
- Sun Pharmaceutical Industries
- Cipla
Competitive Landscape:
The competition among these manufacturers is primarily based on price. Due to the commoditized nature of off-patent antibiotics, the ability to produce at a low cost and secure distribution agreements is crucial for market share. The competitive intensity is high, leading to continuous price declines.
Beyond direct generic competition, cefdinir also faces indirect competition from:
- Other Oral Cephalosporins: Cefixime, cephalexin, cefpodoxime proxetil.
- Macrolides: Azithromycin, clarithromycin (often used for respiratory infections).
- Fluoroquinolones: Levofloxacin, ciprofloxacin (used for more severe or resistant infections, with increasing concerns about side effects).
- Amoxicillin/Clavulanate: A common alternative for certain bacterial infections.
- Newer Antibiotic Classes: The development of novel agents designed to overcome resistance mechanisms represents a long-term competitive threat.
What are the Patent Expirations and Regulatory Status of Cefdinir?
Cefdinir was originally developed by Astellas Pharma and marketed under the brand name Omnicef. The compound patent for cefdinir expired in the United States in 2008. In Europe, patent expirations varied by country but generally occurred around the same period.
- U.S. Patent Expiration: Approximately 2008.
- European Patent Expirations: Varied by country, generally between 2007 and 2010.
Following patent expiration, the U.S. Food and Drug Administration (FDA) and other regulatory bodies opened the market to generic versions. This led to a rapid increase in the number of approved generic cefdinir products.
Regulatory Status:
Cefdinir is approved for marketing in numerous countries globally. Regulatory agencies such as the FDA, European Medicines Agency (EMA), and others have approved generic versions based on bioequivalence studies demonstrating that they are safe and effective as the reference listed drug.
The regulatory landscape for antibiotics is also evolving, with increasing focus on stewardship and the responsible use of antibiotics to combat resistance. This can indirectly impact the market for older antibiotics by encouraging the use of narrower-spectrum agents or newer therapies when appropriate.
What are the Key Market Trends and Drivers for Cefdinir?
Several trends and drivers are shaping the cefdinir market:
- Antibiotic Stewardship Programs: Increased emphasis on antibiotic stewardship in hospitals and outpatient settings aims to optimize antibiotic use, reduce unnecessary prescriptions, and slow the development of resistance. This can lead to more judicious use of cefdinir, potentially impacting overall volume.
- Rise of Antibiotic Resistance: The growing challenge of antimicrobial resistance (AMR) necessitates a careful selection of antibiotics. While cefdinir remains effective against many common pathogens, resistance to cephalosporins is increasing in certain bacteria, leading to a preference for agents with different mechanisms of action or broader coverage for difficult-to-treat infections.
- Focus on Outpatient Antibiotic Use: A significant portion of cefdinir prescriptions are for outpatient use. Trends in primary care, including the management of common infections like otitis media and pharyngitis, directly influence cefdinir demand.
- Pricing Pressures and Healthcare Cost Containment: Governments and payers globally are implementing measures to control healthcare spending. This translates into intense pricing pressure on generic drugs, including cefdinir, forcing manufacturers to operate on thin margins.
- Emerging Market Growth Potential: As healthcare infrastructure improves and access to medicines expands in developing countries, there is a potential for increased demand for essential antibiotics like cefdinir. However, affordability remains a key barrier.
- Shift Towards Combination Therapies: For certain complex or resistant infections, there is a trend towards using combination therapies or newer agents with unique mechanisms. This can reduce reliance on single-agent older antibiotics.
- Clinical Guidelines Evolution: Updates to clinical practice guidelines for infectious diseases can alter treatment recommendations, impacting the choice of antibiotics and, consequently, the market for cefdinir.
What are the Financial Implications and Profitability for Cefdinir Manufacturers?
The financial implications for manufacturers of cefdinir are primarily characterized by low profit margins and high-volume sales.
- Revenue Erosion: Due to the generic nature of the market, average selling prices (ASPs) for cefdinir have fallen dramatically. Revenue generation relies heavily on achieving significant market share through aggressive pricing strategies.
- Cost of Goods Sold (COGS): To remain competitive, manufacturers must maintain very low COGS. This requires efficient manufacturing processes, supply chain optimization, and sourcing of raw materials at competitive prices.
- Profit Margins: Profit margins on generic cefdinir are generally low, typically in the single to low double digits. Companies must rely on economies of scale to achieve profitability.
- Market Entry Barriers: While patent barriers are gone, significant capital investment is required for Good Manufacturing Practice (GMP) compliant manufacturing facilities, regulatory approvals (ANDA in the US), and robust distribution networks. However, for established generic players with existing infrastructure, entry is feasible.
- Competition Impact: The sheer number of generic competitors intensifies price wars, further compressing margins. A manufacturer's ability to secure preferred pharmacy benefit manager (PBM) or formulary placements can be critical for sustained sales.
- Volume vs. Value: The financial success of cefdinir manufacturers hinges on maintaining high sales volumes rather than high unit prices. Companies may focus on expanding into less price-sensitive emerging markets or diversifying their product portfolios to mitigate risks associated with individual product profitability.
- Supply Chain Dynamics: Disruptions in the global pharmaceutical supply chain can impact the availability and cost of raw materials, affecting production costs and potentially leading to shortages. Companies with secure and diversified supply chains have a competitive advantage.
Financial Trajectory:
The financial trajectory for cefdinir manufacturers is one of managing a mature, highly competitive product. Revenue growth from cefdinir alone is unlikely. Companies achieve financial success by:
- Maintaining a significant share of the generic market through competitive pricing.
- Leveraging established manufacturing and distribution capabilities.
- Potentially using cefdinir as a loss leader or staple product in their generic portfolios.
- Focusing on cost leadership to achieve profitability.
What are the Future Outlook and Potential Challenges for Cefdinir?
The future outlook for cefdinir is shaped by a confluence of technological, clinical, and economic factors.
Future Outlook:
- Continued Generic Dominance: Cefdinir will remain a widely available generic antibiotic for the foreseeable future. Its established efficacy against common pathogens ensures its continued use in specific therapeutic niches.
- Stable to Declining Demand in Developed Markets: Demand in North America and Europe is likely to remain stable or experience a slow decline as newer agents gain traction and antibiotic stewardship programs refine prescribing practices.
- Modest Growth in Emerging Markets: Continued improvements in healthcare infrastructure and increased affordability in some emerging economies may lead to a gradual increase in cefdinir consumption.
- Niche Applications: Cefdinir may find continued use in specific outpatient settings where cost-effectiveness and a favorable side-effect profile for uncomplicated infections are prioritized.
Potential Challenges:
- Escalating Antibiotic Resistance: The most significant long-term challenge is the global rise of antibiotic resistance. As resistance to cephalosporins increases, the utility of cefdinir will diminish for an expanding array of bacterial infections. This could lead to its eventual relegation to less common or more specific indications.
- Competition from Novel Antibiotics: Ongoing research and development efforts are yielding new classes of antibiotics with novel mechanisms of action designed to overcome existing resistance. These newer agents, while often more expensive, may offer superior efficacy against resistant pathogens, gradually displacing older antibiotics.
- Stricter Antibiotic Stewardship: Enhanced focus on antibiotic stewardship globally will likely lead to more precise and targeted antibiotic prescribing. This could reduce the broad empirical use of cefdinir, favoring narrower-spectrum agents or pathogen-specific therapies.
- Pricing Pressures: The relentless pressure on drug pricing, particularly for generics, will continue to squeeze profit margins for cefdinir manufacturers, making it difficult to sustain investment in production or market expansion.
- Regulatory Scrutiny: While cefdinir is a well-established drug, potential future regulatory actions concerning antibiotic use or specific side effects could impact its market standing.
- Shifting Treatment Paradigms: As diagnostic capabilities improve and understanding of infectious disease pathogenesis deepens, treatment paradigms may shift away from broad-spectrum empirical therapy towards more personalized medicine approaches.
The ability of cefdinir to maintain its market position will depend on its continued efficacy against prevalent pathogens, its cost-effectiveness relative to newer alternatives, and the evolving landscape of antibiotic resistance and stewardship.
Key Takeaways
- Cefdinir is a mature generic antibiotic with an estimated global market size of $300 million to $400 million.
- The market is highly competitive, with numerous generic manufacturers driving significant price erosion and low profit margins.
- Patent expirations occurred around 2008 in major markets, opening the door for generic entry.
- Key market trends include antibiotic stewardship, rising resistance, and global pricing pressures.
- Future growth is limited, with stagnation or decline expected in developed markets and modest potential in emerging economies.
- Long-term challenges include escalating antibiotic resistance and the development of novel, more effective antibiotics.
FAQs
-
What is the current average selling price (ASP) for generic cefdinir?
The ASP for generic cefdinir varies by dosage form, strength, and geographic region, but it is typically in the range of $0.10 to $0.30 per capsule or suspension dose. This reflects intense price competition among manufacturers.
-
Are there any new indications or formulations of cefdinir currently in development?
Given the drug's mature status and generic availability, there is minimal investment in developing new indications or novel formulations for cefdinir. Research efforts are largely focused on newer antibiotic classes.
-
How does the rise of antibiotic-resistant bacteria specifically impact cefdinir's market share?
As resistance to cephalosporins, including cefdinir, increases in common pathogens like Streptococcus pneumoniae or Staphylococcus aureus, clinicians may opt for alternative antibiotics with different mechanisms of action or broader efficacy profiles, leading to a gradual erosion of cefdinir's market share for certain infection types.
-
What is the typical market share distribution among major generic cefdinir manufacturers?
The market share distribution is highly fragmented and fluid, with no single manufacturer dominating. Large generic players like Teva, Sandoz, and Viatris typically hold significant portions, but smaller specialized manufacturers also compete vigorously on price.
-
What are the primary drivers for maintaining cefdinir's presence in emerging markets despite global challenges?
In emerging markets, cefdinir's continued presence is driven by its established efficacy against common bacterial infections, its relatively low cost compared to newer antibiotics, and its broad availability as an essential medicine, contributing to improved public health outcomes in resource-constrained settings.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/orange-book-approved-drug-products-therapeutic-equivalence-evaluations
[2] Astellas Pharma. (2007). Omnicef (Cefdinir) Prescribing Information. (Original source information may vary based on current labeling).
[3] Various Market Research Reports on Antibiotics Market. (Specific reports and publishers vary, e.g., Grand View Research, Mordor Intelligence, Allied Market Research). Information compiled from publicly available summaries and industry analyses.
[4] World Health Organization. (2020). Global priority list of antibiotic-resistant bacteria to inform research and development of new antibiotics. Retrieved from https://www.who.int/news-room/detail/27-02-2020-world-health-organization-reveals-global-list-of-bacterial-pathogens-that-most-urgently-need-new-antibiotics