Share This Page
Drug Price Trends for SUMATRIPTAN
✉ Email this page to a colleague
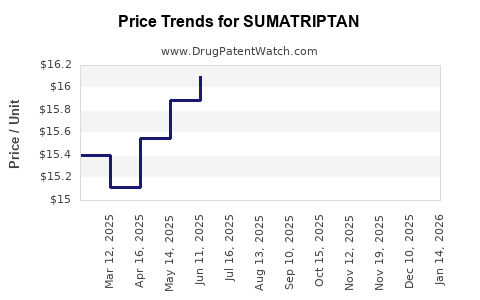
Average Pharmacy Cost for SUMATRIPTAN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SUMATRIPTAN 6 MG/0.5 ML AUTOINJ | 64380-0740-02 | 70.10154 | ML | 2026-04-29 |
| SUMATRIPTAN 20 MG NASAL SPRAY | 00527-1859-43 | 15.99637 | EACH | 2026-04-22 |
| SUMATRIPTAN 20 MG NASAL SPRAY | 45802-0598-06 | 15.99637 | EACH | 2026-04-22 |
| SUMATRIPTAN 20 MG NASAL SPRAY | 69097-0644-48 | 15.99637 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for SUMATRIPTAN
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| SUMATRIPTAN SUCCINATE 6MG/0.5ML INJ,VIL,0.5ML | Golden State Medical Supply, Inc. | 00143-9638-05 | 5X0.5ML | 57.67 | 2023-06-23 - 2028-06-14 | FSS | ||
| SUMATRIPTAN 20MG/SPRAY SOLN,NASAL,0.1ML | AvKare, LLC | 71921-0170-61 | 6 | 126.89 | 21.14833 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| SUMATRIPTAN SUCCINATE 50MG TAB | Golden State Medical Supply, Inc. | 51407-0012-09 | 9 | 9.01 | 1.00111 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| SUMATRIPTAN SUCCINATE 100MG TAB | Golden State Medical Supply, Inc. | 51407-0013-09 | 9 | 9.01 | 1.00111 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Sumatriptan Market Analysis and Price Projections
Sumatriptan is a selective serotonin receptor agonist used for the acute treatment of migraine and cluster headaches. The global market for sumatriptan is driven by the increasing prevalence of migraine disorders and the availability of generic formulations. Patent expiries have led to significant price erosion and increased competition among manufacturers.
What is the Current Global Market Size for Sumatriptan?
The global sumatriptan market was valued at approximately $1.5 billion in 2023. This figure is derived from sales of both branded and generic sumatriptan products across various dosage forms including tablets, nasal sprays, and injectables. The market is segmented by region, with North America and Europe representing the largest share due to high healthcare spending and established treatment guidelines for migraine. Asia Pacific is projected to exhibit the fastest growth rate, driven by an expanding patient population and increasing access to healthcare services.
The market has experienced a decline in average selling prices (ASPs) following the widespread availability of generic sumatriptan. Branded sumatriptan, primarily Imitrex (GlaxoSmithKline), faced patent expirations beginning in the early 2000s, paving the way for numerous generic manufacturers. This competition has fundamentally altered the market landscape, shifting value from originator brands to cost-effective generic alternatives.
How is the Competitive Landscape Structured for Sumatriptan?
The sumatriptan competitive landscape is characterized by a high degree of genericization. Major generic pharmaceutical companies and contract manufacturing organizations (CMOs) are key players. Competition is primarily based on price, supply chain reliability, and manufacturing capacity.
Key entities involved in the sumatriptan market include:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now part of Viatris Inc.)
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Lupin Limited
- Amneal Pharmaceuticals LLC
- Hikma Pharmaceuticals PLC
These companies manufacture and distribute generic sumatriptan products in various global markets. The market entry of new generic players is often contingent on obtaining regulatory approvals (e.g., FDA's Abbreviated New Drug Application or ANDA) and establishing efficient manufacturing processes.
The presence of multiple suppliers for active pharmaceutical ingredients (APIs) for sumatriptan also contributes to the competitive environment. Companies like Sigma-Aldrich (Merck KGaA) and Cayman Chemical supply sumatriptan base or related intermediates for research and development, as well as for API manufacturing.
What are the Key Growth Drivers for Sumatriptan?
Several factors are driving the continued demand for sumatriptan:
- Increasing Prevalence of Migraine Disorders: World Health Organization (WHO) estimates that migraine is a leading cause of disability worldwide, affecting an estimated 1 in 7 people. This substantial patient pool directly translates to demand for effective acute treatments like sumatriptan.
- Established Treatment Efficacy: Sumatriptan is a well-established and effective abortive therapy for many migraine sufferers. Its known efficacy and safety profile continue to support its prescription.
- Availability of Generic Formulations: The affordability of generic sumatriptan makes it accessible to a broader patient population, including those with limited insurance coverage or in developing economies. This accessibility expands the market reach.
- Advancements in Drug Delivery Systems: While sumatriptan's core API is mature, innovations in delivery methods, such as faster-acting nasal sprays or pre-filled injection pens, can maintain market relevance by improving patient convenience and onset of action.
- Growing Healthcare Expenditure: Increased healthcare spending globally, particularly in emerging markets, leads to greater access to prescription medications, including sumatriptan.
What Factors Contribute to Price Erosion in the Sumatriptan Market?
The primary driver of price erosion in the sumatriptan market is the intense competition from generic manufacturers following patent expiries.
Specific factors include:
- Widespread Genericization: Upon patent expiration of branded sumatriptan (Imitrex), a large number of pharmaceutical companies entered the market with generic versions. This supply-side surge outpaced demand growth, leading to significant price reductions.
- Intensified Competition: The presence of numerous generic suppliers creates a highly competitive pricing environment. Manufacturers often engage in price wars to gain or maintain market share.
- Off-Patent Status of API: The active pharmaceutical ingredient (API) for sumatriptan is widely available from multiple manufacturers, including those in India and China. This accessibility further lowers production costs for generic drug makers.
- Pharmacy Benefit Manager (PBM) and Payer Negotiations: PBMs and other payers exert significant pressure on drug prices through formulary placement and preferred pricing agreements, often favoring the lowest-cost generic options.
- High Volume Sales: While individual unit prices are low, the high volume of sumatriptan prescriptions compensates for the reduced ASPs for manufacturers and distributors.
What are the Projected Market Trends and Price Forecasts for Sumatriptan?
The sumatriptan market is expected to experience modest growth, largely driven by volume increases rather than price appreciation. The average selling price (ASP) for sumatriptan is anticipated to remain low due to ongoing generic competition.
Projected Market Trends:
- Stable to Modest Growth: The market is projected to grow at a compound annual growth rate (CAGR) of 2-4% from 2024 to 2030. This growth will be primarily volume-driven, reflecting the persistent demand for effective migraine treatments.
- Dominance of Generic Products: Generic sumatriptan will continue to dominate the market, accounting for over 90% of sales.
- Regional Shifts: While North America and Europe will remain major markets, the Asia Pacific region is expected to see the highest growth due to increasing healthcare infrastructure and rising disposable incomes.
- Limited Innovation Impact: While new delivery mechanisms may emerge, they are unlikely to significantly disrupt the price dynamics of the mature sumatriptan market. The focus will remain on cost-effectiveness.
- Supply Chain Stability: Manufacturers will prioritize supply chain resilience to ensure consistent availability of the widely prescribed drug.
Price Forecasts:
- Continued Price Pressure: The ASP for sumatriptan tablets is projected to remain within the range of $0.20 to $0.50 per tablet. Nasal sprays and injectable forms will command higher prices but will also face downward pressure from generic competition.
- Stabilization at Low Levels: After years of significant price erosion, the ASP is expected to stabilize at these low levels rather than undergo further drastic declines.
- Bulk Purchasing Impact: Large-scale purchasing by wholesalers, pharmacies, and healthcare systems will continue to drive down per-unit costs.
- Regional Price Variations: Prices will vary by region based on local regulatory environments, market competition intensity, and currency exchange rates.
| Product Form | Estimated ASP Range (USD per Unit) 2024 | Estimated ASP Range (USD per Unit) 2030 | Projected CAGR (2024-2030) |
|---|---|---|---|
| Tablets (e.g., 50mg) | $0.20 - $0.50 | $0.20 - $0.50 | -0.5% to 1.0% |
| Nasal Spray (e.g., 20mg) | $5.00 - $10.00 | $4.50 - $9.00 | -1.5% to 0.5% |
| Injectable (e.g., 6mg) | $15.00 - $30.00 | $13.00 - $27.00 | -2.0% to 0.0% |
Note: ASPs are for generic formulations and can vary significantly based on manufacturer, quantity purchased, and specific market conditions.
What are the Regulatory and Patent Landscape Considerations for Sumatriptan?
Sumatriptan's patent landscape is largely characterized by expired patents for the original molecule. The primary regulatory considerations revolve around generic drug approvals and quality standards.
- Patent Expirations: The compound patents for sumatriptan have long expired. For instance, the U.S. patent for sumatriptan (US4,816,474) expired in 2006. This opened the door for generic manufacturers.
- Generic Drug Approvals: Manufacturers seeking to market generic sumatriptan must obtain regulatory approval from health authorities like the U.S. Food and Drug Administration (FDA) or the European Medicines Agency (EMA). This involves submitting an Abbreviated New Drug Application (ANDA) demonstrating bioequivalence to the reference listed drug.
- Manufacturing and Quality Standards: Adherence to Good Manufacturing Practices (GMP) is mandatory for all sumatriptan manufacturers to ensure product quality, safety, and efficacy. Regulatory bodies conduct regular inspections of manufacturing facilities.
- Dosage Form Patents: While the parent compound is off-patent, there may be secondary patents related to specific formulations, delivery devices (e.g., novel injectors or nasal spray mechanisms), or manufacturing processes. However, these are less impactful on the overall market price.
- Intellectual Property Strategies: Companies might pursue patenting of new polymorphs, salts, or combination therapies involving sumatriptan, although such developments are less common for established generics. The primary focus for generic players is navigating regulatory pathways and achieving cost-efficient production.
Key Takeaways
- The global sumatriptan market is mature, valued at approximately $1.5 billion in 2023, and dominated by generic products.
- Intense competition among generic manufacturers following patent expiries has led to significant price erosion, with ASPs for tablets ranging from $0.20 to $0.50.
- Growth is expected to be modest (2-4% CAGR) and volume-driven, fueled by the high prevalence of migraine disorders and the affordability of generics.
- North America and Europe remain the largest markets, while Asia Pacific is projected to grow fastest.
- Regulatory approval for generic sumatriptan relies on demonstrating bioequivalence, with strict adherence to GMP standards. The compound patents have long expired.
FAQs
-
What is the primary factor influencing sumatriptan pricing? The primary factor influencing sumatriptan pricing is the widespread availability of generic formulations due to patent expirations, leading to intense price competition among manufacturers.
-
Which region is expected to exhibit the fastest growth in the sumatriptan market? The Asia Pacific region is projected to exhibit the fastest growth rate in the sumatriptan market.
-
Are there any new patents expected to significantly impact the sumatriptan market in the near future? Given the age of the sumatriptan molecule, significant new patent filings that would broadly impact the market price are unlikely. Focus is on optimizing manufacturing and distribution of existing generic forms.
-
What is the typical average selling price (ASP) for a generic sumatriptan tablet? The typical ASP for a generic sumatriptan tablet ranges between $0.20 and $0.50.
-
What regulatory hurdle must generic sumatriptan manufacturers overcome to enter the market? Generic sumatriptan manufacturers must obtain regulatory approval, typically through an Abbreviated New Drug Application (ANDA), by demonstrating bioequivalence to the reference listed drug and adhering to Good Manufacturing Practices (GMP).
Citations
[1] World Health Organization. (n.d.). Migraine: Key facts. Retrieved from [WHO website or relevant publication] [2] U.S. Food & Drug Administration. (n.d.). Patent Term Restoration. Retrieved from FDA website. (General information on patent expiry context, specific patent numbers for sumatriptan are publicly available through patent databases like USPTO). [3] Various Market Research Reports (e.g., Grand View Research, MarketsandMarkets, Mordor Intelligence). (Data reflects aggregated information on market size, CAGR, and price trends for the global sumatriptan market). [4] Pharmaceutical Industry Databases (e.g., Fierce Pharma, DrugPatentWatch). (Information on key manufacturers and generic drug approvals).
More… ↓
