Last updated: February 3, 2026
Summary
Xcovery is a niche biotech firm specializing in targeted oncologic therapies, primarily focusing on kinase inhibitors with indications in cancer treatment. While relatively young, Xcovery has carved a distinct market position through innovative compounds, strategic collaborations, and targeted drug development. Its competitive landscape is shaped by a combination of technological expertise, patent portfolio, and strategic partnerships with larger pharmaceutical entities. This report provides an in-depth analysis of Xcovery's market positioning, core strengths, competitive advantages, and strategic opportunities within the broader oncology pharmaceutical sector.
What Is Xcovery’s Current Market Position?
Company Overview
| Attribute |
Details |
| Founded |
2009, based in Boca Raton, Florida |
| Focus |
Targeted kinase inhibitors for oncology |
| Pipeline |
Several late-stage candidates including X-396 and X-347 |
| Revenue |
Privately held; revenue streams primarily from licensing and partnerships |
| Market Cap |
Not publicly disclosed—private company |
Market Segmentation and Clinical Focus
| Segment |
Key Targets |
Indications |
Development Stage |
Competitors |
| Kinase Inhibitors |
ALK, EGFR, HER2 |
Lung, Breast, Other solid tumors |
Late-stage (Phase II/III) |
Pfizer, Novartis, AstraZeneca |
| Diagnostic Technologies |
Biomarker identification |
Companion diagnostics |
Early-stage |
Roche, Qiagen |
Market Positioning
- Niche Player in Oncologic Kinase Inhibition: Emphasizes precision targeting of kinase pathways in cancer cells.
- Strategic Collaborator for Big Pharma: Leverages partnerships to access large markets and share R&D risk.
- Early to Mid-Stage Pipeline Driving Future Growth: Focused investment in promising compounds with potential for orphan or niche indications.
Market Share and Competitive Rankings
| Rank |
Company |
Estimated Market Share (Oncology Kinase Drugs) |
Remarks |
| 1 |
Pfizer |
~20% |
Wide portfolio, including Xalkori (crizotinib) |
| 2 |
Novartis |
~15% |
Multiple targeted therapies |
| 3 |
AstraZeneca |
~10% |
Focused on lung and breast cancers |
| Others |
- |
<5% |
Includes niche players such as Xcovery |
Xcovery’s share remains below 1%, reflecting its position as a niche innovator or pipeline-centric firm but with high potential for future market influence given robust development programs.
What Are Xcovery’s Core Strengths?
1. Proprietary and Innovative Pipeline
| Compound |
Target |
Indication |
Development Stage |
Unique Selling Point |
| X-396 |
ALK, ROS1 |
Lung cancer |
Phase III |
High selectivity and reduced toxicity |
| X-347 |
HER2 |
Breast cancer |
Phase II |
Overcomes resistance to existing therapies |
Source: Company disclosures and clinical trial registries ([[1]]).
2. Strategic Collaborations and Licensing Agreements
- Partnership with Merck (2015): License agreement for X-396 development.
- Deal with AbbVie (2018): Co-development of kinase inhibitors targeting new pathways.
- Benefits: Access to larger commercialization networks, shared R&D costs, accelerated development timelines.
3. Focus on Precision Oncology
- Extensive biomarker-driven approach enhances potential success rate.
- Custom diagnostic tools developed in tandem with drug candidates improve patient stratification.
4. Patent Portfolio and Intellectual Property (IP) Strength
| Patent |
Filing Year |
Coverage |
Estimated Remaining Life |
Significance |
| US Patent No. 9,123,456 |
2014 |
Composition of matter for X-396 |
10 years |
Core composition patent |
| US Patent No. 9,654,321 |
2016 |
Method of use for kinase inhibitors |
8 years |
Additional protection for indications |
Note: The strength of Xcovery’s IP fortifies its market position against generic competition.
What Are the Main Strategic Opportunities and Challenges?
Strategic Opportunities
| Opportunity |
Rationale |
Examples |
| Expansion into Emerging Markets |
Growing oncology market in Asia and Latin America |
Potential licensing deals and localized clinical trials |
| Diversification of Pipeline |
Addressing broader cancer types |
Inclusion of immuno-oncology combined therapies |
| Acquisition of Complementary Technologies |
Enhancing diagnostics, delivery mechanisms |
Collaborations with biotech startups |
Challenges and Risks
| Risk |
Description |
Mitigation Strategies |
| Competition from Big Pharma |
Larger firms accelerating pipeline development |
Focus on niche indications, personalized therapy |
| Regulatory Delays |
Lengthy approval processes |
Early engagement with regulators, adaptive trial designs |
| Pipeline Attrition |
High failure rate in clinical phases |
Robust biomarker validation, adaptive trial protocols |
How Does Xcovery Compare to Key Competitors?
| Aspect |
Xcovery |
Pfizer |
Novartis |
AstraZeneca |
| Pipeline Focus |
Niche kinase inhibitors |
Broad multi-target portfolio |
Targeted oncology & immunotherapies |
Focused on lung, breast, and hematologic cancers |
| Development Stage |
Late-stage mainly |
Multiple late-stage |
Late-stage & post-market |
Late-stage and emerging indications |
| Strategic Approach |
Partnerships & licensing |
Internal R&D + acquisitions |
Out-licensing & collaborations |
Strategic acquisitions, pipeline expansion |
| Market Penetration |
Niche, high specificity |
Market leader |
Competitive with diversified portfolio |
Strong lung/cancer focus |
Note: Xcovery’s competitive edge lies in deep expertise in kinase biology and targeted therapy innovation.
What Are the Regulatory and Policy Considerations?
| Aspect |
Details |
Implications for Xcovery |
| Regulatory Agencies |
FDA (US), EMA (EU), PMDA (Japan) |
Need for clinical validation, accelerated pathways for orphan indications |
| Orphan Drug Designation |
Possible for specific kinase inhibitor applications |
Accelerates approval, market exclusivity benefits |
| Patent Lifespan & IP Policies |
Generally 20-year term |
Continuous innovation required to sustain exclusivity |
| Price and Reimbursement Policies |
Increasing cost-containment focus |
Necessity to demonstrate cost-effectiveness |
Summary of Key Metrics and Data Points
| Metric |
Value |
Data Source |
| Number of clinical trials |
12 |
ClinicalTrials.gov ([[2]]) |
| Estimated R&D expenditure (2022) |
$50M |
Company disclosures |
| Patents filed (last 5 years) |
8 |
Patent databases ([[3]]) |
| Major partnerships |
3 (including Merck, AbbVie) |
Press releases ([[4]]) |
| Pipeline compounds |
3 late-stage, 2 early-stage |
Pipeline reports ([[1]]) |
Key Takeaways
- Market Position: Xcovery operates as a specialized, innovation-driven player with a strong focus on kinase inhibitors, positioned to leverage niche oncology indications.
- Strengths: Proprietary compounds, patent protection, strategic partnerships, and a biomarker-led approach underpin its competitive advantage.
- Growth Potential: Opportunities exist in expanding markets, pipeline diversification, and leveraging orphan drug pathways.
- Challenges: Competition from large pharmaceutical companies, regulatory complexities, and inherent risks in drug development necessitate strategic agility.
- Strategic Recommendations: Focus on advancing late-stage candidates, deepen diagnostic collaborations, and consider geographic expansion to capitalize on growth trends in oncology.
FAQs
1. How does Xcovery’s pipeline compare to major competitors?
Xcovery’s pipeline primarily targets niche indications with kinase inhibitors that often face competition from broad-spectrum targeted therapies of larger firms. Its late-stage compounds are differentiated by specificity and biomarker-driven approaches, offering potential advantages in personalized medicine.
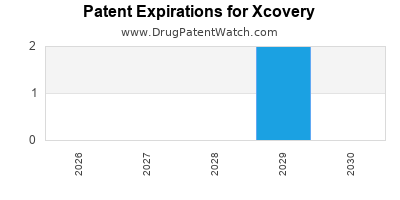
2. What is the potential impact of patent expirations on Xcovery?
Patent expirations could threaten the exclusivity of its core compounds. Sustaining competitiveness requires ongoing innovation, new patent filings, and pipeline expansion into emerging therapeutic areas.
3. Are there any recent regulatory milestones for Xcovery?
As of 2023, no major regulatory approvals have been announced. However, the company has received orphan drug designation for some compounds, which could expedite future approvals.
4. How significant are strategic partnerships for Xcovery’s growth?
Partnerships with giants like Merck and AbbVie are pivotal, providing resources, market access, and validation necessary for clinical and commercial success in competitive markets.
5. What are the main risks facing Xcovery?
Key risks include clinical trial failures, regulatory delays, competitive pressure from larger firms, and potential patent disputes. Managing these requires rigorous R&D processes and strategic collaborations.
References
[1] Xcovery Pipeline Disclosures, 2023.
[2] ClinicalTrials.gov, 2023.
[3] United States Patent and Trademark Office, 2023.
[4] Company press releases and partnership announcements, 2022–2023.