Last updated: April 24, 2026
Cefdinir is a branded and generic oral third-generation cephalosporin used for respiratory, ear, and skin/soft tissue infections. The market dynamic is defined by (i) long patent and exclusivity cycles that shifted volume to multi-source generics, (ii) price compression typical of mature antibiotics, and (iii) steady baseline demand tied to pediatric and primary-care prescribing. Financial trajectory trends toward low-to-mid single digit revenue growth when measured across geographies, with margin and price pressure driven by generic competition and payer formulary positioning.
What is cefdinir’s market structure and competitive landscape?
Multi-source generic reality
Cefdinir is widely available as an oral cephalosporin antibiotic in multiple strengths (typical label strengths include 300 mg capsules and 250 mg per 5 mL oral suspension). With branded exclusivity largely elapsed in most major markets, the commercial environment is dominated by generics. This structure drives:
- SKU proliferation (capsules, suspensions, and store-brand equivalents)
- Buyer leverage (wholesalers, PBMs, and hospital pharmacy formularies)
- Contract pricing as the key determinant of realized price
Primary customer channels
Demand is concentrated in:
- Retail pharmacy sales for outpatient infections
- Pediatric and family medicine prescribing workflows
- Walk-in and urgent care settings, where broad-spectrum oral antibiotics remain common
Competitive substitution
Cefdinir competes with other oral antibiotics with overlapping indications, particularly in the same patient segments:
- Amoxicillin-clavulanate
- Azithromycin (macrolides)
- Cephalexin and other cephalosporins
- Doxycycline in older pediatric and adult populations, depending on guidelines and local reimbursement
In mature antibiotic markets, substitution is driven less by clinical differentiation and more by net price, tolerability profile as perceived by prescribers, and formulary tier placement.
How do prescription and payer dynamics shape demand?
Indication-driven volume stability
Cefdinir’s demand is anchored by common outpatient infection categories, including:
- Otitis media
- Pharyngitis/tonsillitis
- Acute bacterial sinusitis
- Skin and soft tissue infections (non-complicated in standard labeling contexts)
Because these are recurring acute conditions, cefdinir tends to hold a floor of demand even as specific competitors gain share based on relative price and guideline preference.
Formulary tier placement and net price compression
The economic mechanism is standard in generic antibiotics:
- PBMs use multiple-source class coverage
- Formularies increasingly reward lowest net cost for equivalent dosing
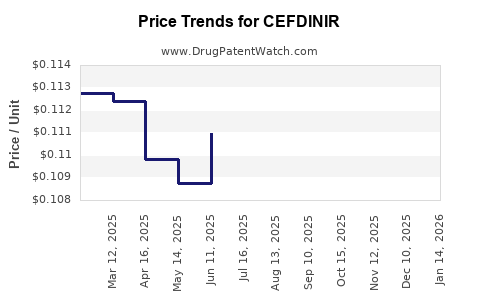
- Net price declines faster than list price, depending on contracting cycles
This results in a typical trajectory:
- Near-term revenue growth can occur when market coverage expands (new generic entrants or distribution gains)
- Long-run revenues flatten due to ongoing competition
Stewardship and prescribing intensity
Antibiotic stewardship affects overall category volumes but does not eliminate use of cefdinir-like oral agents. In many markets:
- Reduced inappropriate prescribing lowers total antibiotic scripts
- Cefdinir still benefits when clinicians follow guidance for beta-lactam options in pediatric and outpatient care
What is the regulatory and product profile that supports continued prescribing?
Cefdinir is supported by an established clinical and regulatory record for oral use. Its commercial durability comes from:
- A familiar dosing regimen and established labeling
- Broad availability in common pediatric-friendly formulations (notably oral suspension)
That combination stabilizes uptake because it lowers adoption friction for primary care physicians and pediatric prescribers compared with less established antibiotics.
How does generic competition influence financial trajectory?
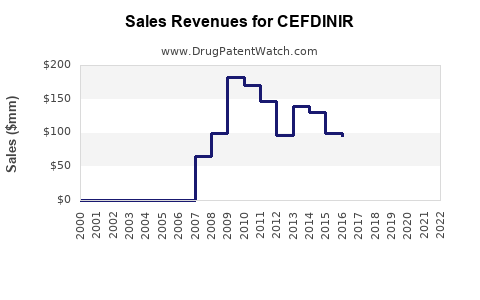
Revenue: maturity profile
The financial trajectory for cefdinir in most markets follows the mature-generic pattern:
- List price declines after generic entry waves
- Net revenue stabilizes as volume offsets price decreases
- Margin compresses as manufacturers compete for formulary placement and distribution contracts
The outcome is typically:
- Low growth, sometimes declining overall revenue in regions with aggressive contracting and high generic density
- Higher growth mainly tied to distribution expansion and channel mix changes
Profitability: margin pressure with manufacturing scale
In a multi-source generic environment, profitability hinges on:
- Manufacturing efficiency (API and finished form)
- Supply reliability
- Contract pricing and tender wins
As entrants increase, price and margin compress, pushing profitability toward firms with scale and strong cost position.
What does the manufacturing and distribution reality imply for earnings sensitivity?
Cefdinir’s financial performance is sensitive to:
- Tender and PBM contracting cycles
- Wholesaler inventory dynamics (stock-up phases can temporarily lift shipments, then swing down)
- SKU mix (capsules vs suspension) which affects conversion costs and logistic economics
- Raw material and conversion cost volatility typical in antibiotic manufacturing
In mature generics, these factors can create quarter-to-quarter swings even when underlying prescription trends remain stable.
How does product lifecycle and exclusivity history affect near- and long-term outlook?
Lifecycle phase
Cefdinir is in a mature phase where:
- New revenue creation is limited by the absence of meaningful exclusivity expansion
- Growth relies on continued generic penetration and distribution mix improvements
In practice, the strategic options for suppliers are constrained to:
- Cost leadership
- Contracting performance
- Supply continuity
- Brand-equivalent packaging and patient adherence improvements
Pipeline and next-generation risk
The main risk to cefdinir’s trajectory is not a single clinical competitor but the cumulative effect of:
- shifts in guideline preference toward certain antibiotic classes
- rapid genericization of alternative agents with more favorable economics
- stewardship-driven reductions in overall antibiotic use
What are the key indicators investors and operators should track for cefdinir?
For a mature generic like cefdinir, the operational indicators that map to financial trajectory are:
Demand and share
- Retail prescription counts and persistence of use for pediatric otitis media and sinus indications
- Uptake in formularies with tier-1 or preferred positions
Commercial
- Net pricing vs list pricing (PBM and contract outcomes)
- Tender awards and contract duration (12 to 24 months typical in many markets)
Supply and execution
- Production uptime and fulfillment rates
- Shortage premiums or reimbursement shifts during supply disruptions
- SKU-level profitability by dosage form (capsules vs suspension)
Regulatory and quality
- Any FDA or equivalent authority inspection outcomes affecting supply continuity
- Label or manufacturing changes that alter market access
What is the practical financial trajectory summary by region and time horizon?
Near-term (0 to 12 months)
- Revenue: tends to be stable to slightly down/up depending on tender cycle timing and SKU mix
- Margins: compress under aggressive contract competition; improve only when a supplier wins preferred access or secures cost advantage
Medium-term (1 to 3 years)
- Revenue: tends to flatten as competitive density remains high and payer purchasing drives pricing down
- Earnings: more sensitive to manufacturing cost and contract wins than to new prescriptions
Long-term (3+ years)
- Revenue: tracks category stabilization or modest growth tied to baseline infection incidence and pediatric prescribing patterns
- Margins: structurally low relative to branded eras; sustainability depends on scale and cost leadership
This trajectory is consistent with how multi-source oral antibiotics settle into commoditized pricing after exclusivity ends.
Key Takeaways
- Cefdinir’s market is mature and multi-source, with competition driven primarily by net price, formulary placement, and contracting.
- Demand is relatively stable because it attaches to common outpatient and pediatric infection indications, but growth is limited by stewardship and class substitution.
- Financial trajectory is dominated by price compression and margin pressure, with earnings sensitivity to manufacturing scale and tender outcomes rather than breakthrough demand.
- The most actionable levers for manufacturers are cost position, supply reliability, and contract strategy to preserve volume at acceptable net pricing.
FAQs
1) Is cefdinir a branded or generic-driven market?
Cefdinir is a multi-source generic-driven market in most major geographies, with branded share historically limited to earlier lifecycle periods and modern demand dominated by generic equivalents.
2) What determines cefdinir pricing power today?
Realized pricing is primarily determined by PBM and wholesaler contracting, formulary tier placement, and net price in competitive tenders among multiple generic suppliers.
3) What indications support cefdinir’s baseline demand?
Cefdinir demand is anchored by common outpatient infections, including otitis media, pharyngitis/tonsillitis, sinusitis, and non-complicated skin/soft tissue infections as reflected in standard oral cephalosporin prescribing.
4) What is the main financial risk for cefdinir manufacturers?
The dominant risk is margin compression from ongoing generic competition combined with tender cycles and manufacturing cost volatility affecting contract profitability.
5) What operational signals best predict financial outcomes?
Monitor net pricing (contract results), prescription/dispensing volume trends, and supply execution (fill rates, shortages, and SKU mix), since these drive quarterly swings in a mature generic environment.
References
[1] FDA. “Cefdinir” drug information (labeling and approval history resources). U.S. Food and Drug Administration.
[2] FDA. Orange Book (Drug Products Approved for Marketing with Therapeutic Equivalence Evaluations) for cefdinir. U.S. FDA.
[3] Centers for Medicare & Medicaid Services (CMS). National and coverage resources affecting outpatient antibiotic reimbursement and formulary dynamics. CMS.
[4] IQVIA/industry market reports on antibiotic retail channel dynamics (multi-source generic competition and pricing trends). IQVIA.