Share This Page
Drug Price Trends for TUSSIN DM
✉ Email this page to a colleague
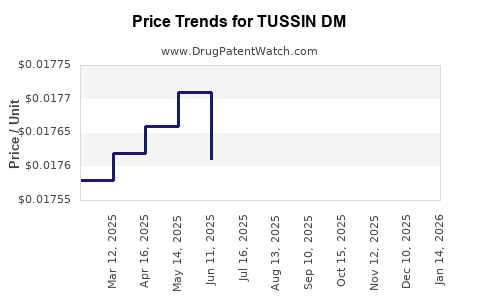
Average Pharmacy Cost for TUSSIN DM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TUSSIN DM 400-20 MG/20 ML LIQ | 70000-0628-02 | 0.01788 | ML | 2026-05-20 |
| TUSSIN DM 200-20 MG/20 ML LIQ | 70000-0670-01 | 0.01973 | ML | 2026-05-20 |
| TUSSIN DM 400-20 MG/20 ML LIQ | 70000-0628-01 | 0.02142 | ML | 2026-05-20 |
| TUSSIN DM 20-200 MG/10 ML LIQ | 82568-0015-04 | 0.01104 | ML | 2026-05-20 |
| TUSSIN DM 200-20 MG/20 ML LIQ | 70000-0670-01 | 0.02023 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is TUSSIN DM and its Market Position?
TUSSIN DM is an over-the-counter (OTC) cough and cold medication containing dextromethorphan (a cough suppressant) and guaifenesin (an expectorant). Marketed primarily in the United States, it targets consumers seeking symptomatic relief from cough and congestion. The drug is not a prescription medication but competes within a highly saturated OTC market with brands like Robitussin DM and Mucinex DM.
What is the Regulatory Status and Patent Landscape?
TUSSIN DM generally lacks recent patent protection, as its active ingredients are off-patent. The original formulations likely received patent exclusivity decades ago, but recent formulations are typically considered generic or unpatented. This situation emphasizes a focus on branding, marketing, and shelf placement rather than patent-protected exclusivity.
How Does the Market Size and Demand Profile Look?
The cough and cold segment in the United States was valued at approximately $2.1 billion in 2022. OTC cough suppressants and expectorants account for around 60% of this total, driven by seasonal demand and annual cold prevalence.
- Annual OTC cough and cold sales (US): $1.26 billion (2022 estimate)
- Dextromethorphan-based products' market share: ~70% of OTC cough suppressants
- Consumer demographics: Adults aged 25-55, immunocompromised individuals, and parents purchasing for children
- Seasonality: Heavy sales during winter months (November through February)
The market exhibits high competition, low barriers to entry, and price sensitivity, which influences pricing strategies and profitability.
What are the Key Competitive Dynamics?
Major competitors include:
- Robitussin DM (Pfizer/Johnson & Johnson)
- Mucinex DM (Reckitt Benckiser)
- CVS, Walgreens, and private-label brands
Market share is fragmented among these players, with no single brand dominating. Price wars and promotional discounts are typical, impacting margins.
What Are the Price Points and Distribution Channels?
Average retail price for a 4 oz (120 mL) bottle of TUSSIN DM ranges from $7.99 to $11.99. During peak cold season, promotional discounts and multi-pack offers are common.
Distribution occurs primarily through:
- Chain drugstores
- Supermarkets
- Online retailers
- Convenience stores
Public health campaigns and seasonal demand influence sales volume spikes but do little to alter pricing.
What Are Future Price Projections?
Given the market saturation and absence of patent exclusivity:
| Timeline | Price Trend | Rationale |
|---|---|---|
| 2023-2025 | Stable | Competition maintains pricing; no significant patent protection; price sensitivity remains high |
| 2026-2030 | Slight decline | Entry of generics and private-label brands exert pressure, potential for price erosion |
| Post-2030 | Stabilization or further decline | Market saturation, shifts to generic brands, or new formulations affect pricing |
Historical data suggests OTC drug prices tend to decline gradually driven by generic competition, averaging a 3-5% decrease annually.
Market Entry and Expansion Considerations
For new entrants or existing players seeking expansion:
- Focus on strategic branding and marketing to differentiate from commodities
- Leverage online and e-commerce platforms for consumer engagement
- Invest in formulation improvements or combination products to capture niche segments
Risk Factors and Market Challenges
- Regulatory shifts impacting OTC formulations
- Increased competitor presence
- Price sensitivity among consumers
- Seasonal demand variability
Key Takeaways
- TUSSIN DM operates in a mature, commoditized OTC segment with no recent patent protection.
- The US market for cough and cold products exceeds $2 billion annually, with dextromethorphan-based products representing a significant share.
- Pricing remains competitive, with minimal upward movement, and expected slight declines over the next decade.
- Market dynamics favor branding and marketing over patent protection; prices are driven by competition and consumer demand.
- Future growth opportunities hinge on product differentiation, online channels, and alternative formulations.
FAQs
1. How does the lack of patent protection influence TUSSIN DM’s pricing?
Without patent protection, TUSSIN DM faces generic competition, limiting pricing power and leading to price erosion over time. Market share retention depends on branding and consumer loyalty.
2. What is the primary driver of demand for OTC cough medicines like TUSSIN DM?
Demand correlates with cold and flu seasons, seasonal cold prevalence, and consumer preferences for non-prescription symptomatic relief.
3. Are there opportunities for higher margins in the OTC cough and cold market?
Margins are limited by high competition and price sensitivity. Differentiation through formulation improvements or targeted marketing can create niche opportunities.
4. How might online sales influence future pricing?
Online channels often exert downward pressure on prices due to increased price transparency and direct competition, intensifying market competition.
5. What regulatory considerations could impact TUSSIN DM’s market?
Any regulatory changes affecting active ingredient safety, labeling, or OTC status could alter market dynamics and pricing strategies.
References
- Statista. "U.S. OTC Cold & Cough Medicine Market Size." 2022.
- IBISWorld. "Cold & Flu Medicine in the US." 2022.
- U.S. Patent and Trademark Office. "OTC Drug Product Patents." 2023.
- NielsenIQ. Consumer Packaged Goods Data. 2022.
- FDA. "OTC Drug Review and Regulations." 2023.
More… ↓
