Last updated: February 20, 2026
What Is Jazz Pharmaceuticals’ Market Position in Its Therapeutic Areas?
Jazz Pharmaceuticals focuses on central nervous system (CNS) disorders, hem-oncology, and sleep medicine. Its portfolio includes branded drugs mainly in narcolepsy, oncology, and rare diseases. As of 2023, Jazz holds a prominent position in narcolepsy treatment through Xyrem (sodium oxybate), which commands significant market share in sleep disorder therapies. It also has a growing presence in oncology with drugs like Zepzelca (lurbinectedin) and Rylaze (asparaginase erwinia chrysanthemi).
Market share estimates place Jazz as the second-largest player in narcolepsy drugs, trailing only major pharmaceutical companies such as Eli Lilly and UCB. Its share of the sleep disorder market exceeds 60% in the U.S., driven by patent protections and reputation for efficacy. In oncology, Jazz’s Zepzelca holds niche but strategic penetration in relapsed small-cell lung cancer (SCLC).
How Does Jazz Pharmaceuticals’ Portfolio Strengths Drive Its Competitive Advantage?
1. Focus on Rare and Niche Indications
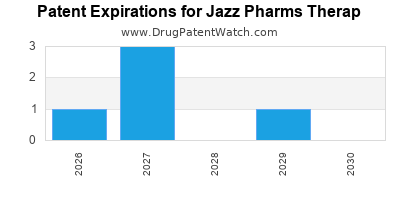
Jazz operates predominantly in orphan drugs and niche markets. This focus reduces direct competition, allows premium pricing, and benefits from supportive regulatory pathways. Its flagship product, Xyrem, benefits from orphan drug status, granting market exclusivity until at least 2028.
2. Proprietary Formulation and Delivery
Jazz emphasizes unique formulations including the sodium oxybate delivery system for Xyrem, which ensures consistent bioavailability. Its R&D has facilitated formulation improvements, increasing patient adherence and reducing side effects.
3. Built-In Market Access and Reimbursement Strategies
Jazz’s established relationships with payers and providers enable faster reimbursement cycles. For Xyrem, the company offers comprehensive support programs, boosting compliance and minimizing generic erosion risks pre-patent expiry.
4. Strategic Acquisitions and Collaborations
Jazz acquires or collaborates to expand its portfolio. The 2018 acquisition of Clavis Pharma added oncology assets, and partnerships with biotech firms expand its pipeline with early-stage candidates.
5. Regulatory and Patent Protections
Market exclusivity stems from patents and orphan drug designations. The process for generic competition is delayed until patent expiration, giving Jazz a competitive edge in high-margin segments.
What Are Key Strategic Opportunities and Risks for Jazz Pharmaceuticals?
Opportunities
- Pipeline Expansion in CNS and Oncology: Jazz's pipeline includes candidates targeting rare CNS disorders and unmet oncology needs, such as JDQ443, a potential first-in-class gamma-secretase inhibitor for ALK+ NSCLC.
- Global Market Penetration: Increasing presence in emerging markets with high unmet needs offers upside, especially in China and India, where orphan drugs are gaining regulatory incentives.
- Product Line Diversification: Developing oral formulations of existing drugs to expand patient access and reduce dependence on injectable products.
Risks
- Patent Expirations: Xyrem’s patent is scheduled to expire by 2028, risking generic erosion and revenue decline.
- Market Competition: Eli Lilly’s upcoming licensed Xyrem biosimilar treatments could diminish Jazz’s market share in sleep disorders.
- Regulatory Challenges: Expedited approval pathways might favor generic entrants, reducing Jazz’s exclusivity period.
- Pipeline Uncertainties: Clinical trial failures or delays in new candidate approvals could reduce future growth potential.
How Does Jazz Pharmaceuticals Compare to Its Major Competitors?
| Company |
Market Focus |
Key Products |
Revenue (2022) |
Market Share in Sleep Disorders |
R&D Investment (2022) |
Patent Expiry Risk |
| Eli Lilly |
CNS, Oncology |
Xyrem (biosimilar), Lilly Patents pending |
$28B |
Largest in narcolepsy |
$4.2B |
2028 |
| UCB |
CNS, Epilepsy |
Vimpat, Nayzilam |
$6.2B |
Second-largest in sleep meds |
$1.4B |
2027 |
| Jazz Pharmaceuticals |
CNS, Hem-oncology, Sleep disorders |
Xyrem, Zepzelca, Rylaze |
$2.9B |
Second in narcolepsy |
$500M |
2028 (Xyrem) |
| Convatec |
Pharma-Device Focus |
Multiple niche pharma devices |
$2B |
Limited |
N/A |
N/A |
Jazz retains a competitive advantage in niche markets due to its focus on rare diseases and orphan drugs, although it faces threats from biosimilars and generics in its leading products.
What Strategic Moves Should Jazz Consider for Sustained Growth?
- Invest in pipeline diversification, specifically in early-stage or accelerated approval indications.
- Strengthen global presence in emerging markets with regulatory incentives for rare diseases.
- Enhance formulation technologies to improve adherence and reduce treatment costs.
- Develop strategic collaborations to offset R&D risks and expand therapeutic reach.
Key Takeaways
- Jazz Pharmaceuticals commands a leading position in narcolepsy therapies with Xyrem, supported by strong patent protections until at least 2028.
- The company’s ASIC focus on niche markets shields it from direct competition but faces patent expiry risks.
- Strategic pipeline expansion and global market penetration are critical for ongoing growth but are counterbalanced by regulatory and market access risks.
- Competitor analysis reveals Eli Lilly and UCB as primary rivals, especially in sleep disorders and CNS markets.
- Maintaining innovation in formulations and expanding indications will be vital for Jazz’s market relevance.
FAQs
1. When will Jazz’s flagship product Xyrem face generic competition?
Patent protection is expected to expire in 2028, opening the market to biosimilars and generics.
2. What are Jazz’s primary therapeutic areas?
Central nervous system disorders (notably narcolepsy), hem-oncology, and rare diseases.
3. How does Jazz differentiate itself in the sleep disorder market?
Through proprietary formulations, orphan drug exclusivities, and patient adherence strategies.
4. What are the main competitive threats to Jazz?
Patent expirations, biosimilar emergence, regulatory changes, and clinical trial outcomes.
5. Which emerging markets are strategic for Jazz?
China, India, and Latin America, due to increasing orphan drug regulations and unmet need.
References
[1] U.S. Food and Drug Administration. (2023). FDA-approved drugs in 2023.
[2] Jazz Pharmaceuticals. (2022). Annual report.
[3] IQVIA. (2023). Market analysis for sleep disorder and oncology drugs.
[4] EvaluatePharma. (2022). Global pharmaceutical pipeline and market projections.
[5] IMS Health. (2022). Prescription drug market share analysis.