Last updated: January 1, 2026
Summary
Puma Biotechnology (NASDAQ: PBYI) is a leading oncology-focused biopharmaceutical company specializing in the development and commercialization of targeted therapies for breast cancer and other solid tumors. This analysis examines Puma's current market position, core strengths, competitive landscape, strategic initiatives, and future outlook within the burgeoning oncology market. The assessment integrates recent product launches, clinical pipeline advancements, market dynamics, and competitive challenges to guide stakeholders in making informed decisions.
What is Puma Biotech’s Market Position in Oncology?
| Aspect |
Details |
| Market Capitalization |
Approx. $800 million (as of Q1 2023) |
| Key Focus |
HER2-positive breast cancer, lung, gastric, and other solid tumors |
| Flagship Product |
Neratinib (Nerlynx) — approved for early-stage HER2-positive breast cancer |
| Market Share (Breast Cancer) |
Estimated 15-20% of targeted HER2-positive breast cancer market in U.S. (post-approval) |
| Global Presence |
Primarily U.S. with ongoing efforts for broader international approval |
The company's niche positioning centers on Neratinib's strategic differentiation as an irreversible tyrosine kinase inhibitor for HER2-positive breast cancer, capitalizing on an unmet need for extended adjuvant therapy to prevent recurrence.
What are Puma’s Core Strengths?
1. Robust Clinical Development Pipeline
| Pipeline Stage |
Indications & Candidates |
Key Data & Milestones |
| Phase 3 |
NEfERT Trial: HER2-positive breast cancer |
Demonstrated efficacy and safety for Neratinib plus trastuzumab |
| Phase 2/3 |
Lung, gastric, and other solid tumors |
Ongoing; targeting expanding indications |
| Preclinical |
Novel TKIs and antibody-drug conjugates |
Diversification of portfolio |
Strategic Advantage: Puma's focused pipeline is designed around precision oncology, leveraging strong clinical data to address specific patient populations with high unmet needs.
2. Regulatory Approvals & Market Access
| Achievements |
Details |
| FDA Approval (2017) |
Neratinib (Nerlynx) for extended adjuvant treatment |
Post-approval label expansion in 2021 for unselected patients |
| FDA Breakthrough Therapy Designation |
For certain lung and gastric indications |
Accelerates development timelines |
| Market Access Strategies |
Collaborations with payers, patient assistance programs |
Mitigates barriers to adoption |
Strategic Advantage: Strong regulatory backing bolsters credibility and facilitates market entry.
3. Niche Focus and Specialized Expertise
- Deep specialization in HER2-driven cancers offers a competitive advantage.
- High-value partnerships with big pharma (e.g., collaboration with Pfizer for HER2-positive breast cancer).
4. Financial Strength & Strategic Collaborations
- Revenue from Neratinib sales supports pipeline expansion.
- Collaborations expand research capabilities and pipeline potential.
What are the Key Competitive Challenges and Landscape?
1. Competitive Oncology Portfolio
| Major Competitors |
Market Focus |
Differentiators |
| Genentech (Roche) |
HER2-targeted therapies (e.g., trastuzumab, tucatinib) |
Market leader with established offerings and extensive pipeline |
| Novartis |
CDK4/6 inhibitors, targeted agents |
Diversified oncology portfolio |
| Seagen (Seagen) |
Antibody-drug conjugates, targeted therapies |
Innovative delivery platforms |
| Eli Lilly & Co. |
Emerging lung and breast cancer agents |
Expanding pipeline |
Competitive Dynamics: Puma faces stiff competition from large, integrated biopharma firms with more extensive sales, marketing infrastructures, and broader indications.
2. Market Penetration and Commercialization Barriers
- Limited commercial infrastructure compared to big pharma.
- Prescriber familiarity and established treatment protocols favor market incumbents.
3. Regulatory and Pricing Pressures
- Increasing scrutiny on oncology drug pricing.
- Potential hurdles in gaining approvals for new indications or international markets.
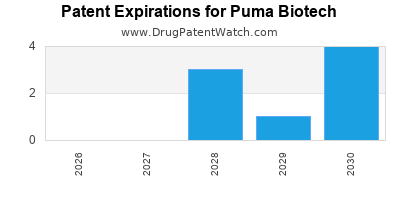
4. Patent and Patent Expiry Risks
- Patent exclusivity for Neratinib expiring in 2026 in the U.S., risking generic competition.
What Strategic Initiatives Could Puma Pursue?
1. Pipeline Expansion & Diversification
- Accelerate clinical trials for lung, gastric, and other solid tumors.
- Invest in novel modalities such as antibody-drug conjugates and immuno-oncology agents.
2. Global Market Penetration
- Accelerate efforts for approvals in Europe, Asia, and emerging markets.
- Collaborate with regional distributors to expand access.
3. Strategic Collaborations & Partnerships
| Target Areas |
Potential Partners |
Benefits |
| Immuno-oncology |
Large pharma with immune checkpoint expertise |
Synergy in combination therapies |
| Biomarker Development |
Diagnostic firms |
Better patient stratification |
| International Expansion |
Local pharma and distributors |
Accelerated market entry |
4. Commercial Infrastructure Enhancement
- Expand sales force coverage.
- Leverage digital health tools to improve prescriber engagement.
5. Intellectual Property & Patent Strategy
- Secure additional patent protections.
- Explore patent extensions via orphan drug designations.
Future Outlook and Market Trends
| Trend |
Impact on Puma |
| Growing Oncology Market |
CAGR of ~7% projected through 2027, driven by prevalence |
Puma positioned in high-growth niche |
| Shift Toward Precision Medicine |
Enhances demand for targeted agents like Neratinib |
Puma's focused pipeline aligns with trends |
| Increased Use of Combination Therapies |
Present opportunities for partnership and pipeline diversification |
Puma can develop combination regimens |
| Pricing and Regulatory Pressures |
Potential impact on revenues and market access |
Strategic planning needed |
Comparison with Major Competitors
| Parameter |
Puma Biotechnology |
Roche/Genentech |
Novartis |
Seagen |
| Market Cap |
~$800 million |
~$220 billion |
~$200 billion |
~$25 billion |
| Focus |
HER2-positive cancers |
Broad oncology |
Diverse targeted therapies |
Targeted and antibody-drug conjugates |
| Pipeline Maturity |
Core product (Neratinib), early pipeline |
Extensive, mature pipeline |
Diversified; significant R&D |
Innovative, expanding pipeline |
| Global Footprint |
Primarily U.S. |
Worldwide |
Worldwide |
Primarily U.S., expanding internationally |
Key Performance Indicators (KPIs) for Puma
| KPI |
Baseline (Q1 2023) |
Target |
Strategic Focus |
| Neratinib Sales |
$60 million |
$150 million (by 2025) |
Market expansion & new indications |
| Pipeline Progress |
3 candidates in clinical trials |
5+ candidates advancing |
R&D acceleration |
| Market Penetration |
20% in HER2-positive breast cancer |
40%+ in developed markets |
Commercial expansion |
| Global Approvals |
U.S. only |
Europe, Asia cases |
International growth |
Conclusion: Strategic Outlook for Puma Biotechnology
Puma's niche focus on HER2-positive breast cancer positions it well within a high-growth, high-need segment. The recent launch and continued expansion of Neratinib offer near-term revenue potential, while pipeline investments are critical for long-term growth. Competitive pressures from industry giants necessitate strategic collaborations, pipeline diversification, and global market penetration. Puma's agility and targeted approach could enable it to carve out a sustainable foothold despite challenges.
Key Takeaways
- Puma's primary market opportunity lies in HER2-positive breast cancer, with a solid regulatory and clinical foundation for Neratinib.
- The company’s strengths include targeted specialization and ongoing pipeline development, positioning it as a niche innovator.
- Competitive threats include large pharma entrenched players, patent expiries, and pricing pressures, requiring strategic agility.
- Future growth hinges on expanding indications, accelerating pipeline progress, and global outreach.
- Investors and stakeholders should monitor pipeline milestones, regulatory milestones outside the U.S., and partnerships to assess Puma's trajectory.
Frequently Asked Questions (FAQs)
1. How does Puma Biotechnology differentiate itself from competitors like Roche or Novartis?
Puma concentrates on highly selective targeted therapies for HER2-positive breast cancers, leveraging niche expertise and rapid clinical development. Unlike Roche's broad portfolio, Puma’s focused pipeline allows for more nimble innovation but faces resource limitations compared to industry giants.
2. What are the growth prospects for Neratinib beyond breast cancer?
Clinical trials are exploring Neratinib’s efficacy in lung, gastric, and other solid tumors, with some indications showing promise. Successful expansion into these areas could significantly boost sales, provided phase 2/3 trials confirm efficacy and safety.
3. What risks does patent expiration pose to Puma?
Patent expiry in 2026 could open avenues for generic competition, impacting revenue streams. Strategies to mitigate this include developing new formulations, expanding indications, and pursuing patent extensions or new compounds.
4. How is Puma planning to expand internationally?
The company aims to obtain regulatory approvals in Europe and Asia, leveraging partnerships and regional collaborations to accelerate market access and build a global footprint.
5. What should investors watch for to assess Puma’s future success?
Key indicators include pipeline milestones, regulatory approvals outside the U.S., sales growth of Neratinib, partnership deals, and management’s strategic initiatives toward pipeline diversification and international expansion.
References
- Puma Biotechnology Inc. Corporate Overview and Recent Results, 2023.
- FDA Approvals and Label Expansion Announcements, U.S. Food and Drug Administration, 2021.
- Market research reports on Oncology Therapeutics, 2022-2023.
- Clinical trial data from ClinicalTrials.gov, 2023.
- Industry analyses from Bloomberg Intelligence, 2023.