PHARMOBEDIENT Company Profile
✉ Email this page to a colleague
What is the competitive landscape for PHARMOBEDIENT
PHARMOBEDIENT has three hundred and thirty-three approved drugs.
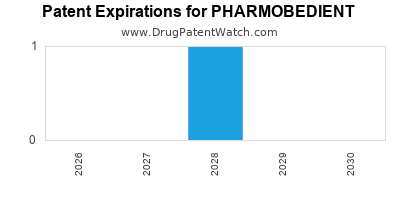
There is one US patent protecting PHARMOBEDIENT drugs. There is one tentative approval on PHARMOBEDIENT drugs.
There are twenty-one patent family members on PHARMOBEDIENT drugs in sixteen countries and six hundred and ninety-seven supplementary protection certificates in nineteen countries.
Summary for PHARMOBEDIENT
| International Patents: | 21 |
| US Patents: | 1 |
| Tradenames: | 259 |
| Ingredients: | 247 |
| NDAs: | 333 |
Drugs and US Patents for PHARMOBEDIENT
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pharmobedient | PAROXETINE HYDROCHLORIDE | paroxetine hydrochloride | TABLET;ORAL | 075716-004 | Mar 8, 2004 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | |||||
| Pharmobedient | ACEBUTOLOL HYDROCHLORIDE | acebutolol hydrochloride | CAPSULE;ORAL | 074288-002 | Apr 24, 1995 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | |||||
| Pharmobedient | AMLODIPINE BESYLATE AND BENAZEPRIL HYDROCHLORIDE | amlodipine besylate; benazepril hydrochloride | CAPSULE;ORAL | 079047-002 | Jul 5, 2011 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for PHARMOBEDIENT
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Pharmobedient | OLUX | clobetasol propionate | AEROSOL, FOAM;TOPICAL | 021142-001 | May 26, 2000 | 6,126,920 | ⤷ Start Trial |
| Pharmobedient | OLUX E | clobetasol propionate | AEROSOL, FOAM;TOPICAL | 022013-001 | Jan 12, 2007 | 6,730,288 | ⤷ Start Trial |
| Pharmobedient | DEMADEX | torsemide | TABLET;ORAL | 020136-001 | Aug 23, 1993 | 4,018,929 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for PHARMOBEDIENT drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Topical Foam | 0.05% | ➤ Subscribe | 2005-06-27 |
| ➤ Subscribe | Inhalation Solution | 0.021% and 0.042% | ➤ Subscribe | 2005-10-19 |
| ➤ Subscribe | Foam | 0.12% | ➤ Subscribe | 2007-08-10 |
| ➤ Subscribe | Delayed-release Tablets | 20 mg | ➤ Subscribe | 2015-06-03 |
International Patents for PHARMOBEDIENT Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Taiwan | 200624124 | ⤷ Start Trial |
| World Intellectual Property Organization (WIPO) | 2006024095 | ⤷ Start Trial |
| Brazil | PI0514806 | ⤷ Start Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for PHARMOBEDIENT Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2563920 | CA 2019 00001 | Denmark | ⤷ Start Trial | PRODUCT NAME: A COMPOUND COMPRISING A MODIFIED OLIGONUCLEOTIDE HAVING A NUCLEOBASE SEQUENCE CONSISTING OF 20 LINKED NUCLEOSIDES ACCORDING TO EP B1 2563920 CLAIM 1 (SEQ ID NO: 80), ...SPECIFICALLY INOTERSEN; AND ITS DERIVATIVES, INCLUDING SODIUM SALTS ...; REG. NO/DATE: EU/1/18/1296 20180710 |
| 1539166 | CA 2013 00059 | Denmark | ⤷ Start Trial | PRODUCT NAME: KOMBINATION AF (A) DEXTROMETHORPHAN OG (B) QUNIDIN, HERUNDER DEXTROMETHORPHANHYDROBROMIDMONOHYDRAT OG QUINIDINSULFATDIHYDRAT; REG. NO/DATE: EU/1/13/833 20130624 |
| 2443246 | SPC/GB21/056 | United Kingdom | ⤷ Start Trial | PRODUCT NAME: ICOSAPENT ETHYL; REGISTERED: UK EU/1/20/1524(FOR NI) 20210329; UK FURTHER MA ON IPSUM 20210329 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.