Lantheus Company Profile
✉ Email this page to a colleague
What is the competitive landscape for LANTHEUS
LANTHEUS has seven approved drugs.
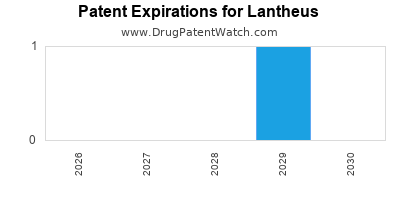
There are thirteen US patents protecting LANTHEUS drugs.
There are one hundred and thirty-five patent family members on LANTHEUS drugs in thirty-two countries and twenty-nine supplementary protection certificates in sixteen countries.
Drugs and US Patents for Lantheus
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lantheus Medcl | GALLIUM CITRATE GA 67 | gallium citrate ga-67 | INJECTABLE;INJECTION | 017478-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | |||||
| Lantheus Medcl | DEFINITY RT | perflutren | INJECTABLE;INTRAVENOUS | 021064-002 | Nov 17, 2020 | RX | Yes | Yes | 11,395,856 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Lantheus Medcl | DEFINITY RT | perflutren | INJECTABLE;INTRAVENOUS | 021064-002 | Nov 17, 2020 | RX | Yes | Yes | 10,022,460 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Lantheus Medcl | DEFINITY | perflutren | INJECTABLE;INTRAVENOUS | 021064-001 | Jul 31, 2001 | RX | Yes | Yes | 10,583,208 | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Lantheus
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Lantheus Medcl | DEFINITY | perflutren | INJECTABLE;INTRAVENOUS | 021064-001 | Jul 31, 2001 | 6,033,645 | ⤷ Start Trial |
| Lantheus Medcl | DEFINITY | perflutren | INJECTABLE;INTRAVENOUS | 021064-001 | Jul 31, 2001 | 5,585,112 | ⤷ Start Trial |
| Lantheus Medcl | ABLAVAR | gadofosveset trisodium | SOLUTION;INTRAVENOUS | 021711-001 | Dec 22, 2008 | 8,394,356 | ⤷ Start Trial |
| Lantheus Medcl | ABLAVAR | gadofosveset trisodium | SOLUTION;INTRAVENOUS | 021711-002 | Dec 22, 2008 | 6,676,929 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for Lantheus Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Israel | 318965 | ⤷ Start Trial |
| Taiwan | I821755 | ⤷ Start Trial |
| Taiwan | I853789 | ⤷ Start Trial |
| China | 115531560 | ⤷ Start Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Lantheus Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0712293 | C300267 | Netherlands | ⤷ Start Trial | PRODUCT NAME: PERFLUTREN BEVATTENDE LIPIDE-MICROBOLLEN; REGISTRATION NO/DATE: EU/1/06/361/001 20060920 |
| 2213652 | 221 2-2015 | Slovakia | ⤷ Start Trial | PRODUCT NAME: FLORBETABEN (18F); REGISTRATION NO/DATE: EU/1/13/906/001 20140224 |
| 2213652 | 504 | Finland | ⤷ Start Trial | |
| 2213652 | C300719 | Netherlands | ⤷ Start Trial | PRODUCT NAME: FLORBETABEN (18F); REGISTRATION NO/DATE: EU/1/13/906 20140220 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.