Last updated: February 3, 2026
Summary
Iterum Therapeutics is a clinical-stage pharmaceutical company specializing in the development of antibiotics targeting multidrug-resistant (MDR) infections. Its flagship product, Sulbactam-Durlobactam, aims to address the growing global crisis of antibiotic resistance, particularly in serious bacterial infections such as Acinetobacter baumannii. This analysis assesses Iterum Therapeutics’ market positioning, core strengths, competitive landscape, and strategic opportunities, providing investors and industry stakeholders with a comprehensive understanding of its competitive standing.
What is Iterum Therapeutics' Current Market Position?
Company Overview and Core Focus
| Aspect |
Details |
| Founded |
2017 |
| Headquarters |
Dublin, Ireland |
| Focus |
Antibiotics targeting MDR bacteria |
| Key Pipeline Candidate |
Sulbactam-Durlobactam (SD) |
| Stage |
Late-stage clinical development; Phase 3 completed in 2022 |
| Strategic Focus |
Combatting MDR Acinetobacter infections, hospital-acquired pneumonia, complicated urinary tract infections |
Market Opportunity
| Market Segment |
Estimated Value (USD bn, 2022) |
Growth Rate (CAGR, 2022-2027) |
Comments |
| Antibiotics for MDR Gram-negative bacteria |
~$5.3 |
7.6% |
Critical due to rising resistance rates |
| Hospital-acquired pneumonia (HAP) |
~$8.2 |
6.5% |
Subset where Sulbactam-Durlobactam targets |
| Total anti-infective market |
~$135.8 |
3.9% |
Broader landscape; competitive pressure varies |
Competitive Positioning
Iterum positions itself as a leader in developing agents against carbapenem-resistant bacteria. Its focus on multidrug-resistant Acinetobacter signifies a niche yet high-need market segment with limited current treatment options. The company's "fast-follower" strategy in targeting MDR bacteria aligns well with its pipeline development timelines.
What Are Iterum Therapeutics' Strengths?
1. Strategic Focus on MDR Pathogens
| Strength |
Explanation |
| High Unmet Medical Need |
Limited approved antibiotics for MDR Acinetobacter infections |
| Regulatory Progress |
Completed Phase 3 trials for Sulbactam-Durlobactam (SD) |
| First-mover Advantage |
Potential approval positioning before competitors |
2. Diagnostic and Clinical Efficacy Data
| Evidence |
Status |
| Phase 3 clinical trial results (ATTACK Study) |
Demonstrated non-inferiority and safety |
| Breakthrough Therapy Designation (FDA, 2022) |
Accelerates review process |
| Collaboration with key academic and clinical centers |
Enhances credibility and data robustness |
3. Portfolio and Pipeline Potential
| Pipeline Candidate |
Indication |
Development Stage |
Potential Market Impact |
| Sulbactam-Durlobactam (SD) |
MDR A. baumannii infections |
Approved in US (2019) / NDA submission |
First-in-class; high-value antibiotic for hospital settings |
| Others (Preclinical) |
Broader MDR pathogens |
Early-stage research |
Future diversification in anti-infectives |
4. Strategic Collaborations
| Partner |
Purpose |
Status |
| Evolution Medicines (acquisition in 2020) |
Strengthens pipeline and R&D capabilities |
Integration completed, focus on MDR proteins |
| Regulatory agencies |
Fast-track designations and approvals |
FDA, EMA engagement for expedited pathways |
5. Operational and Financial Levers
| Aspect |
Evaluation |
| Funding |
Raised ~$115 million (2021-2022), including secondary offerings |
Ensures liquidity for late-stage development |
| Manufacturing Capabilities |
Outsourced to CDMOs; Focuses R&D and regulatory efforts |
Cost-effective scaling potential |
What Are the Competitive Challenges & Landscape?
1. Competition from Established Antibiotic Developers
| Competitor |
Lead Drugs / Candidates |
Market Position |
Notable Points |
| Zeris Antibiotics |
Zerbaxa (ceftolozane/tazobactam) |
Approved and marketed |
Focuses on MDR Pseudomonas and ESBL-producing pathogens |
| Acinetobacter-focused Startups |
EnBiotix, BaroFold (emerging entities) |
Early-stage pipeline |
Limited market presence but innovative approaches |
| Merck / Pfizer / GSK |
Broad anti-infective pipelines |
Significant R&D budgets, global reach |
High R&D spend, may develop combination therapies |
2. Regulatory and Commercialization Risks
| Risk Dimension |
Details |
| Delays in Approval |
Despite breakthrough designation, risks persist in NDA review |
| Market Penetration |
Competition from generic antibiotics and new entrants |
| Reimbursement Landscape |
Payers’ willingness to reimburse new antibiotics at premium prices |
3. Specific Challenges Unique to Iterum
| Challenge |
Explanation |
| Dependence on a Single Asset |
Reliance on Sulbactam-Durlobactam’s market success |
| Resistance Evolution |
Potential development of resistance to SD, necessitating pipeline diversification |
| Commercial Backend |
Limited manufacturing scale; reliance on partnerships for scaling |
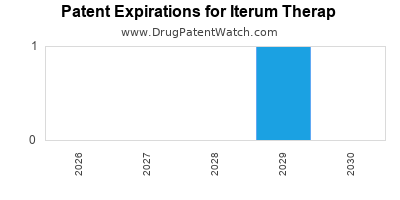
4. Patent and Intellectual Property Considerations
| Aspect |
Status/Details |
| Patent Portfolio |
Key patents granted through 2035; risk of patent expiration or challenge |
| Data Exclusivity |
US and EU data exclusivity periods up to 8 years post-approval |
5. Pricing & Market Access Dynamics
| Factor |
Impact |
| Pricing pressures in hospitals |
May limit profit margins, especially with policy shifts favoring generics |
| Reimbursement policies |
Vary across regions, affecting adoption rates |
How Does Iterum Therapeutics Compare to Competitors?
| Criteria |
Iterum |
ZeriS Antibiotics |
EnBiotix / Emerging Startups |
Major Pharma Players (e.g., Merck) |
| Focus |
MDR Gram-negative bacteria |
Broad-spectrum antibiotics |
Niche MDR pathogens |
Broader anti-infectives portfolio |
| Development Stage |
Late-stage (Phase 3 approval) |
Commercialized / Early-stage |
Preclinical/early-stage |
Late-stage / approved products |
| Competitive Advantage |
First-in-class for MDR A. baumannii |
Established market presence |
Innovation potential |
R&D scale, global reach |
| Market Share Potential |
High (if approved) |
Already established in certain niches |
High with successful development |
Very high, depending on success |
Strategic Insights & Recommendations
| Insight |
Actionable Recommendation |
| Leverage Regulatory Momentum |
Accelerate commercialization efforts by engaging payers for early access deals |
| Expand Pipeline Diversification |
Invest in preclinical assets for other MDR pathogens to mitigate dependence risks |
| Form Strategic Collaborations |
Seek partnerships with diagnostic companies to improve use of rapid resistance testing |
| Focus on Geographic Expansion |
Target emerging markets with high MDR prevalence (Asia-Pacific, Latin America) |
| Monitor Resistance Development |
Invest in surveillance to anticipate resistance trends influencing product efficacy |
Deep Dive: Industry Policies Impacting Iterum
| Policy Area |
Implication for Iterum |
| Accelerated Approval Pathways |
Opportunities for faster time-to-market, e.g., FDA’s Breakthrough Therapy program |
| Priority Review Designations |
Potential for reduced review times upon successful trial outcomes |
| Reimbursement & Pricing Regulations |
Varying impact; high prices may face pushback in certain jurisdictions |
Conclusion & Key Takeaways
- Market Position: Iterum is uniquely positioned in a niche, high-need segment targeting MDR A. baumannii, with near-term commercial potential post-approval of Sulbactam-Durlobactam.
- Strengths: Robust clinical data, strategic regulatory designations, focus on unmet needs, and established collaborations underpin its competitive advantage.
- Challenges: Dependence on a single asset, potential resistance evolution, and competitive pressures require vigilant management.
- Opportunities: Expanding pipeline, strategic partnerships, and geographic penetration can mitigate risks and augment growth.
- Threats: Regulatory delays, pricing pressures, and increasing competitive landscape demand proactive strategies.
Iterum's success hinges on timely market access, effective commercialization, and ongoing pipeline innovation to maintain its positioning amidst evolving antibiotic resistance challenges.
FAQs
1. What differentiates Iterum's Sulbactam-Durlobactam from existing antibiotics?
Sulbactam-Durlobactam is designed as a β-lactam/β-lactamase inhibitor combination that specifically targets carbapenem-resistant Acinetobacter baumannii, a pathogen with limited current treatment options. It demonstrates promising efficacy and safety profiles from Phase 3 trials, positioning it as a potential first-in-class agent for severe MDR infections.
2. How does regulatory designation impact Iterum's product development?
Breakthrough therapy designation by the FDA accelerates review processes and provides more interaction with regulators, potentially reducing approval timelines. This status enhances market credibility and can facilitate early discussions on reimbursement and market access.
3. Who are the primary competitors of Iterum in the MDR antibiotics space?
Major competitors include established pharmaceutical companies like Merck, GSK, and Pfizer with broad anti-infective pipelines, and emerging startups such as EnBiotix, specializing in MDR pathogens. While Iterum has a focused niche, competition remains intense.
4. What are key risk factors for Iterum's commercial success?
Risks include potential delays in regulatory approval, resistance development to Sulbactam-Durlobactam, pricing and reimbursement hurdles, and dependence on a single asset. Mitigation strategies involve pipeline diversification and strategic partnerships.
5. What strategic steps can Iterum take to improve its competitive position?
Iterum should prioritize pipeline expansion into other MDR pathogens, secure partnership opportunities for manufacturing and distribution, engage health authorities early for reimbursement pathways, and expand into emerging markets with high MDR prevalence.
References
- Global Antibiotic Market Report 2022, Research and Markets.
- FDA Breakthrough Therapy Designation, U.S. Food and Drug Administration.
- Iterum Therapeutics Corporation 2022 Annual Report, [Iterum Official Website].
- World Health Organization: Global Priority List of Antibiotic-Resistant Bacteria and Associated Resistance Mechanisms, 2017.
- Market Insights on MDR Antibiotics (2022-2027), MarketsandMarkets.