Tris Pharma Inc Company Profile
✉ Email this page to a colleague
What is the competitive landscape for TRIS PHARMA INC, and when can generic versions of TRIS PHARMA INC drugs launch?
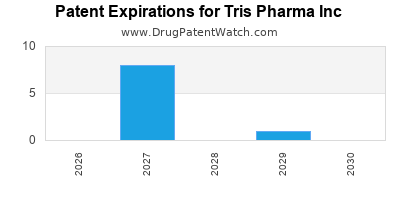
TRIS PHARMA INC has twenty-eight approved drugs.
There are eleven US patents protecting TRIS PHARMA INC drugs.
There are nineteen patent family members on TRIS PHARMA INC drugs in fourteen countries and twenty-eight supplementary protection certificates in ten countries.
Summary for Tris Pharma Inc
| International Patents: | 19 |
| US Patents: | 11 |
| Tradenames: | 28 |
| Ingredients: | 23 |
| NDAs: | 28 |
| Drug Master File Entries: | 6 |
| Patent Litigation for Tris Pharma Inc: | See patent lawsuits for Tris Pharma Inc |
Drugs and US Patents for Tris Pharma Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Tris Pharma Inc | DYANAVEL XR 5 | amphetamine; amphetamine aspartate/dextroamphetamine sulfate | TABLET, EXTENDED RELEASE;ORAL | 210526-001 | Nov 4, 2021 | RX | Yes | No | 9,675,704 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Tris Pharma Inc | DEXTROAMPHETAMINE SULFATE | dextroamphetamine sulfate | TABLET;ORAL | 206095-001 | Aug 18, 2022 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Tris Pharma Inc | DYANAVEL XR | amphetamine; amphetamine aspartate/dextroamphetamine sulfate | SUSPENSION, EXTENDED RELEASE;ORAL | 208147-001 | Oct 19, 2015 | RX | Yes | Yes | 11,590,228 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for Tris Pharma Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Japan | 5479086 | ⤷ Try a Trial |
| Japan | 2009530298 | ⤷ Try a Trial |
| Brazil | PI0709606 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Tris Pharma Inc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1539166 | 122013000090 | Germany | ⤷ Try a Trial | PRODUCT NAME: KOMBINATION AUS: (A) DEXTROMETHORPHAN ODER EINEM PHARMAZEUTISCH VERTRAEGLICHEN SALZ, Z.B. DEXTROMETHORPHAN-HYDROBROMID UND INSBESONDERE DEXTROMETHORPHAN-HYDROBROMID-MONOHYDRAT; UND (B) CHINIDIN ODER EINEM PHARMAZEUTISCH VERTRAEGLICHEN SALZ DAVON, Z.B.CHINIDIN-SULFAT UND INSBESONDERE CHINIDIN-SULFAT-DIHYDRAT; REGISTRATION NO/DATE: EU/1/13/833 20130624 |
| 1499331 | SPC/GB13/034 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: IZINOVA CONCENTRATE FOR ORAL SOLUTION. THE ACTIVE SUBSTANCE IS A MIXTURE OF 3 SALTS:SODIUM SULPHATE ANHYDROUS, MAGNESIUM SULPHATE HEPTAHYDRATE AND POTASSIUM SULPHATE.; REGISTERED: BE BE434323 20130220; UK PL34926/0016 20130313 |
| 0058146 | 2001C/045 | Belgium | ⤷ Try a Trial | PRODUCT NAME: DICHLORHYDRATE DE LEVOCETIRIZINE; NAT RER. NO/DATE: 194 IS 90 F3 20011022; FIRST REG.: DE 49903.00.00 20010103 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.